Flexor Tendon Tenolysis: An Intraoperative Masterclass for Restoring Digital Function
Key Takeaway
This masterclass guides fellows through flexor tendon tenolysis, a critical procedure for restoring digital motion post-injury. We cover detailed surgical anatomy, meticulous preoperative planning, and granular intraoperative execution, emphasizing precise adhesiolysis techniques. Learn to identify and address adhesions, manage pulleys, and navigate potential pitfalls, ensuring optimal functional outcomes and patient recovery through expert surgical and rehabilitative strategies.
Comprehensive Introduction and Patho-Epidemiology
The surgical management of flexor tendon adhesions represents one of the most formidable challenges in the armamentarium of the hand and upper extremity surgeon. Flexor tendon tenolysis is an intricate, highly demanding procedure that transcends mere scar resection; it is a masterclass in restoring the delicate biomechanics of digital kinematics. When a patient presents with a profound discrepancy between passive and active range of motion following a previous flexor tendon repair, the surgeon is confronted with a dense, scarred landscape. These fibrous tethers, which act as unyielding anchors within the fibro-osseous tunnel, severely limit the gliding capacity of the tendons, effectively neutralizing the mechanical advantage of the musculotendinous unit. The overarching goal of tenolysis is to liberate these structures, thereby restoring the fundamental ability to grasp, pinch, and manipulate objects, which are the cornerstones of human hand function.
The pathophysiology of flexor tendon adhesions is deeply rooted in the complex biology of tendon healing. Following trauma and subsequent surgical repair, the tendon undergoes a triphasic healing process: inflammatory, proliferative, and remodeling. Unfortunately, the very mechanisms designed to bridge the tendon gap also orchestrate the formation of extratendinous adhesions. The extracellular matrix, a dynamic scaffold composed of collagen, proteoglycans, fibronectin, and elastin, plays a paradoxical role. Degradation products from the initial trauma act as potent chemotactic signals, recruiting fibroblasts, leukocytes, and endothelial cells. Within days of the injury, T cells and macrophages infiltrate the site, stimulating synovial cells to upregulate fibronectin production. By the end of the first week, an influx of type I and type III collagen thickens the epitenon to five to seven cell layers, utilizing the fibronectin as a provisional scaffold. This robust fibroblastic response bridges the tendon to the surrounding synovial sheath, periosteum, and volar plate, creating the restrictive adhesions we encounter clinically.
Historically, injuries within Zone II—infamously dubbed "no man's land" by Bunnell—were considered nearly insurmountable due to the high propensity for adhesion formation in this tightly constrained anatomical space. Early surgical philosophies often advocated for delayed definitive treatment or primary tendon grafting, hoping to circumvent the inevitable scarring. However, these strategies proved largely ineffective and often led to profound stiffness. Modern hand surgery acknowledges that adhesions are a biologic reality of the healing process, exacerbated by factors such as severe initial trauma, imprecise surgical technique, prolonged immobilization, and infection. We now classify these adhesions into three primary morphological types: loose adhesions arising from subcutaneous tissue that permit some tendon glide; moderately dense adhesions originating from the synovial sheath or pulleys that significantly restrict motion; and dense, unyielding adhesions arising from the bony floor or volar plate that often penetrate the dorsal aspect of the tendon, completely obliterating excursion.
The epidemiology of flexor tendon adhesions requiring surgical intervention remains significant despite advances in primary repair techniques and early active motion protocols. Literature suggests that up to 20% of patients undergoing primary flexor tendon repair in Zone II may eventually require a tenolysis to achieve optimal functional outcomes. The decision to proceed with tenolysis is dynamic and highly individualized, heavily influenced by the patient's physiological response to injury, their psychological resilience, and the structural integrity of the involved tendon and surrounding sheath. As we navigate this scarred landscape, our intraoperative decision-making must be precise, guided by a profound understanding of the underlying patho-epidemiology and a commitment to meticulous, tissue-sparing surgical technique.
