Total Hip Arthroplasty for Metastatic Lesions: An Intraoperative Masterclass
Key Takeaway
This masterclass provides an exhaustive, real-time guide to Total Hip Arthroplasty for metastatic bone disease. We cover intricate surgical anatomy, meticulous preoperative planning, and granular intraoperative execution, emphasizing technique and rationale. Fellows will learn to navigate complex acetabular and femoral reconstructions, manage intraoperative challenges, and optimize postoperative recovery for these challenging oncology patients.
Comprehensive Introduction and Patho-Epidemiology
The surgical management of metastatic bone disease (MBD) affecting the proximal femur and acetabulum represents one of the most formidable challenges in orthopedic oncology and adult reconstruction. Total Hip Arthroplasty (THA) in this setting is profoundly distinct from routine primary arthroplasty for osteoarthritis. Here, the orthopedic surgeon operates within a compromised biomechanical and biological milieu, facing distorted anatomy, depleted bone stock, and the ever-present risk of catastrophic hemorrhage. Our fundamental objective is to execute a durable, rigid reconstruction that affords immediate mechanical stability, thereby alleviating debilitating pain and restoring functional independence for a patient with a limited, yet highly precious, life expectancy.
Pathophysiology of Bone Metastases
To effectively treat these lesions, we must first understand the underlying cellular mechanisms driving bone destruction. Metastatic bone disease is a pervasive sequela of advanced malignancy, affecting over half of the 1.2 million patients newly diagnosed with cancer annually. The skeleton is the third most common site for metastatic spread, following the lung and liver. The proximal femur is the most frequently affected long bone, harboring approximately 25% of all appendicular skeletal metastases. This predilection is largely attributed to the "seed and soil" hypothesis; the rich vascularity and abundant hematopoietic red marrow of the proximal femur provide a fertile, nutrient-rich environment for circulating tumor cells. Consequently, nearly 75% of all oncologic surgical interventions for bone metastases are localized around the hip joint.
Systemically, patients with extensive marrow infiltration often present with a "leukoerythroblastic reaction" on peripheral blood smears. This phenomenon, characterized by the premature release of nucleated red blood cells and immature myeloid cells into the peripheral circulation, is a critical hematologic indicator of significant marrow space occupation by tumor, often accompanied by underlying anemia and thrombocytopenia.
Osteolytic versus Osteoblastic Lesions
Metastatic lesions are broadly categorized into osteoblastic and osteolytic variants, though mixed patterns are common. Osteoblastic metastases, classically associated with prostate carcinoma, are characterized by disorganized, woven bone formation. While these lesions are typically less painful and present a lower immediate risk of pathologic fracture due to the preservation of some structural integrity, the woven bone is biomechanically inferior to native lamellar bone. It is paramount to recognize that not all prostate metastases are strictly blastic; mixed or lytic variants exist and carry a higher fracture risk.
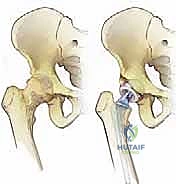
Conversely, osteolytic metastases—frequently originating from breast, renal cell, thyroid, and lung carcinomas—are hallmarked by aggressive bone destruction. This osteolysis is rarely caused by the direct mechanical pressure of the tumor mass. Instead, tumor cells secrete a cascade of cytokines and parathyroid hormone-related protein (PTHrP), which upregulate the Receptor Activator of Nuclear Factor Kappa-B Ligand (RANKL) pathway. This stimulates native osteoclast differentiation and hyperactivation, leading to rapid, painful bone resorption. Furthermore, tumors such as renal cell carcinoma, thyroid carcinoma, and multiple myeloma are notorious for profound neovascularization. These hypervascular lesions present a severe risk of massive, life-threatening intraoperative hemorrhage, necessitating meticulous preoperative planning and, frequently, prophylactic angiographic embolization.
Detailed Surgical Anatomy and Biomechanics
Navigating the hip joint in the presence of metastatic disease demands an exhaustive, three-dimensional understanding of pelvic and femoral anatomy. The tumor often obliterates standard surgical landmarks, requiring the surgeon to rely on deep anatomical relationships to safely achieve exposure and definitive fixation.
