FHL Transfer for Achilles Tendinosis: An Intraoperative Masterclass
Key Takeaway
Step into the operating room for a comprehensive masterclass on Flexor Hallucis Longus (FHL) transfer for Achilles tendinosis. This guide covers detailed anatomy, meticulous preoperative planning, and a granular, real-time surgical execution from incision to closure. We'll explore critical pearls, potential pitfalls, and essential postoperative rehabilitation for optimal patient outcomes in this challenging condition.
Comprehensive Introduction and Patho-Epidemiology
Chronic Achilles tendinosis represents one of the most structurally debilitating and therapeutically challenging conditions encountered by the orthopedic foot and ankle surgeon. Far from a simple inflammatory "tendinitis," this pathology is defined by a profound, progressive mucoid degeneration of the tendon's microscopic architecture. It is a failed healing response characterized by the disorganized proliferation of tenocytes, an insidious shift from resilient Type I collagen to biomechanically inferior Type III collagen, and the chaotic ingrowth of non-functional neovessels and nociceptive nerve fibers. This complex interplay of mechanical overload and intrinsic biologic failure ultimately renders the tendon structurally deficient, highly symptomatic, and acutely vulnerable to catastrophic rupture.
The epidemiological profile of chronic Achilles tendinosis typically highlights patients in their fourth to sixth decades of life. These individuals often present with a frustratingly prolonged history of posterior heel pain, progressive functional decline, and an inability to participate in previously tolerated athletic or daily activities. The natural history of this condition frequently follows a predictable continuum: it may originate as a localized retrocalcaneal bursitis or paratenonitis, subsequently evolving into midsubstance or insertional tendinosis as the relentless mechanical impingement from a prominent calcaneal tuberosity (Haglund’s deformity) takes its toll. Left unaddressed, the degenerative cascade accelerates, leading to macroscopic intrasubstance tearing and eventual mechanical failure.
For patients who have exhausted a rigorous, protracted course of nonoperative modalities—including eccentric loading protocols, immobilization, orthotic management, and biologic injections—surgical intervention becomes the definitive pathway to restoring function. When preoperative imaging and intraoperative assessment dictate the necessary excision of greater than 50% of the diseased Achilles tendon cross-sectional area, primary repair alone is biomechanically insufficient. In these advanced scenarios, the Flexor Hallucis Longus (FHL) transfer has emerged as the gold standard for augmentation.
The FHL tendon is uniquely suited for this role due to its robust tensile strength, its axis of contractility which closely mirrors that of the native Achilles, and its phase-matched firing pattern during the human gait cycle. Furthermore, the low-lying, highly vascularized muscle belly of the FHL provides an invaluable biologic advantage. When transferred into the posterior ankle, this muscle belly drapes directly over the relatively avascular "watershed" zone of the Achilles tendon, delivering a rich, localized blood supply that aggressively promotes host tissue healing, mitigates the risk of postoperative wound necrosis, and substantially enhances the overall durability of the reconstruction.
Detailed Surgical Anatomy and Biomechanics
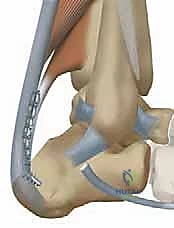
A profound, three-dimensional mastery of hindfoot anatomy is the absolute prerequisite for executing a safe and biomechanically sound FHL transfer. The Achilles tendon, the largest and strongest tendon in the human body, is formed by the confluence of the gastrocnemius and soleus aponeuroses. Unlike tendons enveloped by a true synovial sheath, the Achilles is surrounded by a highly vascularized paratenon composed of a single layer of cells. This paratenon is the primary conduit for the tendon's extrinsic blood supply, which is supplemented distally by osseous branches from the calcaneal insertion and proximally by intramuscular branches.