Detailed Surgical Anatomy and Biomechanics
Before initiating any surgical incision, a comprehensive mastery of the critical anatomy and biomechanics of the digital flexor tendon system is non-negotiable. This intricate system is a marvel of biological engineering, designed to transmit massive forces across multiple articulations while maintaining a remarkably low-friction gliding environment. The digital flexor system primarily comprises two tendons: the Flexor Digitorum Superficialis (FDS) and the Flexor Digitorum Profundus (FDP). The FDS bifurcates at the level of the proximal phalanx, forming two slips that wrap around the FDP at Camper's chiasm before inserting onto the volar base of the middle phalanx. Its primary biomechanical role is proximal interphalangeal (PIP) joint flexion. The FDP continues distally through this bifurcation to insert onto the volar base of the distal phalanx, serving as the primary flexor of the distal interphalangeal (DIP) joint while secondarily contributing to PIP and metacarpophalangeal (MP) joint flexion.

These tendons glide within a highly constrained fibro-osseous tunnel, particularly within Zone II, which extends from the proximal edge of the A1 pulley to the insertion of the FDS. This tunnel is lined by a synovial sheath that facilitates a frictionless glide and provides a pathway for synovial diffusion, a critical component of tendon nutrition. The mechanical efficiency of the flexor tendons is maintained by a complex retinacular pulley system that prevents bowstringing during digital flexion. The annular pulleys (A1 through A5) are robust, transverse fibrous bands. The A2 pulley, originating from the proximal phalanx, and the A4 pulley, originating from the middle phalanx, are the biomechanical keystones of this system. Their preservation is absolutely critical; compromise of the A2 or A4 pulleys drastically alters the moment arm of the tendons, leading to severe bowstringing, a significant increase in the work of flexion, and a profound loss of digital motion and grip strength. The thinner, oblique cruciform pulleys (C1 through C3) intersperse the annular pulleys, allowing the sheath to collapse dynamically during digital flexion without impinging on the tendons.
The neurovascular anatomy surrounding the flexor tendon sheath is equally critical and highly vulnerable during tenolysis. Running along the midaxial lines of each digit are the proper digital nerves and arteries, typically organized as radial and ulnar neurovascular bundles. The digital nerves, positioned volar to the arteries, provide critical tactile sensation and sympathetic innervation to the digit. These structures are intimately associated with the flexor sheath and are frequently encased in the same dense scar tissue that tethers the tendons. Cleland's and Grayson's ligaments compartmentalize these bundles, but profound scarring can distort these normal anatomical planes, rendering the nerves highly susceptible to iatrogenic transection or traction injury during dissection. The digital arteries provide the primary vascular perfusion to the digit, and their preservation is paramount to prevent digital ischemia, particularly in digits with a history of severe crush injury or replantation.
The intrinsic blood supply to the flexor tendons themselves is a crucial consideration during tenolysis. Tendon nutrition relies on a dual system: vascular perfusion via the vincula system and avascular diffusion from the synovial fluid. The vincula brevia and longa are delicate, mesentery-like structures that carry segmental blood vessels from the digital arteries to the dorsal aspect of the tendons. During tenolysis, aggressive circumferential dissection or indiscriminate stripping of the dorsal tendon surface can easily obliterate these tenuous vascular tethers. If the vincula are destroyed, the tendon becomes entirely reliant on synovial diffusion, which may be insufficient in a heavily scarred, avascular bed, precipitating ischemic necrosis or spontaneous rupture. Therefore, the surgeon must balance the need for complete mechanical release with the biological imperative of preserving the tendon's fragile microcirculation.
Exhaustive Indications and Contraindications
The decision to proceed with a flexor tendon tenolysis requires meticulous clinical judgment and a rigorous assessment of the patient's functional status. The primary, overarching indication for tenolysis is a localized, mechanical restriction of tendon glide following a primary repair, tendon graft, or trauma, characterized by a significant discrepancy between passive and active range of motion. We define this clinical scenario as a patient who has achieved a soft tissue equilibrium but has reached a definitive functional plateau. This plateau is typically recognized when there has been no measurable improvement in active digital motion over a continuous period of 4 to 8 weeks, despite the patient's strict adherence to an intensive, expertly guided hand therapy protocol. It is critical to confirm that the underlying tendon is structurally intact; tenolysis of a ruptured or severely attenuated tendon is an exercise in futility and necessitates a completely different reconstructive algorithm.