The Acetabulum: Columns, Walls, and the Weight-Bearing Dome
The acetabulum is a complex hemispherical socket formed by the triradiate confluence of the ilium, ischium, and pubis. From a reconstructive standpoint, it is conceptualized as two primary columns and two walls. The anterior column extends from the anterior iliac crest down through the anterior half of the acetabulum to the pubic symphysis. The posterior column, which is massive and critical for load transfer, originates at the superior gluteal notch, traverses the posterior half of the acetabulum, and terminates at the ischial tuberosity. The anterior and posterior walls provide structural containment for the femoral head.
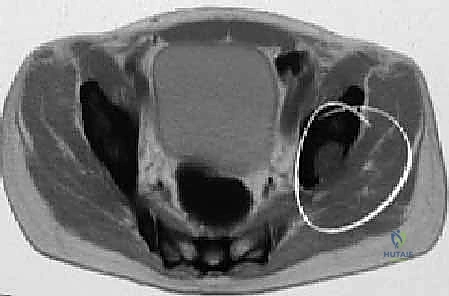
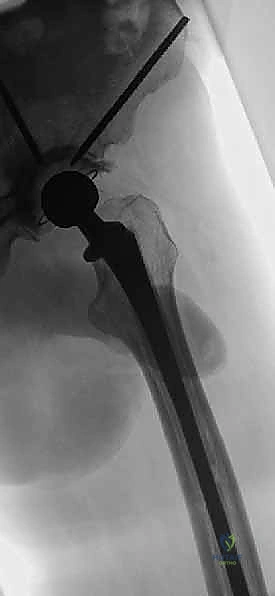
The acetabular dome, the superior weight-bearing roof, is the paramount region for achieving stable prosthetic fixation. Metastatic infiltration of the dome or the medial wall (lamina papyracea) severely compromises the mechanical integrity of the pelvis. Unrecognized or inadequately addressed defects in these regions inevitably lead to catastrophic implant subsidence, protrusio acetabuli, and construct failure.
The Proximal Femur: Trabecular Architecture and Stress Risers
The proximal femur is an architectural marvel designed to withstand immense biomechanical forces. The internal structure is defined by intersecting systems of trabecular bone: the principal compressive group extending from the medial cortex into the superior femoral head, and the principal tensile group arcing from the lateral cortex into the inferior head. The intersection of these groups leaves a central area of relative weakness known as Ward's triangle.
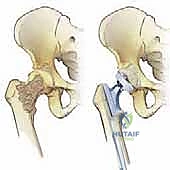
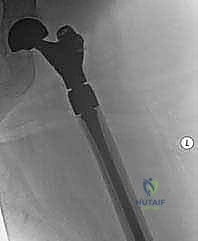
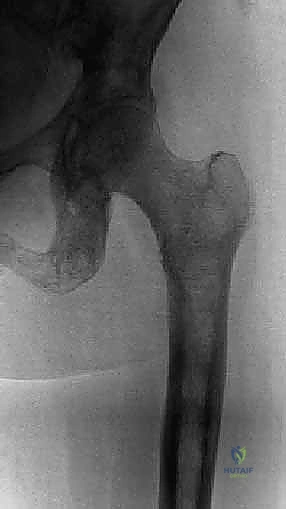
Metastatic osteolysis disrupts this intricate trabecular network, creating massive stress risers. The intertrochanteric and subtrochanteric regions are subjected to the highest bending moments in the human body. A lytic lesion in these areas drastically reduces the bone's load-bearing capacity. Biomechanical studies demonstrate that a cortical defect involving greater than 50% of the bone diameter reduces torsional strength by over 60%, placing the patient at imminent risk of a devastating pathologic fracture during routine weight-bearing or even simple bed mobility.
Critical Neurovascular Relationships
Regardless of the surgical approach, intimate knowledge of the surrounding neurovascular structures is non-negotiable. The distorted anatomy caused by tumor expansion can displace these structures from their native beds.