Crucially, injection studies have consistently demonstrated a relative zone of hypovascularity—the "watershed" area—located approximately 2 to 6 centimeters proximal to the calcaneal insertion. This precarious vascular anatomy is the primary reason this specific segment is disproportionately susceptible to degenerative tendinosis and spontaneous rupture. Furthermore, the osteology of the posterior calcaneus plays a pivotal mechanical role. A posterosuperior prominence, or Haglund’s deformity, acts as a mechanical fulcrum, impinging directly against the anterior aspect of the Achilles tendon during ankle dorsiflexion, thereby initiating and perpetuating the degenerative cycle at the enthesis.

The Flexor Hallucis Longus muscle originates expansively from the inferior two-thirds of the posterior fibular surface, the lower interosseous membrane, and the intermuscular septum. It is a powerful bipennate muscle whose belly extends remarkably far distally, often reaching the level of the tibiotalar joint line. As the FHL tendon courses distally, it traverses the fibro-osseous tarsal tunnel, passing deep to the flexor retinaculum, posterior to the medial malleolus, and grooving the posterior aspect of the talus and the undersurface of the sustentaculum tali. It then proceeds into the plantar vault, crossing dorsal to the flexor digitorum longus (FDL) at the Master Knot of Henry, before finally inserting into the base of the distal phalanx of the hallux.
Navigating the deep posterior compartment to harvest the FHL requires meticulous respect for the adjacent neurovascular structures. The tibial nerve and the posterior tibial artery, along with its venae comitantes, lie immediately medial and superficial to the FHL within the tarsal tunnel. The medial calcaneal nerve branches proximally from the tibial nerve and crosses the surgical field superficially. The surgeon must maintain a precise plane of dissection, identifying and gently retracting the neurovascular bundle medially while incising the deep transverse fascia to expose the FHL muscle belly. Biomechanically, the FHL is an ideal transfer candidate; it is the second strongest plantarflexor of the foot (behind the triceps surae), fires synergistically with the Achilles during the push-off phase of gait, and possesses an axis of pull that perfectly replicates the vector required for powerful plantarflexion.
Exhaustive Indications and Contraindications
Patient selection is the cornerstone of a successful outcome in Achilles reconstructive surgery. The decision to proceed with an FHL transfer is not made lightly; it is reserved for patients exhibiting severe, intractable pathology that has proven refractory to comprehensive, prolonged conservative management. The primary intraoperative indication for FHL augmentation is the necessity to debride more than 50% of the native Achilles tendon's cross-sectional area to reach healthy, viable collagen. Attempting a primary tubularization or repair of a tendon compromised to this degree invites a high probability of catastrophic postoperative rupture and persistent pain.
Beyond the intraoperative assessment of tendon viability, specific clinical scenarios strongly favor FHL transfer. These include chronic, neglected Achilles tendon ruptures with a gap exceeding 3 to 5 centimeters, where primary end-to-end apposition is impossible even with maximal plantarflexion. Additionally, older, low-demand patients with severe insertional calcific tendinosis and massive retrocalcaneal exostoses benefit immensely from the robust biologic and mechanical reinforcement provided by the FHL. Patients presenting with recurrent ruptures following previous primary repairs, or those with significant soft tissue compromise requiring local vascularized muscle coverage, are also prime candidates for this procedure.
Conversely, absolute contraindications must be rigorously respected to prevent disastrous complications. Active local or systemic infection is an absolute barrier to any elective reconstructive procedure. Severe peripheral arterial disease (PAD) fundamentally compromises the healing potential of the posterior heel skin—an area already notorious for wound breakdown—making surgical intervention exceptionally hazardous. Patients who are non-ambulatory or possess profound neurological deficits precluding rehabilitation are not suitable candidates. Relative contraindications include poorly controlled diabetes mellitus, heavy active tobacco use, and advanced Charcot neuroarthropathy, all of which substantially elevate the risk of postoperative complications and require extensive preoperative optimization.
