Congenital Knee Dislocation Repair: An Intraoperative Masterclass in Pediatric Orthopaedic Surgery

Key Takeaway
This masterclass details surgical repair of congenital knee dislocation (CDK). Fellows will learn comprehensive anatomy, meticulous intraoperative techniques including percutaneous, mini-open, and extensile V-to-Y quadriceps advancements. We cover preoperative planning, detailed execution, critical pearls, and postoperative management, ensuring optimal outcomes for this challenging pediatric deformity. Gain a granular understanding of every surgical micro-step.
Comprehensive Introduction and Patho-Epidemiology
Congenital Dislocation of the Knee (CDK) represents one of the most visually striking and biomechanically profound deformities encountered in pediatric orthopaedic surgery. Distinct from a simple positional recurvatum—which often resolves spontaneously or with minimal intervention—true CDK involves a fixed, rigid anterior displacement of the tibia relative to the distal femur. If left unrecognized or inadequately treated, this pathology severely compromises the development of normal joint kinematics, obliterates the child’s potential for physiological ambulation, and leads to cascading deformities in the ipsilateral hip and foot. As a rare clinical entity, with an estimated incidence of 1 in 100,000 live births, CDK demands a high index of suspicion, a thorough understanding of its multifactorial etiology, and a meticulous, stepwise approach to surgical reconstruction.
The patho-epidemiology of CDK is broadly stratified into isolated (idiopathic) cases and those associated with underlying syndromic or neuromuscular conditions. Isolated CDK is frequently attributed to intrauterine packaging issues, such as oligohydramnios, breech presentation, or a lack of intrauterine space, which mechanically forces the fetal knee into prolonged hyperextension. Conversely, syndromic CDK is intimately linked with connective tissue disorders and neuromuscular dysplasias, most notably Larsen syndrome, Arthrogryposis Multiplex Congenita (AMC), and myelomeningocele. In these syndromic patients, the dislocation is rarely an isolated phenomenon; it is typically accompanied by congenital hip dysplasia (DDH) and rigid clubfoot (talipes equinovarus), creating a triad of lower extremity deformities that require highly coordinated, staged surgical management.
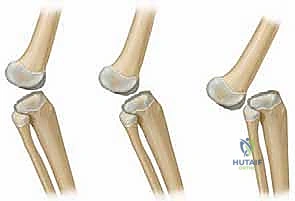
Clinically, the severity of the deformity is best understood through the lens of the Curtis and Fisher classification system, which delineates the pathology into three distinct grades. Grade I represents congenital hyperextension, where the articular surfaces remain in contact, and the deformity can often be managed with serial casting. Grade II denotes anterior subluxation, characterized by partial loss of articular congruity and a distinct mechanical block to flexion. Grade III is the frank, complete anterior dislocation of the tibia on the femur, a rigid deformity that almost universally necessitates extensile surgical release. In Grade III presentations, the clinician will invariably note deep transverse skin creases over the anterior aspect of the knee—a hallmark sign of chronic, fixed hyperextension that has impeded normal soft tissue expansion during fetal development.
Understanding the natural history of CDK is paramount for operative decision-making. While Grade I hyperextension may respond to conservative measures such as gentle manipulation and Pavlik harness application, Grades II and III involve profound structural alterations that resist non-operative correction. The chronicity of the dislocation leads to secondary morphological changes in the cartilaginous anlagen of the distal femur and proximal tibia. The anterior femoral condyles become flattened, and the tibial plateau may develop an anterior slope, further destabilizing the joint. Therefore, timely surgical intervention—typically performed between 6 and 12 months of age—is critical to reduce the joint concentrically, stimulate normal chondral modeling, and provide the child with a stable, functional limb for the onset of independent ambulation.