Patient selection is arguably the most critical determinant of a successful outcome in flexor tendon tenolysis. The surgeon must evaluate not only the mechanical deficit but also the patient's psychological readiness and compliance. The ideal candidate is highly motivated, cognitively intact, and fully comprehends that the surgical procedure represents only half of the therapeutic equation. They must be willing to endure the rigors of immediate, often painful, postoperative active motion therapy. Furthermore, the soft tissue envelope must be pristine. All prior surgical incisions or traumatic wounds must be completely healed, with soft, pliable skin and an absence of induration, erythema, or active inflammation. The skeletal architecture must be stable, with all fractures fully consolidated, as the forces generated during post-tenolysis rehabilitation can easily displace a tenuous nonunion.
Conversely, the contraindications for tenolysis are absolute and must be rigorously respected to prevent catastrophic complications. Active or latent infection within the operative field is an absolute contraindication, as surgical intervention will rapidly disseminate the pathogen, potentially leading to deep space infections or osteomyelitis. Severe, unyielding joint contractures that cannot be corrected passively preclude isolated tenolysis; if the joint itself cannot be mobilized, releasing the tendon will yield no functional benefit. In such cases, a staged approach involving primary capsulectomy or joint release followed by delayed tenolysis, or a combined procedure if deemed safe, must be considered. Inadequate soft tissue coverage, characterized by thin, adherent, or irradiated skin, is a strong relative contraindication, as the surgical approach may lead to wound breakdown and exposed tendon.
Furthermore, profound neurovascular compromise in the affected digit is a significant red flag. Performing a tenolysis in a dysvascular digit risks precipitating frank gangrene, while surgery in an insensate digit often yields poor functional results due to the lack of proprioceptive feedback necessary for effective rehabilitation. Finally, a patient who exhibits poor compliance, lacks motivation, or has secondary gain issues (such as contentious workers' compensation claims) is a poor candidate. The success of tenolysis is intrinsically linked to the patient's postoperative effort; performing this complex procedure on a non-compliant patient will inevitably result in recurrent adhesions, profound stiffness, and a deeply dissatisfied patient.
| Category | Indications for Flexor Tendon Tenolysis | Contraindications for Flexor Tendon Tenolysis |
|---|---|---|
| Clinical Status | Significant discrepancy between Passive ROM and Active ROM | Active or latent deep space infection |
| Therapy Progress | Functional plateau (no improvement for 4-8 weeks) | Severe, unyielding passive joint contractures |
| Tissue Integrity | Intact, continuous flexor tendon (FDS and/or FDP) | Attenuated, ruptured, or structurally incompetent tendon |
| Soft Tissue Envelope | Healed, pliable skin with resolved inflammation | Poor soft tissue coverage (adherent scars, skin grafts directly on tendon) |
| Skeletal/Vascular | Consolidated fractures, stable skeletal architecture | Unhealed fractures, severe digital ischemia, insensate digit |
| Patient Factors | Highly motivated, compliant, understands rehab demands | Poor compliance, lack of motivation, severe cognitive impairment |
Pre-Operative Planning, Templating, and Patient Positioning
The foundation of a successful flexor tendon tenolysis is laid long before the patient enters the operating theater. Meticulous preoperative planning begins with an exhaustive clinical examination designed to isolate the specific anatomical source of the motion deficit. The surgeon must systematically differentiate flexor tendon adhesions from intrinsic muscle contractures, extensor mechanism tethering, and capsular stiffness. The Bunnell intrinsic tightness test is mandatory; limited PIP flexion with the MP joint extended, which improves when the MP joint is flexed, confirms intrinsic tightness rather than isolated flexor adhesions. Similarly, the Landsmeer test is utilized to evaluate the oblique retinacular ligament. A thorough assessment of the "seesaw effect"—where passive flexion of one joint forces extension of another—can help localize non-articular contractures.
