Elbow Arthroscopy & Mini-Arthrotomy for Capitellar OCD and Panner's Disease: A Masterclass
Key Takeaway
Join us in the OR for a masterclass on elbow arthroscopy and mini-arthrotomy for capitellar osteochondritis dissecans (OCD) and Panner's disease. We'll meticulously cover patient selection, anatomical considerations, step-by-step intraoperative execution, crucial pearls and pitfalls, and comprehensive postoperative management to ensure optimal outcomes for these challenging pediatric and adolescent conditions.
Comprehensive Introduction and Patho-Epidemiology
Osteochondritis dissecans (OCD) of the capitellum and its developmental counterpart, Panner's disease, represent a spectrum of lateral compartment elbow pathology frequently encountered in the young, athletic population. These conditions are characterized by a focal, idiopathic alteration in the subchondral bone of the capitellum, which can subsequently compromise the overlying articular cartilage. While they share a common anatomical location and are both exacerbated by repetitive radiocapitellar microtrauma, their distinct epidemiological profiles, pathophysiological mechanisms, and natural histories dictate vastly different clinical management strategies. Understanding these nuances is critical for the orthopedic surgeon, as inappropriate intervention can lead to lifelong elbow dysfunction, whereas timely, well-executed surgical management—such as advanced arthroscopy or mini-arthrotomy—can restore joint congruity and allow a return to high-level athletics.
Panner's disease is classically defined as an osteochondrosis of the capitellar ossific nucleus. It predominantly affects preadolescent children, typically boys between the ages of 6 and 10 years. The etiology is fundamentally tied to a transient disruption of the vascular supply to the developing epiphysis, compounded by the repetitive compressive forces inherent in youth sports. Because the capitellum relies on a precarious, end-arterial blood supply during this developmental window, it is highly susceptible to ischemic necrosis. However, the natural history of Panner's disease is overwhelmingly benign. With strict activity modification and brief periods of immobilization, the ossific nucleus undergoes predictable stages of fragmentation, revascularization, and eventual reossification. Surgical intervention is exceedingly rare and is generally reserved for atypical cases presenting with recalcitrant mechanical symptoms due to loose body formation.
In stark contrast, osteochondritis dissecans (OCD) of the capitellum manifests in adolescents, typically between the ages of 10 and 17, whose capitellar ossification centers have already matured or fused. This condition is intrinsically linked to repetitive valgus overload, most commonly seen in overhead throwing athletes (such as baseball pitchers) and upper-extremity weight-bearing athletes (such as gymnasts). The repetitive valgus stress places immense compressive and shear forces across the radiocapitellar joint. Over time, this repetitive microtrauma overwhelms the reparative capacity of the subchondral bone, leading to focal avascular necrosis, microfracture, and eventually structural failure. Unlike Panner's disease, capitellar OCD has a high propensity for progression. If left untreated, the subchondral bone failure leads to the destabilization, separation, and eventual detachment of the overlying articular cartilage, resulting in intra-articular loose bodies and early-onset osteoarthritis.
The pathogenesis of both conditions underscores the vulnerability of the lateral elbow compartment to biomechanical stress. The capitellum's blood supply is derived from descending extraosseous branches of the brachial artery, which enter posteriorly and traverse anteriorly. This creates a relative "watershed" zone in the anterior and central portions of the capitellum—the exact areas subjected to maximum radiocapitellar compression during throwing and weight-bearing. When a patient presents with vague, activity-related lateral elbow pain, a high index of suspicion must be maintained. Early identification of capitellar OCD, prior to articular surface collapse, offers the best opportunity for joint preservation techniques, whereas advanced lesions necessitate complex reconstructive procedures such as osteochondral autograft transfer.
Detailed Surgical Anatomy and Biomechanics
A profound understanding of elbow osteology, ligamentous constraints, and neurovascular topography is the foundation of safe and effective elbow arthroscopy. The elbow joint is a highly congruent, complex synovial hinge consisting of three distinct articulations: the ulnohumeral joint, the radiocapitellar joint, and the proximal radioulnar joint. For capitellar pathology, our focus is primarily on the radiocapitellar articulation. The capitellum is the hemispherical articular eminence of the lateral distal humerus, oriented anteriorly and inferiorly. It articulates with the concave fovea of the radial head. This articulation is not merely a hinge; it functions as a trochoid joint, acting as a secondary stabilizer to valgus stress and facilitating complex multi-planar motions including flexion, extension, pronation, and supination.