Detailed Surgical Anatomy and Biomechanics
To execute a successful reconstruction of a congenital knee dislocation, the orthopaedic surgeon must possess an intimate understanding of the radically altered pathoanatomy and paradoxical biomechanics that define this condition. The primary anatomical culprit, and the central focus of our surgical release, is the quadriceps femoris muscle complex. In the setting of CDK, the quadriceps is not merely contracted; it is profoundly dysplastic. The muscle belly is often hypoplastic, heavily fibrosed, and abnormally adherent to the anterior cortex of the distal femur. This dense adherence acts as a rigid, unyielding tether, physically preventing the proximal migration of the patella and blocking any attempt at knee flexion. The quadriceps tendon itself is markedly shortened and thickened, losing its normal viscoelastic properties.
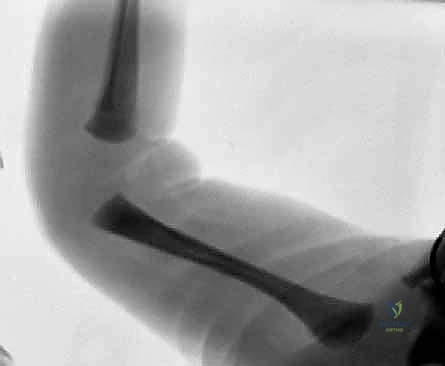
The patellofemoral articulation is significantly distorted. Because the patella is entirely cartilaginous in infants and thus radiolucent on standard radiographs, its position must be inferred clinically and confirmed via ultrasonography or direct intraoperative visualization. In CDK, the patella is almost universally displaced laterally and proximally, resting high on the lateral femoral condyle. Furthermore, the suprapatellar pouch—the crucial synovial recess that normally unfolds to accommodate knee flexion—is frequently hypoplastic or completely obliterated by intra-articular adhesions. The anterior knee articular capsule is markedly tight, contracted, and thickened, forming a secondary mechanical barrier that must be meticulously released to achieve concentric reduction of the tibiofemoral joint.
The biomechanical alterations extend to the posterior structures of the knee, creating a paradoxical functional paradigm. The hamstring muscles, which typically function as primary knee flexors, are frequently subluxated anteriorly past the mechanical axis of the knee joint. Due to this anterior translation, the hamstrings undergo a functional inversion, acting as secondary knee extensors and actively exacerbating the recurvatum deformity. The cruciate ligaments exhibit high variability; they may be attenuated, elongated, or entirely absent, particularly in syndromic cases. However, despite their morphological abnormalities, the cruciates are rarely the primary block to reduction. The menisci, interestingly, are usually present and morphologically normal, though they may be compressed or distorted by the abnormal tibiofemoral contact pressures.

Navigating the neurovascular anatomy in an infant with CDK requires exceptional vigilance, as the chronic dislocation alters the normal course and tension of critical structures. Medially, the saphenous nerve, its infrapatellar branch, and the great saphenous vein lie superficially and are vulnerable during medial skin flap elevation. Deeper, the femoral artery and vein transition through the adductor hiatus to become the popliteal vessels. Laterally, the common peroneal nerve is at significant risk; it courses around the fibular neck, and its superficial and deep branches can be easily compromised during lateral retinacular releases or division of a tight iliotibial band. Posteriorly, the popliteal artery, popliteal vein, and tibial nerve are intimately associated with the posterior capsule. The greatest risk to these posterior structures is not direct surgical transection, but rather a severe traction injury (neurapraxia or vascular intimal tear) that can occur during the acute reduction of a chronically dislocated, shortened limb.
Exhaustive Indications and Contraindications
The decision to proceed with surgical intervention for Congenital Dislocation of the Knee is dictated by the severity of the deformity, the patient's underlying syndromic status, and the response to initial conservative management. Surgical reconstruction is unequivocally indicated for infants presenting with Grade II (subluxation) or Grade III (complete dislocation) CDK who have failed a rigorous, 4-to-6-week trial of conservative therapy. Conservative measures typically involve serial casting, gentle manipulation, or the use of a Pavlik harness, aimed at gradually stretching the anterior structures. When these modalities fail to achieve a stable, concentric reduction with at least 90 degrees of passive flexion, operative intervention becomes mandatory to prevent irreversible cartilaginous deformation and functional disability.