Advanced imaging modalities, while not always strictly necessary, serve as invaluable adjuncts in complex cases. High-resolution plain radiographs are mandatory to evaluate joint congruity, rule out occult osteoarthritis, assess for malunited fractures that may be impinging on the flexor sheath, and identify the presence and position of any retained orthopedic hardware. In cases where the continuity of the flexor tendon is questionable, dynamic ultrasound or Magnetic Resonance Imaging (MRI) can provide critical diagnostic clarity. Ultrasound offers the advantage of real-time, dynamic assessment of tendon excursion and can effectively map the extent of adhesions. MRI, particularly with contrast, can differentiate between dense scar tissue and a partial tendon rupture, allowing the surgeon to prepare for a potential tendon reconstruction or grafting procedure if the tendon is found to be non-viable intraoperatively.
The timing of the tenolysis is a critical variable that significantly impacts the ultimate outcome. The consensus among master hand surgeons is that a minimum of 3 to 6 months must elapse following the initial tendon repair or grafting procedure. This mandatory waiting period allows the initial biological cascade of fibroplasia to subside, the scar tissue to mature and soften, and the inflammatory hyperemia to resolve. Intervening prematurely, while the tissue is still in the active remodeling phase, virtually guarantees a massive recurrence of adhesions and a worse functional outcome. The surgeon must exercise patience and rely on the clinical indicators of a soft tissue equilibrium—pliable scars, absence of edema, and a definitive plateau in therapy—rather than an arbitrary timeline.
Patient positioning and the choice of anesthesia are paramount to the intraoperative strategy. The patient is positioned supine with the affected extremity extended on a radiolucent hand table. A lead hand splint is frequently utilized to stabilize the wrist and unaffected digits, providing a rigid platform for micro-dissection. The modern gold standard for flexor tendon tenolysis is the Wide Awake Local Anesthesia No Tourniquet (WALANT) technique. By utilizing a mixture of lidocaine for anesthesia and epinephrine for hemostasis, the surgeon avoids the need for a proximal tourniquet and general anesthesia. This revolutionary approach allows the patient to remain fully conscious and comfortable, enabling them to actively flex and extend their digits on command during the procedure. This real-time, active intraoperative feedback is invaluable; it allows the surgeon to visualize the exact mechanical restrictions, confirm the complete release of all adhesions, ensure the integrity of the tendon under physiological load, and immediately demonstrate the restored motion to the patient, which serves as a powerful psychological motivator for the grueling postoperative rehabilitation ahead.
Step-by-Step Surgical Approach and Fixation Technique
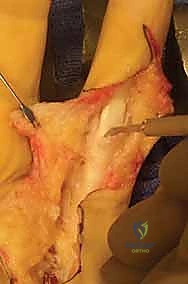
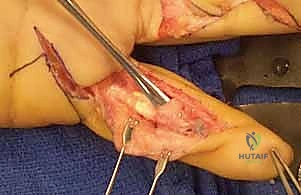
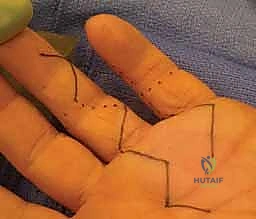
The surgical execution of a flexor tendon tenolysis demands a meticulous, almost microscopic attention to detail, balancing aggressive scar resection with absolute respect for the surviving anatomy. The incision is typically planned utilizing either a classic Brunner zigzag approach or a midaxial incision, depending on the location of previous scars and the anticipated need for exposure. The Brunner approach provides unparalleled, wide exposure of the entire volar aspect of the digit and flexor sheath, allowing for comprehensive visualization. Regardless of the incision chosen, the initial dissection must be deliberate and superficial, elevating thick fasciocutaneous flaps to preserve the subdermal vascular plexus. The immediate priority upon entering the subcutaneous space is the definitive identification, mobilization, and protection of the digital neurovascular bundles. In a heavily scarred digit, these structures are frequently displaced or encased in dense fibrosis. Utilizing loupe magnification and blunt dissection techniques, the nerves and arteries must be traced from normal, unscarred proximal tissue into the zone of injury, securing them with vessel loops to prevent iatrogenic transection.