The biomechanics of the throwing motion directly implicate the radiocapitellar joint in the pathogenesis of OCD. During the late cocking and early acceleration phases of throwing, the elbow is subjected to tremendous valgus torque. The primary restraint to this torque is the anterior bundle of the medial collateral ligament (MCL). However, up to 30% of this valgus restraint is provided by the radiocapitellar articulation acting as a bony buttress. In athletes with subtle MCL insufficiency or those who simply generate forces exceeding physiological limits, the radiocapitellar joint experiences severe compressive and shear stress. This "valgus extension overload" syndrome concentrates forces squarely on the relatively avascular anterocentral capitellum, leading to the characteristic subchondral fatigue failure seen in OCD.
Navigating the lateral compartment surgically requires meticulous respect for the surrounding neurovascular structures. The lateral portal placements rely on exploiting safe muscular intervals, specifically the plane between the anconeus and the extensor carpi ulnaris (ECU) or the extensor carpi radialis brevis (ECRB). The anconeus, originating from the posterior aspect of the lateral epicondyle, serves as an excellent landmark. Deep to these muscular layers lies the lateral collateral ligament (LCL) complex, specifically the lateral ulnar collateral ligament (LUCL), which must be protected to prevent iatrogenic posterolateral rotatory instability (PLRI).
The most critical neurovascular structure at risk during lateral elbow arthroscopy and capitellar drilling is the posterior interosseous nerve (PIN). The PIN, the deep motor branch of the radial nerve, courses anterior to the radiocapitellar joint before diving into the arcade of Frohse within the supinator muscle. The nerve wraps around the radial neck, placing it in immediate proximity to the lateral capsule. Any arthroscopic portal placed too far anteriorly, or any percutaneous drilling trajectory initiated too inferiorly or advanced too deeply, carries a catastrophic risk of PIN transection or thermal injury. Additionally, the lateral antebrachial cutaneous nerve (LABCN) runs superficially in the subcutaneous tissues of the anterolateral forearm and is highly susceptible to injury during skin incisions and blunt dissection for portal establishment.
Exhaustive Indications and Contraindications
The decision-making algorithm for surgical intervention in capitellar OCD is dictated by patient age, skeletal maturity, lesion stability, and the presence of mechanical symptoms. Nonoperative management, consisting of absolute cessation of the offending activity, hinged bracing, and physical therapy, is the gold standard for Panner's disease and for early, stable OCD lesions in skeletally immature patients (open capitellar physis). Surgery is considered only when conservative measures have demonstrably failed after a minimum of 3 to 6 months, or when the initial presentation reveals an unstable lesion that is unlikely to heal spontaneously.
Indications for arthroscopic intervention in capitellar OCD include the presence of symptomatic loose bodies, mechanical symptoms such as catching or locking, and MRI evidence of articular cartilage separation or subchondral fluid indicative of instability. In skeletally mature adolescents with intact but softened cartilage over a necrotic subchondral bed, arthroscopic transarticular or extra-articular retrograde drilling is indicated to stimulate revascularization. For unstable lesions with salvageable cartilage fragments, arthroscopic or mini-open internal fixation using bioabsorbable pins or compression screws is warranted. When the osteochondral fragment is fragmented, detached, or unsalvageable, debridement and bone marrow stimulation (microfracture) are indicated for smaller lesions (typically <10mm). For larger, uncontained lesions (>10mm or involving the lateral margin), osteochondral autograft transfer (OATS) via a mini-arthrotomy is the definitive procedure to restore the articular contour.