* Sciatic Nerve: Exiting the greater sciatic foramen, it courses deep to the piriformis and superficial to the short external rotators. It is highly vulnerable during posterior approaches, particularly during aggressive posterior retraction, posterior column screw placement, or when tumor extends posteriorly through the greater sciatic notch.
* Femoral Nerve and Vessels: Located within the anterior femoral triangle. They are at risk during anterior approaches or when resecting tumors involving the anterior column or superior pubic ramus.
* Superior and Inferior Gluteal Neurovascular Bundles: Supplying the abductors and gluteus maximus, respectively. Injury to the superior gluteal nerve during superior retraction leads to devastating postoperative abductor weakness and a persistent Trendelenburg gait.
* Obturator Nerve and Vessels: Traversing the obturator foramen. They are at extreme risk during aggressive medial acetabular reaming or when dissecting a tumor that has breached the quadrilateral plate.
Exhaustive Indications and Contraindications
The decision to proceed with operative intervention in a patient with metastatic disease requires a multidisciplinary approach involving orthopedic oncology, medical oncology, radiation oncology, and palliative care. The primary goal is palliative: to relieve pain, restore ambulatory function, and facilitate nursing care.
The Mirels Scoring System
The Mirels scoring system remains the gold standard for quantifying the risk of pathologic fracture in long bones and guiding the indication for prophylactic stabilization. It evaluates four parameters, scoring each from 1 to 3:
- Site of Lesion: Upper limb (1), Lower limb (2), Peritrochanteric region (3). The peritrochanteric area scores highest due to extreme biomechanical stress.
- Pain Level: Mild (1), Moderate (2), Functional/Severe pain on weight-bearing (3).
- Lesion Characteristics: Blastic (1), Mixed (2), Lytic (3).
- Size of Lesion (Cortical Extent): Less than 1/3 of cortex (1), 1/3 to 2/3 of cortex (2), Greater than 2/3 of cortex (3).
A cumulative Mirels score of 7 or less suggests a low risk of fracture (typically <5%), and these patients may be managed non-operatively with radiation therapy and bisphosphonates. A score of 8 indicates a moderate risk (15%), often warranting surgical consideration. A score of 9 or greater indicates a high impending fracture risk (>33%), making prophylactic surgical stabilization definitively indicated.
Indications and Contraindications Table
| Parameter | Operative Indications (THA / Reconstruction) | Absolute & Relative Contraindications |
|---|---|---|
| Fracture Status | Displaced pathologic fracture of femoral neck/head. Impending fracture (Mirels $\ge$ 8). Avulsion of lesser trochanter. |
Undisplaced fracture responding to radiation (Relative). Mirels score $\le$ 7 without functional pain. |
| Life Expectancy | Estimated survival > 6 weeks. | Estimated survival < 4-6 weeks (Absolute). Patient will not survive the perioperative rehabilitation period. |
| Patient Status | Intractable pain unresponsive to medical management. Loss of ambulatory function. |
Medically unfit for anesthesia (ASA V). Uncorrectable coagulopathy. Active, overwhelming systemic infection. |
| Tumor Biology | Failure of conservative therapy (radiation/systemic). Radioresistant tumors (e.g., Renal Cell Carcinoma). |
Highly responsive tumors where radiation alone provides rapid structural consolidation (e.g., Lymphoma, Myeloma - Relative). |
Pre-Operative Planning, Templating, and Patient Positioning
The success of a metastatic THA is dictated long before the skin incision is made. Meticulous preoperative planning is the bulwark against catastrophic intraoperative complications.
Comprehensive Imaging and Systemic Staging
A standard radiographic series (AP pelvis, full-length AP and lateral femur) is mandatory but insufficient. Plain films are notoriously insensitive, only revealing lytic destruction after 30-50% of the trabecular bone has been obliterated.
Advanced imaging is absolutely critical:
* Detailed Pelvic and Femoral CT: A fine-cut CT scan (without contrast, unless assessing vascularity) is the cornerstone of preoperative templating. It accurately defines the true extent of cortical destruction and acetabular bone loss, which is underestimated by plain radiographs in up to 83% of cases.
* Total Body Bone Scan / PET-CT: Essential for identifying synchronous skeletal lesions. A distal femoral lesion, if missed, could act as a stress riser at the tip of a standard-length femoral stem, leading to a periprosthetic fracture.