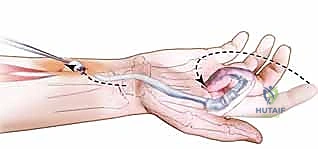
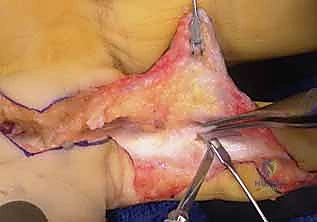
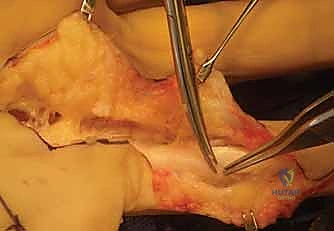
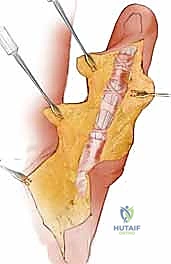
Once the neurovascular structures are safeguarded, attention is turned to the flexor tendon sheath. The objective is to systematically release the tendons from the surrounding scar while meticulously preserving the critical A2 and A4 pulleys. Adhesiolysis begins at the proximal and distal margins of the scar bed, working from areas of relatively normal anatomy toward the epicenter of the adhesions. Specialized instruments, such as Freer elevators, right-angle dural hooks, and specifically designed tenolysis knives, are employed. The surgeon must slide the elevator along the volar, medial, and lateral aspects of the tendon, sequentially breaking the fibrous tethers. Extreme caution must be exercised when dissecting the dorsal aspect of the tendon to avoid obliterating the delicate vincula brevia and longa, which provide the essential vascular supply. If dense adhesions are encountered at the level of the A2 or A4 pulleys, the surgeon must carefully dilate the pulley using a blunt probe or perform a very limited, step-cut lengthening of the pulley edge, absolutely avoiding complete division, which would result in catastrophic bowstringing.
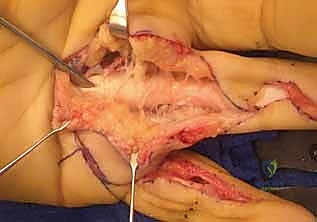
As the adhesions are systematically dismantled, the surgeon must continuously assess the structural integrity of the flexor tendons. It is not uncommon to uncover partial tears, severe fraying, or attenuation of the tendon substance that was previously masked by the dense scar tissue. If a partial tear involving less than 30-40% of the tendon cross-sectional area is identified, it should be meticulously debrided and the epitenon repaired with fine, non-absorbable monofilament suture (e.g., 6-0 Prolene) to restore a smooth gliding surface and prevent mechanical catching. However, if the tendon is found to be severely attenuated, structurally incompetent, or completely ruptured, the primary tenolysis must be aborted. The surgeon must pivot seamlessly to a salvage procedure, typically involving the excision of the scarred tendon remnant and the placement of a silicone Hunter rod to initiate the first stage of a two-stage tendon reconstruction.
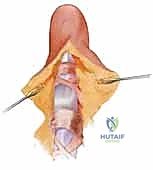
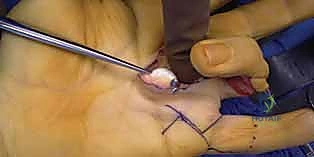
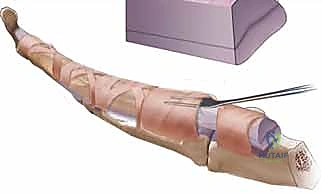
The culmination of the procedure under the WALANT technique is the intraoperative active motion test. The patient is instructed to actively flex and extend the digit with maximum effort. The surgeon observes the excursion of the tendon, looking for any residual tethering, mechanical impingement at the pulley margins, or signs of bowstringing. If full, unhindered active flexion is achieved, the tenolysis is deemed complete. Meticulous hemostasis is then achieved using bipolar electrocautery to minimize postoperative hematoma formation, which is a potent stimulus for recurrent scarring. The wound is irrigated copiously, and the skin is closed with non-absorbable sutures, avoiding any deep dermal sutures that could inadvertently tether the underlying tendon. A bulky, non-compressive soft dressing is applied, and the patient is immediately transitioned to the postoperative rehabilitation phase.