Contraindications to surgical intervention are equally important to recognize. Asymptomatic lesions discovered incidentally on imaging should generally not be subjected to prophylactic surgery, as the natural history may favor spontaneous stabilization, particularly in younger patients. Active local or systemic infection is an absolute contraindication to elective arthroscopy or arthrotomy. Severe, advanced degenerative joint disease with extensive osteophyte formation and global cartilage loss represents a relative contraindication, as isolated management of the capitellar lesion is unlikely to provide meaningful clinical relief. Furthermore, severe medical comorbidities precluding safe general or regional anesthesia must be carefully weighed against the benefits of the procedure.
| Category | Indications for Surgery | Contraindications to Surgery |
|---|---|---|
| Clinical Presentation | Refractory pain after 3-6 months of rest; Mechanical symptoms (catching, locking); Symptomatic loose bodies. | Asymptomatic incidental findings; Pain resolving with conservative management. |
| Imaging Findings | Unstable lesions (subchondral fluid on MRI); Detached osteochondral fragments; Lesion size >1cm requiring grafting. | Stable, undisplaced lesions in skeletally immature patients; Advanced global osteoarthritis. |
| Patient Factors | High-level athletes demanding return to sport; Skeletally mature patients with failed healing. | Active local or systemic infection; Severe medical comorbidities precluding anesthesia. |
| Specific Procedures | Microfracture: Small (<1cm), contained lesions. OATS: Large (>1cm), uncontained lesions. | OATS: Lack of adequate donor site; Active joint infection. |
Pre-Operative Planning, Templating, and Patient Positioning
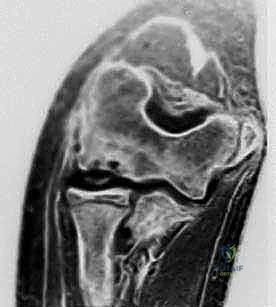
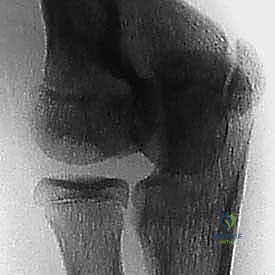
Meticulous preoperative planning is the cornerstone of successful elbow arthroscopy and osteochondral reconstruction. The process begins with a comprehensive review of high-quality imaging. Standard anteroposterior (AP), lateral, and oblique radiographs are scrutinized to assess the skeletal maturity of the capitellum, the size of the radiolucent defect, and the presence of radiopaque loose bodies.
However, plain radiographs consistently underestimate the true extent of articular involvement. Therefore, Magnetic Resonance Imaging (MRI) without contrast is the gold standard for preoperative templating. MRI allows for precise measurement of the lesion's dimensions in both sagittal and coronal planes, assessment of the overlying articular cartilage integrity, and detection of subtle subchondral edema or fluid interfaces that signify lesion instability.

Based on the MRI findings, the surgical team must anticipate the necessary equipment. Elbow arthroscopy in pediatric and adolescent patients is performed in a highly constrained space. Standard knee or shoulder arthroscopes are excessively bulky and increase the risk of iatrogenic cartilage scuffing. Therefore, a dedicated small-joint setup utilizing a 2.9 mm or 2.7 mm, 30-degree arthroscope is mandatory. The surgical tray must include appropriately sized shavers (typically 3.0 mm), delicate grasping forceps, arthroscopic awls or drills for microfracture, and bioabsorbable fixation devices if fragment stabilization is a possibility. If an OATS procedure is anticipated, the appropriate osteochondral harvesting and delivery systems must be available, and the patient's ipsilateral knee must be prepped and draped as a donor site.
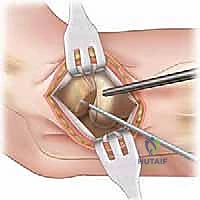
Patient positioning is a critical variable that dictates surgical exposure and safety. While the supine suspended and prone positions are viable alternatives, the lateral decubitus position is widely preferred for lateral compartment pathology due to its excellent access to the radiocapitellar joint and ease of conversion to a mini-arthrotomy. The patient is placed in the lateral decubitus position with the operative arm superior. An axillary roll is placed, and all bony prominences are meticulously padded to prevent compressive neuropathies. The operative arm is draped free and supported over a well-padded bolster or a dedicated arm positioner, allowing the elbow to rest at approximately 90 degrees of flexion.