* MRI: Invaluable for assessing soft tissue extension, neurovascular involvement, and the precise intramedullary extent of the tumor.
Preoperative Embolization
For highly vascular tumors—specifically Renal Cell Carcinoma, Thyroid Carcinoma, and Multiple Myeloma—preoperative selective arterial embolization is a critical adjunct. Embolization should be performed within 24 to 48 hours prior to surgery. Failure to embolize these hypervascular lesions can result in rapid, massive exsanguination upon entering the tumor capsule, obscuring the surgical field and precipitating hemorrhagic shock.
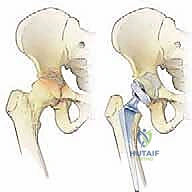
Implant Selection and Templating Strategy
The reconstructive paradigm in metastatic disease relies on immediate, rigid fixation. Biological ingrowth (uncemented fixation) is unreliable due to poor bone quality, the systemic effects of chemotherapy, and the necessity for postoperative radiation therapy, which halts osteointegration.
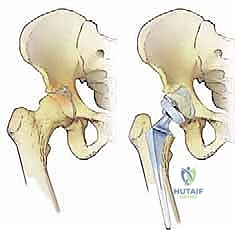
Therefore, cemented arthroplasty components are the gold standard.
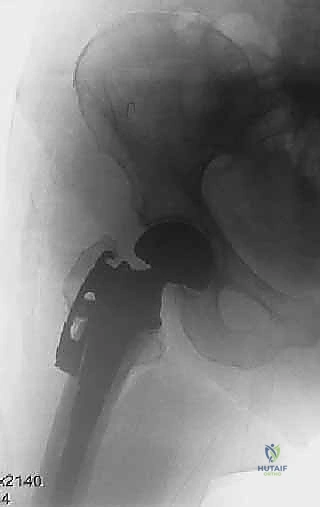
* Femoral Reconstruction: We template for cemented, long-stemmed components. The stem must bypass the most distal aspect of the lytic lesion by a minimum of two cortical bone diameters to prevent stress shielding and subsequent fracture.
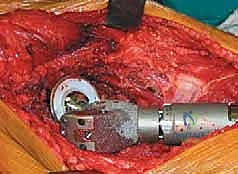
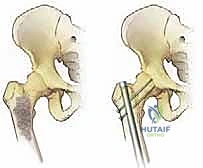
* Acetabular Reconstruction: Based on the CT scan, we classify acetabular defects (often using the Harrington classification). Class I (intact walls/roof) may accept a standard cemented cup. Class II (medial wall deficiency) requires protrusio rings or cages. Class III (massive roof/column defects) necessitates complex reconstruction with anti-protrusio cages, massive structural allografts, or custom triflange components, augmented with copious polymethylmethacrylate (PMMA) bone cement.
Patient Positioning
General endotracheal anesthesia is typically employed. The patient is placed in the lateral decubitus position on a radiolucent operative table, utilizing a vacuum beanbag for rigid stabilization. The affected hip is positioned superiorly.
Meticulous padding of all bony prominences (axillary roll, fibular head, medial malleoli) is critical to prevent iatrogenic neuropathies in these often cachectic patients. The sterile field must be draped widely to allow for an extensile approach, ensuring access from the iliac crest down to the distal femur. Intraoperative fluoroscopy must be positioned to allow unhindered AP and lateral imaging of the entire proximal femur and pelvis throughout the procedure.
Step-by-Step Surgical Approach and Fixation Technique
We will execute this procedure utilizing an extensile posterior approach. This approach provides unparalleled visualization of the posterior column, acetabulum, and the entire proximal femur, facilitating complex reconstructions and the insertion of long-stemmed prostheses.
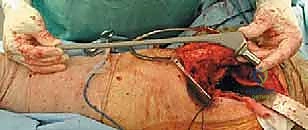
1. Extensile Incision and Deep Dissection
The skin incision is curvilinear, beginning approximately 5 cm posterior and superior to the greater trochanter, extending distally over the trochanter, and continuing along the longitudinal axis of the femoral shaft. The length of the distal extension is dictated by the templated length of the femoral stem.