Furthermore, the threshold for surgical intervention is significantly lowered in patients with syndromic associations, such as Arthrogryposis Multiplex Congenita or Larsen syndrome. In these populations, the soft tissues are inherently rigid and fibrotic, rendering conservative management almost universally ineffective. Early surgical release—often performed between 3 and 6 months of age—is advocated in these complex cases to facilitate the concurrent or subsequent management of associated deformities, particularly developmental dysplasia of the hip (DDH). A reduced, flexed knee is a biomechanical prerequisite for the successful treatment of DDH, as it relaxes the hamstrings and allows for proper positioning in a spica cast or harness.
Contraindications to surgical repair are relatively few but absolute when present. Active local or systemic infection precludes any elective orthopaedic reconstruction. Severe medical comorbidities, such as unstable congenital heart defects or profound respiratory compromise (often seen in severe syndromic presentations), may necessitate delaying the procedure until the infant is medically optimized for general anesthesia. Additionally, in older, neglected cases of CDK presenting in late childhood or adolescence, isolated soft tissue releases are generally contraindicated. In these skeletally mature or near-mature patients, the adaptive bony changes are fixed, and attempting a soft tissue reduction will inevitably result in severe neurovascular compromise; these patients require complex bony procedures, such as distal femoral shortening osteotomies, to achieve reduction safely.
| Category | Specific Criteria | Clinical Rationale / Management Impact |
|---|---|---|
| Absolute Indications | Failed conservative management (4-6 weeks) | Persistent anterior tethering leads to irreversible joint dysplasia. |
| Absolute Indications | Grade III Complete Dislocation | Mechanical block precludes any spontaneous resolution. |
| Relative Indications | Syndromic CDK (Arthrogryposis, Larsen) | Early surgery required due to extreme soft tissue rigidity. |
| Relative Indications | Concomitant severe DDH | Knee must be flexed to treat the hip dysplasia effectively. |
| Absolute Contraindications | Active systemic or local infection | High risk of septic arthritis and catastrophic failure. |
| Absolute Contraindications | Severe cardiorespiratory instability | Anesthetic risks outweigh the benefits of early joint reduction. |
| Relative Contraindications | Neglected presentation in older children | Soft tissue release alone will cause neurovascular stretch injury; requires osteotomy. |
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the cornerstone of a successful CDK reconstruction, bridging the gap between clinical assessment and intraoperative execution. The evaluation begins with high-quality, orthogonal plain radiographs of the affected limb. Anteroposterior (AP) and true lateral views of the knee are essential, though obtaining a true lateral can be challenging due to the severe recurvatum. These radiographs confirm the direction and magnitude of the tibiofemoral dislocation and allow the surgeon to assess the ossification centers of the distal femur and proximal tibia. While the patella remains unossified and invisible on plain films in this age group, the overall alignment and any secondary bony dysplasias (such as anterior sloping of the tibial plateau) must be carefully documented.
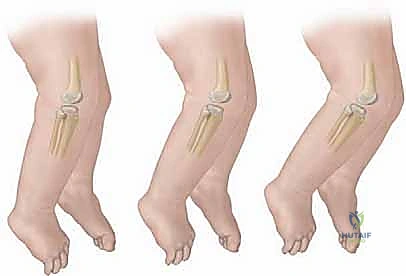
Advanced imaging modalities, particularly high-resolution ultrasonography, have become invaluable adjuncts in the preoperative workup of infantile CDK. Ultrasound provides real-time, dynamic visualization of the unossified cartilaginous structures. It allows the surgeon to accurately map the position of the laterally displaced patella, assess the depth of the trochlear groove, and evaluate the integrity of the extensor mechanism prior to making an incision. Magnetic Resonance Imaging (MRI) is rarely required for isolated, idiopathic cases, but it may be indicated in complex syndromic presentations to delineate severe intra-articular anomalies, such as absent cruciate ligaments or blended meniscocapsular structures, which could alter the surgical approach.