This 90-degree flexed posture is biomechanically advantageous; it relaxes the anterior capsule, opening the radiocapitellar interval, and allows gravity to assist in distracting the joint. A sterile pneumatic tourniquet is applied to the proximal brachium to ensure a bloodless field, which is critical given the small volume of the elbow joint where even minor bleeding can obscure visualization. Intraoperative fluoroscopy (C-arm) should be positioned on the opposite side of the table, draped sterilely, and verified for adequate AP and lateral imaging trajectories before the first incision is made.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution begins with precise anatomical landmark identification. Using a sterile surgical marker, the lateral epicondyle, radial head, olecranon tip, and the course of the ulnar nerve are clearly delineated. Prior to establishing portals, the elbow joint is maximally distended. This is achieved by inserting an 18-gauge spinal needle through the "soft spot"—the center of the triangle formed by the lateral epicondyle, radial head, and olecranon—and injecting 15 to 25 mL of sterile normal saline. Adequate distension pushes the neurovascular structures away from the capsule, significantly reducing the risk of iatrogenic injury during trocar insertion.
The primary viewing portal is the proximal anterolateral portal. This is established approximately 2 cm proximal and 1 cm anterior to the lateral epicondyle, aiming towards the center of the joint. A superficial skin incision is made using a #11 blade, taking care to incise only the dermis to avoid the superficial radial nerve and LABCN. Blunt dissection with a hemostat is performed down to the capsule, followed by the insertion of a blunt trocar and cannula. Once intra-articular placement is confirmed via fluid egress, the 2.9 mm arthroscope is introduced. A thorough diagnostic sweep of the anterior compartment is performed, assessing the coronoid, anterior capsule, and the anterior aspect of the radiocapitellar articulation.
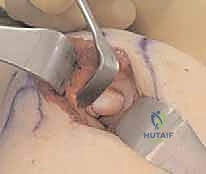
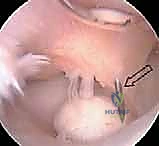
To fully address capitellar pathology, a direct lateral or mid-lateral working portal is established under direct intra-articular visualization. A spinal needle is used to localize the optimal trajectory, ensuring perpendicular access to the capitellar lesion. Once established, an arthroscopic probe is introduced to palpate the articular cartilage. The lesion is systematically evaluated for softening, ballottement, fissuring, or frank detachment. Any loose bodies within the anterior or lateral compartments are identified and carefully extracted using grasping forceps.
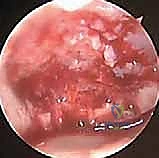
If the lesion consists of an unstable but salvageable osteochondral fragment, in situ fixation is attempted. The fibrous tissue at the base of the crater is debrided using a small curette or shaver to expose bleeding subchondral bone. The fragment is then anatomically reduced and provisionally held with a K-wire. Definitive fixation is achieved using headless bioabsorbable compression screws or pins, countersunk below the level of the articular cartilage to prevent radial head abrasion.
For unsalvageable fragments or chronic defects measuring less than 10 mm in diameter, marrow stimulation via microfracture is the treatment of choice. The necrotic cartilage and sclerotic bone are meticulously debrided back to stable, vertical margins, creating a well-contained "shoulder" of healthy cartilage. Using arthroscopic awls, multiple perforations are made into the subchondral bone plate, spaced approximately 2-3 mm apart, until the release of marrow fat droplets and blood is visualized. This technique recruits mesenchymal stem cells to form a fibrocartilaginous repair tissue.
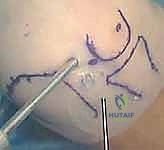
When dealing with large (>10 mm), cystic, or uncontained lesions involving the lateral capitellar margin, arthroscopic techniques are often insufficient, and an Osteochondral Autograft Transfer System (OATS) via a mini-arthrotomy is indicated. The arthroscopy is concluded, and a 4-5 cm lateral incision is made centered over the radiocapitellar joint. The extensor interval (typically between the anconeus and ECU) is split to expose the lateral capsule. An arthrotomy is performed, providing direct visualization of the capitellum.