We sharply incise the subcutaneous tissue down to the fascia lata. The fascia lata is split longitudinally in line with the skin incision. Proximally, the gluteus maximus fibers are split bluntly in line with their orientation, minimizing denervation and muscular trauma. A Charnley or self-retaining retractor is placed to maintain exposure.
We identify the short external rotators (piriformis, superior gemellus, obturator internus, inferior gemellus, and quadratus femoris). The sciatic nerve must be palpated and visually confirmed, typically lying deep to the piriformis and superficial to the obturator internus bursa. We place heavy, non-absorbable tagging sutures in the tendinous insertions of the short external rotators near the greater trochanter and sharply release them. These muscles are reflected posteriorly, acting as a soft-tissue buffer to protect the sciatic nerve during the remainder of the procedure.
2. Capsulotomy, Dislocation, and Femoral Neck Osteotomy
A robust T-shaped or H-shaped capsulotomy is performed to expose the femoral head and neck.
Surgical Warning: Dislocation of the hip must be performed with extreme caution. The proximal femur is often severely compromised by tumor. Forceful internal rotation and adduction can easily cause an iatrogenic spiral fracture of the femoral shaft. If the hip is difficult to dislocate, or if the neck is highly lytic, it is safer to perform the femoral neck osteotomy in situ prior to extracting the femoral head with a corkscrew.
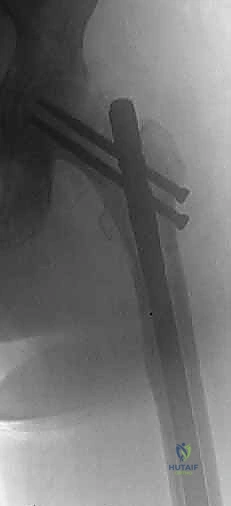
Using preoperative templating as a guide, the femoral neck osteotomy is performed using an oscillating saw, typically 1 to 1.5 cm above the lesser trochanter. The femoral head is removed and sent for histopathological analysis.
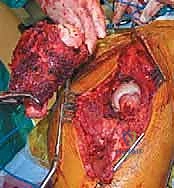
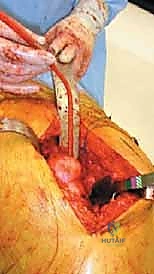
3. Acetabular Preparation and Reconstruction
Exposure of the acetabulum is achieved by placing a blunt Hohmann retractor anteriorly over the anterior column, a retractor inferiorly beneath the transverse acetabular ligament, and a posterior retractor carefully positioned to avoid the sciatic nerve.
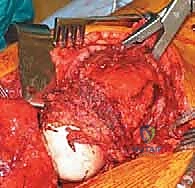
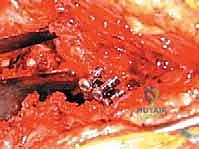
We meticulously debride the labrum, pulvinar, and any gross tumor mass within the acetabular fossa. Tumor debulking should be rapid and aggressive down to bleeding, healthy bone. If significant hemorrhage occurs from a hypervascular tumor bed, the cavity should be immediately packed with laparotomy sponges soaked in hot saline or epinephrine, followed by rapid cementation to achieve hemostasis.
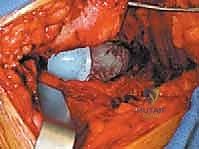
Reaming is performed sequentially. In metastatic disease, we often under-ream the acetabulum to preserve whatever subchondral bone remains.
If a Harrington Class II or III defect is present, standard cementation is inadequate. We must utilize an anti-protrusio cage (e.g., Burch-Schneider cage). The cage is contoured to bridge the defect, securing fixation into the intact ilium proximally and the ischium distally with multiple cortical screws. Once the cage is rigidly fixed, high-viscosity PMMA bone cement is pressurized into the construct, and an all-polyethylene acetabular component is cemented into the cage at the appropriate inclination (40-45 degrees) and anteversion (15-20 degrees).