| Category | Specific Factors |
|---|---|
| Primary Indications | Intraoperative debridement requiring >50% excision of Achilles cross-section; Chronic ruptures with >3-5 cm gap; Severe insertional calcific tendinosis with massive Haglund's deformity; Failed previous primary Achilles repair. |
| Patient Demographics | Typically 40+ years old; Exhausted >6 months of dedicated non-operative treatment (eccentric loading, immobilization, biologics); Intractable pain limiting ADLs. |
| Absolute Contraindications | Active local or systemic infection; Severe Peripheral Arterial Disease (PAD); Non-ambulatory status; Inability to comply with strict, prolonged postoperative rehabilitation protocols. |
| Relative Contraindications | Poorly controlled Diabetes Mellitus (HbA1c > 8.0); Active, heavy tobacco smoking (requires cessation); Advanced systemic inflammatory arthropathies; Charcot neuroarthropathy. |
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning begins with a comprehensive clinical examination. The surgeon must meticulously palpate the Achilles tendon to identify nodular thickening, structural gaps, and areas of maximal point tenderness. The Silfverskiöld test is mandatory to differentiate between isolated gastrocnemius contracture and combined gastrocnemius-soleus complex tightness, although in the setting of chronic tendinosis, the inherent structural lengthening of the diseased tendon often masks underlying contractures. Assessment of hindfoot alignment is critical; a severe varus or valgus deformity may require concomitant osteotomies to protect the transferred tendon from asymmetrical mechanical overload. The integrity of the overlying skin must be scrutinized for prior incisions, thinning, or vascular compromise.
Radiographic evaluation is standard and essential. Weight-bearing lateral radiographs of the foot and ankle are scrutinized to assess the morphology of the calcaneal tuberosity, quantify the extent of intratendinous calcification, and identify the presence of a Haglund’s deformity. The Fowler-Philip angle and parallel pitch lines can be utilized, though clinical correlation remains paramount. When significant calcification is present at the insertion, the surgeon must plan for a substantial calcaneal ostectomy and subsequent reattachment of the Achilles footprint.
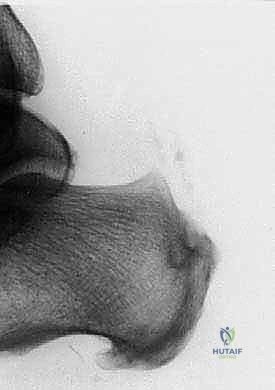
Magnetic Resonance Imaging (MRI) is the definitive modality for characterizing the extent of soft tissue degeneration. Sagittal and axial T1 and T2-weighted sequences provide a precise map of the diseased tissue. The surgeon evaluates the MRI for increased intratendinous signal intensity, fusiform thickening, partial interstitial tearing, and the exact longitudinal extent of the pathology. This advanced imaging allows the surgical team to accurately estimate the volume of tendon requiring debridement, thereby confirming the necessity for an FHL transfer before the first incision is made.
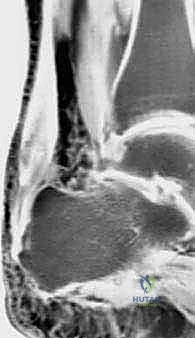
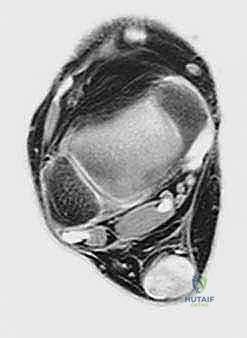
Patient positioning in the operating room is critical for optimal exposure and workflow. The patient is placed in the prone position following the administration of general or regional anesthesia. Careful padding of all bony prominences, particularly the ulnar nerves, knees, and contralateral foot, is mandatory. Chest rolls are utilized to facilitate unencumbered ventilation. A well-padded thigh tourniquet is applied to provide a bloodless surgical field, typically inflated to 250-300 mmHg after exsanguination. Crucially, the operative leg is positioned such that the foot extends freely over the edge of the operating table. This allows the surgeon to dynamically assess ankle dorsiflexion and precisely tension the FHL transfer with the foot in the desired resting equinus position. A C-arm fluoroscopy unit is positioned to allow for unobstructed lateral imaging during the calcaneal ostectomy and hardware placement.