The defect is sized, and a recipient socket is reamed to a precise depth (usually 10-15 mm). Attention is then turned to the ipsilateral knee, where a matching cylindrical osteochondral graft is harvested from a non-weight-bearing region, such as the lateral periphery of the lateral femoral condyle. The graft is carefully transferred and press-fit into the capitellar socket, ensuring the articular surface is perfectly flush with the surrounding native cartilage to maintain normal radiocapitellar kinematics.
Following rigid fixation or graft implantation, the joint is thoroughly irrigated to remove any osseous debris. The capsule is meticulously closed with interrupted absorbable sutures to prevent synovial fistulae or instability. The muscular interval, subcutaneous tissue, and skin are closed in a layered fashion. A sterile compressive dressing is applied, and the arm is placed in a well-padded posterior splint at 90 degrees of flexion to protect the repair during the immediate postoperative phase.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, arthroscopic and open management of capitellar OCD carries a distinct complication profile. The most devastating intraoperative complication is iatrogenic neurovascular injury, specifically to the posterior interosseous nerve (PIN). The PIN is highly vulnerable during the establishment of anterolateral portals and during aggressive percutaneous drilling trajectories. Injury rates are reported to be less than 1% in experienced hands, but the consequences—loss of digit and thumb extension—are profound. Prevention relies on adequate joint distension, superficial skin incisions followed by blunt dissection, and utilizing protective drill guides. If a PIN transection is identified, immediate microsurgical repair or nerve grafting is mandated.
Postoperative stiffness is arguably the most common complication following elbow surgery, with an incidence ranging from 5% to 15% depending on the extent of the procedure and the patient's compliance with rehabilitation. The elbow joint capsule is highly reactive to surgical trauma and hemarthrosis, rapidly forming dense scar tissue. Prevention requires meticulous hemostasis, the use of postoperative drains if extensive bony work was performed, and early, controlled mobilization. If severe stiffness persists beyond 6 months despite aggressive physical therapy, arthroscopic or open capsular release may be necessary as a salvage procedure.
Failure of graft incorporation, non-union of fixed fragments, or deterioration of microfracture fibrocartilage represent significant structural failures. These complications present as recurrent pain, mechanical symptoms, and progressive radiocapitellar arthritis. The incidence of graft failure in OATS procedures is approximately 5-10%. Management of these failures is highly complex. Salvage options for failed primary procedures include revision grafting (using allograft if autograft is depleted), capitellar resurfacing implants, or, in severe end-stage cases in older patients, radial head excision or total elbow arthroplasty. Infection, while rare (<1%), requires immediate aggressive management with arthroscopic irrigation, debridement, and culture-directed intravenous antibiotics.
| Complication | Estimated Incidence | Prevention and Salvage Management |
|---|---|---|
| PIN Neuropathy / Injury | < 1% | Prevention: Joint distension; blunt portal dissection; avoid deep/anterior drilling. Salvage: EMG/NCS at 6 weeks; microsurgical exploration/repair if transected. |
| Postoperative Stiffness | 5% - 15% | Prevention: Meticulous hemostasis; early controlled ROM; continuous passive motion (CPM). Salvage: Aggressive PT; dynamic splinting; arthroscopic capsular release. |
| Graft Failure / Non-union | 5% - 10% | Prevention: Precise graft sizing; flush implantation; strict adherence to weight-bearing restrictions. Salvage: Revision OATS (allograft); capitellar resurfacing; radial head excision. |
| Superficial/Deep Infection | < 1% | Prevention: Strict sterile technique; prophylactic antibiotics; meticulous wound closure. Salvage: Prompt I&D; targeted IV antibiotic therapy; hardware removal if necessary. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following surgical management of capitellar OCD is as critical to the final outcome as the surgery itself. The protocol must carefully balance the need to protect the healing osteochondral interface with the imperative to prevent debilitating capsular contracture. Rehabilitation is generally divided into four distinct phases, tailored to the specific surgical procedure performed (e.g., microfracture vs. OATS).