4. Femoral Preparation and Stem Cementation
The femur is elevated and exposed. We initiate canal preparation with a box osteotome and sequential broaching. Intramedullary tumor is aggressively curetted and cleared using a long flexible reamer or a canal brush. Thorough irrigation with pulsatile lavage is critical to remove tumor debris, fat, and marrow, which minimizes the risk of fat embolism syndrome during cement pressurization.

A prophylactic cerclage wire may be placed just distal to the lesser trochanter prior to broaching to prevent iatrogenic splitting of the compromised proximal femur.
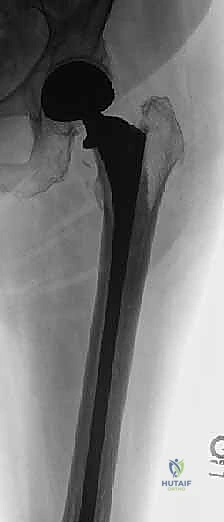
We utilize third-generation cementing techniques. A distal cement restrictor (plug) is placed 1-2 cm distal to the templated tip of the long stem. The canal is meticulously dried. High-viscosity PMMA is injected in a retrograde fashion using a cement gun, ensuring complete filling of the canal and interdigitation into the cancellous bone.
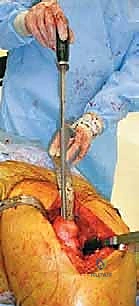
The long-stemmed femoral component is inserted slowly to allow air and fat to escape, reducing the risk of a massive embolic event. The stem is held rigidly in appropriate anteversion (10-15 degrees) until the PMMA has completely polymerized.
5. Reduction, Assessment, and Closure
Once the cement has cured, trial heads are utilized to reduce the hip and assess leg length, offset, and stability in all planes of motion. We check for impingement anteriorly in flexion/internal rotation and posteriorly in extension/external rotation.
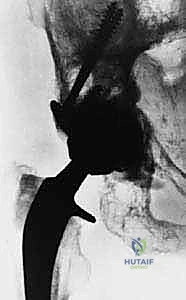
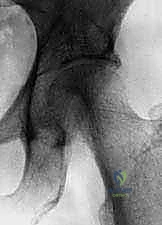
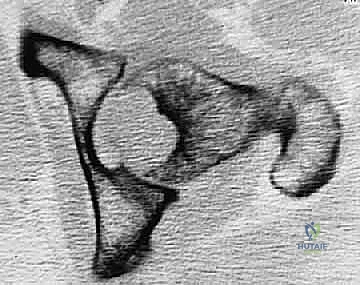
After selecting the definitive femoral head, the hip is finally reduced. Copious irrigation is performed. A deep subfascial drain is routinely placed due to the highly vascular nature of the tumor bed and the extensile dissection.

Meticulous closure is paramount. The short external rotators and posterior capsule are repaired back to the greater trochanter through transosseous drill holes to maximize posterior stability. The fascia lata is closed with heavy interrupted sutures. The subcutaneous tissues and skin are closed in layers.
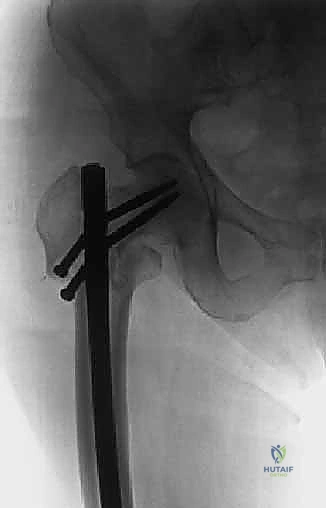
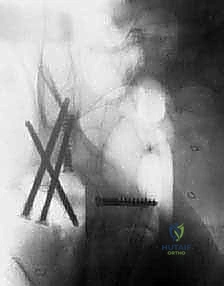
Complications, Incidence Rates, and Salvage Management
Surgical intervention for MBD carries a significantly higher morbidity and mortality profile compared to primary THA. The systemic burden of cancer, malnutrition, prior radiation, and chemotherapy severely compromise the patient's physiological reserve.