Step-by-Step Surgical Approach and Fixation Technique
Incision and Initial Exposure
The procedure commences with a meticulously planned posteromedial incision. A longitudinal incision, typically 10 to 15 centimeters in length, is placed approximately 1 to 2 centimeters medial to the medial border of the Achilles tendon. This specific placement avoids placing the surgical scar directly over the posterior prominence of the heel, thereby mitigating postoperative shoe wear irritation, while simultaneously providing the most direct, unhindered access to the deep posterior compartment for the FHL harvest.

The dissection must be carried down sharply and directly through the subcutaneous tissues to the paratenon. The creation of broad subcutaneous skin flaps is strictly prohibited. The vascular supply to the posterior heel skin is notoriously tenuous, relying on a delicate subdermal plexus. Undermining these flaps severely compromises this blood supply, exponentially increasing the risk of catastrophic wound dehiscence and full-thickness skin necrosis.
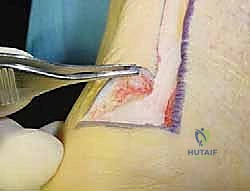
In cases of severe insertional tendinosis where extensive exposure of the calcaneal footprint is required, the longitudinal incision may be modified into an L-shaped or J-shaped extension distally, curving laterally along the superior border of the calcaneal tuberosity. Once the paratenon is reached, it is incised longitudinally, exposing the underlying diseased Achilles tendon.

Achilles Tendon Debridement and Calcaneal Ostectomy
Upon exposing the Achilles tendon, the surgeon is immediately confronted with the macroscopic evidence of chronic tendinosis. The healthy, glistening, tightly bundled, longitudinally oriented collagen fibers are replaced by a thickened, amorphous, dull, and hyperemic mass of tissue. This is classically described as having a "codfish-flesh" or "crab-meat" appearance. Intratendinous calcifications, ranging from fine stippling to massive, rock-hard ossicles, are frequently encountered embedded within the diseased matrix.
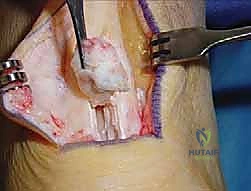
Aggressive and unapologetic debridement is paramount. The surgeon utilizes a scalpel to systematically excise all degenerative, mucoid, and calcified tissue. The goal is to resect back to healthy, viable tendon margins, characterized by normal color, firm texture, and distinct fascicular architecture. It is exceedingly common to remove more than 50% of the tendon's cross-sectional area; leaving diseased tissue behind in an attempt to preserve continuity is a primary cause of persistent postoperative pain and surgical failure.
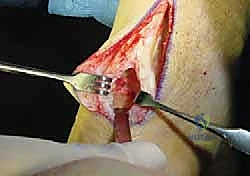
Following the soft tissue debridement, attention is directed to the calcaneus. The retrocalcaneal bursa, which is typically hypertrophied and inflamed, is meticulously excised. This exposes the posterosuperior calcaneal prominence (Haglund’s deformity). Utilizing an oscillating saw or a sharp osteotome, a generous calcaneal ostectomy is performed. The cut is directed from posterosuperior to anteroinferior, ensuring the complete removal of the impingement lesion while preserving the structural integrity of the plantar calcaneal tuberosity.
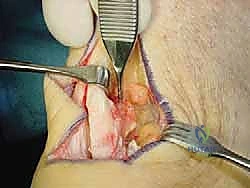
The resected bone is removed, and the remaining osseous surface is smoothed with a rasp or burr. Fluoroscopy is utilized at this stage to confirm the adequacy of the resection and ensure no sharp bony edges remain that could abrade the transferred tendon.
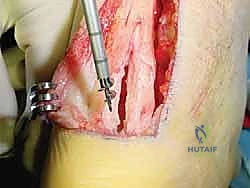
FHL Muscle Identification and Distal Harvest
With the Achilles debridement and calcaneal ostectomy complete, the critical phase of FHL harvesting begins. The deep transverse fascia of the leg, which separates the superficial and deep posterior compartments, is identified anterior and medial to the Achilles tendon. This fascia is carefully incised longitudinally. Immediately deep to this fascial layer lies the robust, low-lying muscle belly of the Flexor Hallucis Longus.