Phase 1: Immediate Postoperative Protection (Weeks 0-2)
The primary goals of this phase are to control pain and inflammation, protect the surgical site, and initiate early, safe range of motion. The patient is typically immobilized in a posterior splint at 90 degrees of flexion for the first 3 to 7 days to allow soft tissue healing. Following this, the patient is transitioned to a hinged elbow brace. Active-assisted and passive range of motion (ROM) exercises for flexion and extension are initiated, usually limiting terminal extension by 15-20 degrees to avoid excessive radiocapitellar compression. Pronation and supination are performed with the elbow flexed to 90 degrees. Strict avoidance of any upper extremity weight-bearing or valgus stress is mandatory.
Phase 2: Intermediate Motion and Light Strengthening (Weeks 2-6)
During this phase, the goal is to achieve full, symmetrical range of motion. The hinged brace is gradually unlocked and eventually discontinued. Passive stretching is incorporated to address any residual capsular tightness. Scapular stabilization exercises and light, submaximal isometric strengthening of the biceps, triceps, and forearm musculature are introduced. However, heavy lifting, pushing, and pulling remain strictly prohibited to protect the maturing fibrocartilage or integrating osteochondral graft. Modalities such as cryotherapy and electrical stimulation may be used as adjuncts for pain and edema control.
Phase 3: Advanced Strengthening and Proprioception (Weeks 6-12)
Once full, painless ROM is achieved and radiographic evidence of healing is observed (if applicable), the patient progresses to isotonic and dynamic strengthening. Closed kinetic chain exercises are cautiously introduced, gradually increasing the axial load on the radiocapitellar joint. Plyometric exercises and sport-specific functional drills are initiated. For throwing athletes, this phase focuses on core strengthening, kinetic chain mechanics, and a highly structured, interval throwing program. The throwing program begins with short-distance, flat-ground tosses and progressively increases in distance and velocity.
Phase 4: Return to Play (Months 3-6+)
The final phase is focused on a safe return to competitive athletics. Progression to this phase requires a completely asymptomatic elbow, full functional strength, and successful completion of the interval throwing or sport-specific program without pain. For patients who underwent microfracture, return to high-impact sports is typically delayed until 4-6 months to allow adequate maturation of the fibrocartilage. For OATS procedures, the incorporation of the bony plug may allow a slightly earlier return to light activities, but full competitive throwing or gymnastics often requires a full 6 months of rehabilitation. Continuous monitoring for recurrent symptoms is essential during this transition back to full activity.
Summary of Landmark Literature and Clinical Guidelines
The evolution of treatment for capitellar OCD has been heavily influenced by several landmark clinical studies and biomechanical investigations. Historically, conservative management was the mainstay, but high failure rates in older adolescents prompted a shift toward surgical intervention. Takahara et al. published critical long-term follow-up data demonstrating that nonoperative management of advanced capitellar OCD lesions in skeletally mature patients yields poor clinical outcomes, with a high incidence of residual pain and early osteoarthritis. This established the modern paradigm that unstable lesions or those in mature athletes require surgical stabilization or reconstruction.
The efficacy of arthroscopic microfracture for small, contained defects was validated by several cohorts, demonstrating excellent short-to-mid-term relief of pain and restoration of function. However, literature from Ruch and others highlighted the limitations of marrow stimulation techniques for larger defects, noting that the resulting fibrocartilage lacks the biomechanical durability of native hyaline cartilage, often leading to deterioration in high-demand throwing athletes. This catalyzed the adoption of osteochondral autograft transfer (OATS).
O'Driscoll and colleagues have provided foundational literature on the precise surgical anatomy and the biomechanical rationale for restoring the radiocapitellar articular contour. Clinical outcome studies evaluating OATS for large capitellar defects have consistently shown superior long-term results compared to microfracture, with higher rates of return to pre-injury levels of sport, particularly in baseball pitchers and gymnasts. Current clinical guidelines, supported by the American Orthopaedic Society for Sports Medicine (AOSSM), strongly recommend MRI for accurate preoperative staging and advocate for OATS or advanced reconstructive techniques for uncontained lesions exceeding 10 mm, ensuring the restoration of joint congruity and the preservation of long-term elbow function.