Intraoperative and Postoperative Challenges
Intraoperative Hemorrhage: As discussed, this is a primary concern. Rapid debulking, immediate packing, and the use of PMMA as a hemostatic agent are critical intraoperative salvage techniques.
Venous Thromboembolism (VTE): Cancer patients exist in a hypercoagulable state. The addition of major pelvic surgery and cement pressurization skyrockets the risk of Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE). Aggressive mechanical and pharmacological prophylaxis is mandatory.
Instability and Dislocation: Dislocation rates in oncologic THA are substantially higher (up to 10-15%) than in primary arthroplasty (1-2%). This is due to extensive soft tissue resection, muscle weakness, and altered geometry. The use of large-diameter femoral heads, constrained liners, or dual-mobility articulations should be strongly considered in patients with massive abductor deficiency.
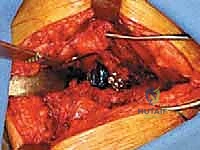

Complications and Salvage Table
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage / Management Strategy |
|---|---|---|---|
| Intraoperative Hemorrhage | 5 - 15% (Higher in RCC/Thyroid) | Hypervascular tumor, inadequate pre-op embolization. | Rapid debulking, packing, PMMA cementation, massive transfusion protocol. |
| Dislocation | 5 - 15% | Extensive soft tissue resection, abductor weakness, non-compliance. | Closed reduction. If recurrent, revision to dual-mobility or constrained liner. |
| Periprosthetic Infection | 3 - 8% | Immunosuppression (chemo/radiation), malnutrition, prolonged operative time. | Aggressive I&D, modular exchange. Two-stage revision often contraindicated due to life expectancy; suppressive antibiotics may be required. |
| Aseptic Loosening / Construct Failure | 2 - 5% (Early) | Disease progression, inadequate initial fixation, failure to bypass stress risers. | Revision arthroplasty with longer stem, massive allograft, or proximal femoral replacement (megaprosthesis). |
| VTE (DVT / PE) | 5 - 10% | Hypercoagulable state of malignancy, endothelial injury, stasis. | Therapeutic anticoagulation, inferior vena cava (IVC) filter if anticoagulation is contraindicated. |
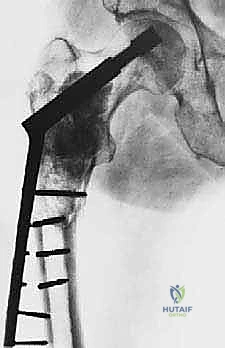
Phased Post-Operative Rehabilitation Protocols
The entire rationale for utilizing cemented arthroplasty components in this patient population is to permit immediate, full weight-bearing. Prolonged bed rest is a death sentence for the metastatic cancer patient, leading to rapid deconditioning, pneumonia, decubitus ulcers, and VTE.
Immediate Postoperative Phase (Days 1-3)
Patients are mobilized out of bed on postoperative day one. Weight-bearing as tolerated (WBAT) is initiated immediately with the assistance of physical therapy and an appropriate walking aid (walker or crutches). Strict posterior hip precautions (no flexion past 90 degrees, no internal rotation, no adduction past midline) are enforced to mitigate the high risk of dislocation.
Integration with Adjuvant Therapies
Coordination with the radiation oncologist is critical. Postoperative radiation therapy is frequently required to achieve local tumor control and prevent disease progression that could compromise the reconstruction. However, radiation severely impairs wound healing. Therefore, radiation therapy is typically delayed for 2 to 3 weeks postoperatively until the surgical incision has completely healed and sutures/staples have been removed.
Systemic chemotherapy may also need to be held perioperatively, depending on the specific agents and the patient's hematologic indices, to prevent profound neutropenia and subsequent periprosthetic joint infection.
Summary of Landmark Literature and Clinical Guidelines
The evolution of surgical management for metastatic disease of the hip is grounded in several seminal works.
- Mirels' Original Classification (1989): H. Mirels' publication in Clinical Orthopaedics and Related Research established the foundational scoring system that remains the international standard for predicting pathologic fractures and guiding prophylactic fixation.
- Harrington's Acetabular Reconstruction (1981): K.D. Harrington's classification of acetabular defects and his pioneering work on the use of threaded pins, anti-pro