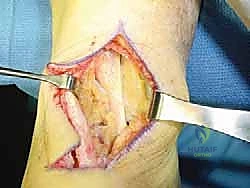
The surgeon must exercise extreme caution during this exposure. The tibial neurovascular bundle is situated immediately medial to the FHL muscle and tendon. Utilizing blunt dissection, the neurovascular bundle is identified, mobilized, and gently retracted medially using a blunt retractor (such as a right-angle or Senn retractor). The FHL muscle belly is then traced distally as it transitions into its tendinous portion and enters the fibro-osseous tarsal tunnel.
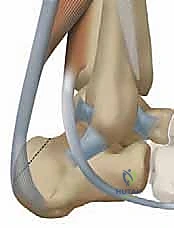
To maximize the length of the harvested tendon, the flexor retinaculum forming the roof of the tarsal tunnel is partially released. The foot is maximally plantarflexed, and the hallux is forcefully dorsiflexed to draw the FHL tendon proximally into the wound. While a double-incision technique (with a second incision in the medial midfoot at Henry’s knot) can be utilized for maximal length, a single posterior incision approach is typically sufficient for most augmentations and minimizes midfoot morbidity.
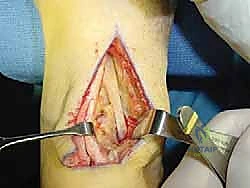
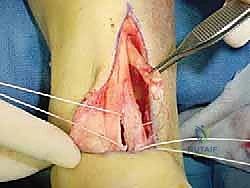
The tendon is sharply transected as far distally as possible within the tarsal tunnel. A heavy, non-absorbable locking whipstitch (e.g., #2 FiberWire or similar high-tensile suture) is immediately placed into the distal stump of the harvested FHL tendon to secure it and prepare it for transfer.
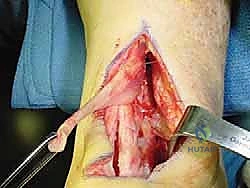
FHL Transfer, Tensioning, and Fixation
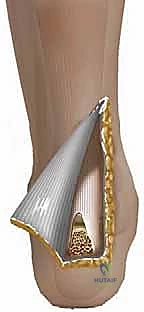
The harvested FHL tendon is now prepared for transfer. The rich, vascular muscle belly of the FHL is pulled distally into the defect left by the Achilles debridement. This muscle belly is deliberately draped over the relatively avascular watershed zone of the remaining Achilles tendon, providing a powerful biologic stimulus for neovascularization and healing.
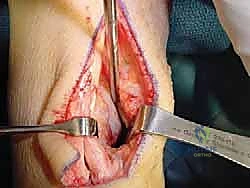
Fixation of the FHL tendon can be achieved through several techniques, including a transosseous tunnel, bio-tenodesis screw fixation into the calcaneus, or a side-to-side anastomosis with the remaining Achilles tendon. For a robust, rigid fixation, a bio-tenodesis technique is highly favored. A guide pin is placed into the superior aspect of the calcaneal tuberosity, aiming plantarly and slightly anteriorly. A blind tunnel is reamed over the guide pin, typically matching the diameter of the harvested FHL tendon (usually 7 to 9 mm).

The whipstitched FHL tendon is passed into the calcaneal tunnel. Proper tensioning of the transfer is the most critical biomechanical step of the procedure. The ankle is positioned in approximately 15 to 20 degrees of resting equinus (plantarflexion), matching the cascade of the contralateral limb. With the foot held rigidly in this position, maximal tension is applied to the FHL tendon, pulling it deeply into the socket.
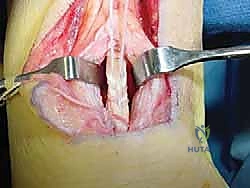
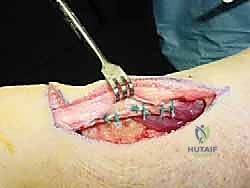
While maintaining this strict tension, an interference screw (typically PEEK or biocomposite) is advanced into the tunnel, securing the tendon against the cortical bone. Fluoroscopy is utilized to confirm the trajectory and final seating of the interference screw.