Patient positioning and operating room setup must be orchestrated with precision, keeping the unique physiological vulnerabilities of the infant in mind. The patient is placed supine on a fully radiolucent operating table to facilitate unimpeded fluoroscopic imaging. Given the infant's delicate skin and immature thermoregulatory system, meticulous padding of all bony prominences—specifically the occiput, sacrum, and bilateral heels—is mandatory to prevent pressure necrosis. A forced-air warming blanket is deployed over the upper body to maintain strict normothermia throughout the procedure, mitigating the risks of coagulopathy and delayed anesthetic emergence. The C-arm fluoroscopy unit is positioned to enter from the contralateral side, allowing for rapid AP and lateral imaging without requiring repositioning of the limb.
Crucially, the entire operative lower extremity, from the iliac crest down to the tips of the toes, is prepped and draped freely into the sterile field. This extensive draping is vital; it permits the surgeon to manipulate the hip and ankle, assessing the global tension of the biarticular muscles (rectus femoris and hamstrings) during the reduction maneuvers. A tourniquet is strictly avoided in this procedure. The application of a tourniquet in an infant with CDK is contraindicated for several reasons: it artificially tethers the quadriceps mechanism, obscuring the true extent of the contracture; it distorts the delicate anatomical landmarks; and it introduces a significant risk of ischemic injury to tissues that are already compromised by chronic dislocation and impending surgical stretch. Hemostasis is instead achieved through meticulous, sharp dissection and the judicious use of bipolar electrocautery.
Step-by-Step Surgical Approach and Fixation Technique
The surgical correction of a rigid Congenital Dislocation of the Knee demands an extensile approach to systematically address every contracted structure blocking flexion. Scalpel in hand, the surgeon must choose between a midline longitudinal incision and a serpentine incision. While a midline approach offers direct access, the serpentine incision is vastly superior in pediatric joint reconstructions. By curving the incision—starting from the tibial tubercle, sweeping medially over the joint line, and extending laterally into the distal thigh—the surgeon prevents the formation of a linear, longitudinal scar that could contract and recreate an extension tether as the child grows.
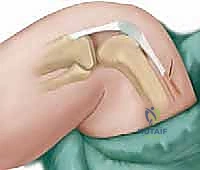
1. Exposure and Mobilization of the Extensor Mechanism
Following the skin incision, thick subcutaneous flaps are developed and elevated meticulously to preserve the delicate subdermal vascular plexus, minimizing the risk of postoperative skin necrosis. The dissection proceeds down to the deep fascia overlying the quadriceps mechanism. The fascial layer is incised longitudinally, revealing the underlying rectus femoris and vastus musculature. At this juncture, the profound pathology of the quadriceps becomes visually apparent: the muscle belly is invariably hypoplastic, fibrotic, and densely adherent to the anterior aspect of the distal femur. The patella, often found resting superolaterally, is identified, and the shortened patellar tendon is traced distally to its insertion on the tibial tubercle.
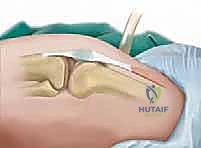
2. The V-to-Y Quadriceps Advancement
The crux of the reconstruction is the V-to-Y quadriceps lengthening, a powerful technique designed to restore length to the extensor mechanism while maintaining its continuity. An inverted V-shaped incision is mapped out over the quadriceps tendon and distal muscle belly. The apex of the "V" is positioned proximally, extending high into the central tendon of the rectus femoris, while the two limbs diverge distally, terminating just proximal to the superior pole of the patella.
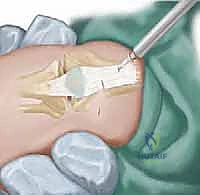
The surgeon first sharply detaches the medial and lateral fibers of the vastus medialis and lateralis from the central rectus tendon. Following this, a comprehensive transverse release of the medial and lateral retinacula is performed, extending from the borders of the patella down to the respective collateral ligaments. This step is critical to centralize the laterally tracking patella. Next, the posterior borders of the vastus lateralis and medialis are divided sharply. Using a blunt periosteal elevator, the surgeon meticulously dissects the central quadriceps muscle flap off the anterior femur. This subperiosteal elevation breaks the dense fibrotic adhesions tethering the muscle to the bone, allowing the entire extensor mechanism to slide proximally.