Complications, Incidence Rates, and Salvage Management
Despite flawless surgical execution, flexor tendon tenolysis carries a formidable risk profile, and complications are an ever-present reality. The most devastating intraoperative or early postoperative complication is iatrogenic tendon rupture. The incidence of tendon rupture following tenolysis is reported to range from 4% to 10% in major clinical series. This catastrophic event typically occurs when a tendon, already compromised by prior trauma and rendered relatively avascular by the dense scar bed, is subjected to the aggressive mechanical forces of adhesiolysis or early postoperative active motion. Furthermore, overzealous sharp dissection that inadvertently scores the tendon substance significantly drastically reduces its tensile strength. When an intraoperative rupture occurs, or if the tendon is deemed too attenuated to withstand rehabilitation, the primary tenolysis is immediately converted to a staged tendon reconstruction with the insertion of a silicone active tendon implant (Hunter rod) to preserve the pulley system and create a pseudosheath for future grafting.
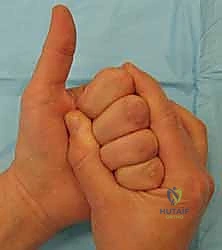
The most frequent complication, and the primary reason for a "failed" tenolysis, is the recurrence of dense adhesions and subsequent digital stiffness. The incidence of recurrent stiffness requiring further intervention or resulting in permanent functional deficit can be as high as 15% to 30%. This recurrence is driven by the profound biological response to surgical trauma; the very act of excising scar tissue incites a new wave of hemorrhage, inflammation, and fibroplasia. Hematoma formation within the dead space created by the tenolysis serves as a rich scaffold for fibroblast proliferation. Management of recurrent stiffness relies heavily on aggressive, sustained hand therapy, static progressive splinting, and serial casting. Revision tenolysis is generally discouraged unless a specific, newly formed mechanical block is identified, as repeated surgical insults yield diminishing returns and exponentially increase the risk of tendon rupture and neurovascular compromise.
Iatrogenic neurovascular injury is a profound risk due to the distorted anatomy. Digital nerve transection or severe traction neuropraxia occurs in approximately 2% to 5% of cases. Intraoperative recognition of a nerve transection mandates immediate microsurgical primary epineurial repair. Missed nerve injuries result in painful neuromas and debilitating loss of protective sensation. Vascular compromise, leading to digital ischemia or necrosis, is rare but can occur if both digital arteries are damaged or if the digit is subjected to excessive tension in the presence of compromised venous outflow. Postoperative infection is another critical concern, occurring in 1% to 3% of patients. Deep space infections within the flexor sheath require emergent operative debridement, copious irrigation, and targeted intravenous antibiotic therapy; failure to aggressively manage an infection will inevitably result in complete destruction of the tendon and the gliding mechanism.

In cases where tenolysis fails unequivocally, either due to recurrent intractable adhesions, tendon rupture, or severe joint contracture, the surgeon must be prepared to offer definitive salvage procedures. If the soft tissue envelope and pulley system remain viable, a two-stage tendon reconstruction utilizing a free tendon graft (e.g., palmaris longus or plantaris) remains the gold standard. However, in older patients, those with severely damaged joint surfaces (osteoarthritis or post-traumatic arthrosis), or those unwilling to undergo further complex reconstructions, arthrodesis of the PIP or DIP joint in a functional position provides a stable, pain-free digit, albeit with sacrificed motion. In the most severe cases of a stiff, painful, and non-functional digit that interferes with the overall utility of the hand, ray amputation may be the most pragmatic and definitive salvage option, allowing the patient to return to their life without the burden of a dysfunctional appendage.
| Complication | Estimated Incidence | Primary Etiology / Risk Factors | Salvage / Management Strategy |
|---|---|---|---|
| Tendon Rupture | 4% - 10% | Attenuated tendon, overzealous dissection, aggressive early therapy | Staged tendon reconstruction (Hunter rod), Arthrodesis |
| Recurrent Adhesions | 15% - 30% | Hematoma, inadequate hemostasis, poor therapy compliance | Aggressive therapy, static progressive splinting, rarely revision |
| Digital Nerve Injury | 2% - 5% | Distorted anatomy, dense scar encased bundles | Immediate microsurgical epineurial repair |
| Pulley Incompetence | 1% - 3% | Iatrogenic division of A2/A4, excessive dilation | Pulley reconstruction (graft), ring splinting |
| Deep Space Infection | 1% - 3% | Contamination, poor soft tissue envelope | Emergent I&D, IV antibiotics, removal of necrotic tissue |
| Complex Regional Pain Syndrome | 2% - 7% | Uncontrolled post-op pain, nerve traction | Multidisciplinary pain management, stellate ganglion blocks |
Phased Post-Operative Rehabilitation Protocols
The surgical execution of a flexor tendon tenolysis represents only the preliminary step in the restoration of digital function; the ultimate success of the procedure is unequivocally dictated by the rigor and precision of the postoperative rehabilitation protocol. It is a widely accepted axiom among hand surgeons that a tenolysis is "50% surgery and 50% therapy." The fundamental objective of the rehabilitation phase is to maintain the tendon glide achieved intraoperatively while mitigating the inevitable biological cascade of edema, inflammation, and recurrent fibroplasia. This requires a delicate, highly supervised balance between aggressive mobilization to prevent adhesions and careful load management to prevent tendon rupture or inflammatory flare-ups. The protocol must be initiated immediately; delaying therapy even by 24 to 48 hours allows organizing hematoma to consolidate into nascent adhesions, severely compromising the surgical outcome.
Phase I of the rehabilitation protocol encompasses the first 0 to 2 weeks postoperatively and is characterized by immediate, controlled active motion. Within 24 hours of surgery, the bulky surgical dressing is removed, and the patient is fitted with a custom, lightweight thermoplastic splint, typically a dorsal blocking splint or a resting hand splint, depending on the surgeon's preference and the specific intraoperative findings. The cornerstone of Phase I is the initiation of active and active-assisted range of motion exercises. Patients are instructed to perform specific tendon gliding exercises—straight fist, hook fist, and full composite fist—multiple times every hour while awake. Edema control is paramount during this phase, utilizing elevation, compressive wrapping (e.g., Coban), and retrograde massage. Excessive edema increases tissue hydrostatic pressure, restricts venous and lymphatic return, and acts as a biological glue that promotes fibrosis. Pain management must be aggressive, utilizing multimodal analgesia, as severe pain will inhibit the patient's ability to perform the necessary active excursions.
Phase II, spanning from 2 to 6 weeks postoperatively, marks a transition toward more intensive mobilization and scar management. As the surgical wounds heal and the initial inflammatory phase subsides, the therapy protocol becomes more vigorous. Blocking exercises are introduced to isolate the excursion of the FDS and FDP tendons independently. If the patient is struggling to achieve full passive extension due to early flexor tightness, dynamic or static progressive extension splinting may be cautiously implemented, carefully monitoring the digit for signs of excessive tension or vascular compromise. Scar management becomes a primary focus; aggressive deep friction massage, silicone gel sheeting, and elastomer pads are utilized to soften the maturing scar tissue and prevent it from tethering to the underlying tendon sheath. The therapist must continuously assess the patient's progress, adjusting the intensity of the exercises based on the tissue's response—pushing hard enough to break micro-adhesions but backing