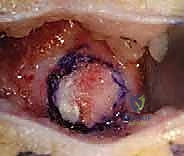
3. Capsular Release and Joint Reduction
Even with a fully mobilized quadriceps, the anterior knee joint capsule remains a formidable barrier to reduction. A wide, transverse anterior capsulotomy is executed, extending from the medial collateral ligament to the lateral collateral ligament. The surgeon must exercise extreme caution during this step to stay superficial to the anterior horns of the menisci and avoid iatrogenic scoring of the delicate cartilaginous femoral condyles.
With the anterior structures released, the surgeon gently manipulates the limb. An assistant provides inline traction while the surgeon translates the tibia posteriorly and flexes the knee. If a mechanical block persists, associated deformities must be addressed. Frequently, the tibia presents in rigid valgus and external rotation, driven by a contracted iliotibial (IT) band. In such instances, a transverse fractional lengthening or complete division of the IT band is performed laterally, which rapidly corrects the rotational and angular malalignment, allowing the tibia to seat concentrically beneath the femur.
4. Extensor Mechanism Repair and Closure
Once concentric reduction is achieved and confirmed via intraoperative fluoroscopy, the knee is ranged to ensure at least 90 to 110 degrees of passive flexion. The knee is then held immobilized in approximately 30 to 40 degrees of flexion—the optimal biomechanical angle for repairing the extensor mechanism.
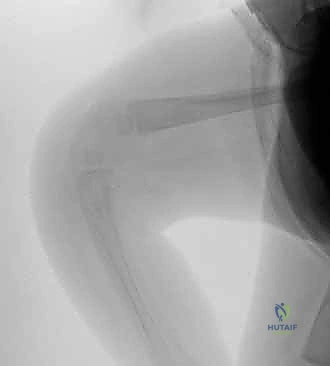
The V-shaped proximal flap of the quadriceps is now advanced distally and sutured into the Y-shaped defect created by the release. This repair must be exceptionally robust to withstand the forces of postoperative rehabilitation. Heavy, non-absorbable braided sutures (e.g., 2-0 or 3-0 Ethibond) are utilized, employing a locking Krackow or modified Kessler technique to securely grasp the tendinous fibers.
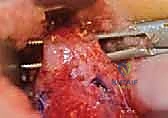
Crucially, while the central tendon is repaired, the medial and lateral retinacula are intentionally left open. Attempting to close the retinacular defects would instantly recreate the anterior tether and compromise the flexion gained during the procedure. The subcutaneous tissues are approximated with interrupted absorbable sutures, and the skin is closed with a running subcuticular stitch or interrupted nylon, depending on the tension of the flaps.
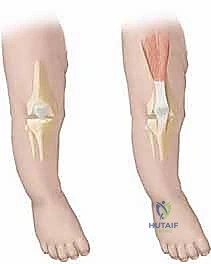
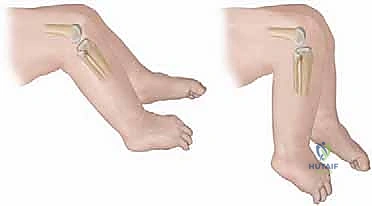
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, the extensile nature of the CDK repair in an infant carries a distinct profile of perioperative and long-term complications. The surgeon must be prepared to identify and manage these issues promptly to preserve the functional outcome of the limb. One of the most immediate and devastating complications is ischemic skin necrosis over the anterior knee. This occurs due to the sudden stretching of the chronically contracted anterior skin envelope during postoperative flexion, compounded by surgical disruption of the subdermal plexus. The use of a serpentine incision mitigates this risk, but if blanching is noted upon closure, the knee must be extended slightly to relieve tension, and the degree of postoperative casting flexion must be reduced.
Neurovascular compromise, specifically stretch neurapraxia of the common peroneal nerve, is a recognized complication when correcting severe valgus and external rotation deformities. The nerve, having adapted to the shortened, dislocated state, is suddenly subjected to significant tension upon anatomic reduction. Postoperatively, the limb must be monitored closely for signs of ischemia or neurological deficit. If a profound, persistent neurapraxia is suspected, immediate removal of the cast and slight extension of the knee may be required to relieve tension on the nerve.
Long-term complications are predominantly biomechanical. Recurrence of the anterior subluxation or hyperextension deformity can occur if the initial release was inadequate, if the V-Y repair fails, or if the postoperative splinting protocol is non-compliant. Conversely, an over-lengthened quadriceps mechanism can result in a persistent extensor lag, leading to a crouch-gait pattern as the child begins to ambulate. Furthermore, because surgery is performed in close proximity to the distal femoral and proximal tibial physes, iatrogenic physeal arrest is a rare but catastrophic complication that can lead to progressive angular deformities or limb length discrepancies, requiring close radiographic surveillance throughout the child's growth.
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage Management / Prevention |
|---|---|---|---|
| Anterior Skin Necrosis | 5% - 10% | Excessive tension on skin flaps during flexion; midline incisions. | Prevent with serpentine incision. Manage with wound care, delayed grafting if severe. Decrease cast flexion. |
| Peroneal Neurapraxia | 2% - 5% | Acute stretch during reduction of severe valgus/ER deformity. | Usually transient. Prevent by avoiding excessive acute correction. Manage with observation and AFO support. |
| Recurrent Hyperextension | 10% - 15% | Inadequate capsular release; failure of V-Y repair; poor splinting. | Requires revision soft tissue release or, in older children, distal femoral flexion osteotomy. |
| Extensor Lag / Weakness | 15% - 20% | Over-lengthening of quadriceps; prolonged immobilization. | Intensive physical therapy. Rarely requires surgical plication of the extensor mechanism. |
| Physeal Growth Arrest | < 2% | Iatrogenic injury to cartilaginous anlagen during dissection. | Monitor with serial scanograms. Manage with epiphysiodesis of contralateral limb or limb lengthening procedures. |
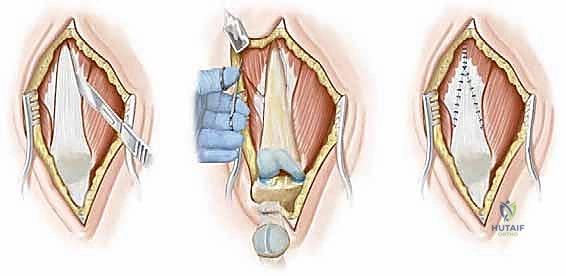
Phased Post-Operative Rehabilitation Protocols
The success of a congenital knee dislocation repair relies heavily on a rigid, phased postoperative rehabilitation protocol. The surgical release merely provides the anatomical potential for motion; it is the postoperative management that secures the functional outcome. Immediately following wound closure in the operating room, the limb is immobilized in a well-padded, long-leg cast. The knee is positioned in the maximum amount of flexion that allows for adequate distal perfusion and avoids undue tension on the anterior skin closure—typically between 45 and 90 degrees of flexion. This cast serves to protect the V-Y advancement repair and maintain the reduced position of the tibia.
Phase 1 of rehabilitation encompasses the first 4 to 6 weeks postoperatively. During this period, strict immobilization in the long-leg cast is maintained. The child is monitored closely for any signs of neurovascular compromise or skin breakdown at the cast edges. After 6 weeks, the cast is bivalved or transitioned to a custom-molded, removable thermoplastic splint.
Phase 2 (weeks 6 to 12) marks the initiation of gentle, passive range-of-motion (ROM) exercises. The parents and physical therapists are instructed to remove the splint multiple times a day to perform passive flexion exercises, aiming to maintain the ROM achieved intraoperatively. Crucially, passive extension is strictly avoided to protect the healing quadriceps repair. The splint must be worn at all times when exercises are not being performed, particularly during sleep, to prevent recurrent anterior subluxation.
Phase 3 (3 to 6 months) introduces
