Pediatric Sacral Fracture & Neurological Deficit: A Detailed Trauma Case Study
Key Takeaway
Pediatric sacral fractures with neurological deficits present with severe pain, lower extremity weakness, perineal numbness, and absent sacral reflexes. Diagnosis involves thorough clinical neurological examination, CT for fracture morphology, and MRI to confirm nerve root compression, edema, or hemorrhage, all crucial for guiding surgical decompression and stabilization planning.
Patient Presentation and History
An 8-year-old male presented to the Level I pediatric trauma center emergency department following a high-energy motor vehicle versus pedestrian incident. The patient was struck by a standard passenger sedan traveling at an estimated velocity of 40 km/h. Eyewitness reports indicated the primary point of impact was the posterior pelvis and sacral region, resulting in the patient being thrown several meters. The mechanism suggests a significant axial loading and shear force vector applied directly to the sacropelvic architecture, a kinematic profile highly associated with complex pelvic ring disruptions and neurovascular compromise.
Upon arrival, the patient was conscious, alert, and oriented, with a Glasgow Coma Scale of 15. He was in significant distress, reporting severe pain in the lower back and bilateral gluteal regions, quantified as 9/10 on an age-appropriate visual analogue scale. Initial Advanced Trauma Life Support evaluation confirmed a patent airway, bilateral breath sounds, and stable hemodynamics. His initial vital signs demonstrated a blood pressure of 105/65 mmHg, a heart rate of 98 beats per minute, and a respiratory rate of 20 breaths per minute. Fluid resuscitation was initiated with warmed crystalloid per pediatric trauma protocols, though no massive transfusion protocol was required.
The history of present illness was notable for a profound inability to mobilize the lower extremities or tolerate any degree of weight-bearing. The patient reported acute onset numbness in the perineal region and bilateral posterior thighs. Furthermore, he reported an inability to initiate micturition since the time of the accident. While acute urinary retention can be secondary to pain and psychological distress in the pediatric trauma patient, the concomitant perineal numbness raised immediate clinical suspicion for a significant sacral neurological injury involving the S2 through S4 nerve roots.
A thorough review of systems and past medical history revealed a previously healthy child with no prior history of spinal deformities, congenital neurological deficits, coagulopathies, or metabolic bone disease. The patient was up-to-date on all childhood immunizations, and there was no family history contributory to skeletal dysplasias or hereditary neuropathies. The absence of pre-existing conditions isolated the current clinical presentation entirely to the acute traumatic event, necessitating a highly focused evaluation of the lumbopelvic junction and the cauda equina.
Clinical Examination Findings
The secondary survey was conducted with meticulous attention to the lumbopelvic region and the lower extremity neurological axes. Inspection of the posterior torso and pelvis revealed significant soft tissue swelling, extensive ecchymosis, and a central superficial abrasion overlying the S2 through S4 dermatomes. The presence of significant fluctuance over the posterior sacrum and bilateral gluteal regions raised immediate concern for a developing Morel-Lavallée lesion, a closed degloving injury resulting from the shearing of the subcutaneous tissue away from the underlying deep fascia. There were no open wounds communicating with the fracture site, and perineal inspection revealed no lacerations or gross hematuria at the urethral meatus.
Palpation of the anterior pelvic ring, including the anterior superior iliac spines, iliac crests, and pubic symphysis, elicited no significant tenderness or palpable step-offs. A gentle compression-distraction maneuver of the iliac wings was performed; however, aggressive manipulation was strictly deferred to prevent exacerbation of the suspected sacral fracture and secondary iatrogenic injury to the already compromised sacral plexus. Tenderness to palpation was exquisitely localized over the entire sacrum and the right sacroiliac joint complex.
Neurological examination of the lower extremities was critical, though challenging due to the patient's acute pain and anxiety. A detailed motor and sensory assessment was performed following the American Spinal Injury Association guidelines, adapted for pediatric compliance. Gross motor function demonstrated profound, asymmetric weakness. Hip extension and abduction were graded 2/5 bilaterally, while hip flexion was 3/5. Knee extension mediated by the L3 and L4 nerve roots, and ankle dorsiflexion mediated by the L4 and L5 nerve roots, were preserved at 4/5 bilaterally. However, ankle plantarflexion mediated by the S1 and S2 nerve roots, and toe flexion mediated by the S2 and S3 nerve roots, were significantly impaired, graded at 2/5 bilaterally.
Sensory examination revealed a distinct pattern of deficit. Light touch and pinprick discrimination were preserved in the L1 through L5 dermatomes. However, there was a marked decrease in sensation within the S2 through S5 dermatomes bilaterally, specifically involving the posterior thighs, the perianal region, and the plantar aspects of the feet.
A digital rectal examination was performed, revealing significantly diminished voluntary anal sphincter tone. The bulbocavernosus reflex was completely absent. Plantar reflexes were bilaterally downgoing, and there was no evidence of clonus or spasticity, consistent with a lower motor neuron or nerve root level injury rather than a central spinal cord lesion. The constellation of bilateral S1-S3 motor weakness, S2-S5 sensory deficits, diminished sphincter tone, and absent bulbocavernosus reflex strongly indicated a severe sacral plexopathy or cauda equina syndrome secondary to sacral root compression or avulsion.
Vascular examination demonstrated palpable and symmetric 2+ dorsalis pedis and posterior tibial pulses bilaterally. Capillary refill was brisk at less than two seconds in all digits, indicating no major macrovascular compromise to the lower extremities. Compartment compartments of the lower legs were soft and compressible.
Imaging and Diagnostics
The initial radiographic evaluation included standard anteroposterior pelvic, inlet, and outlet views. The anteroposterior radiograph demonstrated asymmetric widening of the right sacroiliac joint space and an ill-defined, complex fracture line extending through the right sacral ala, disrupting the superior sacral arcuate lines.
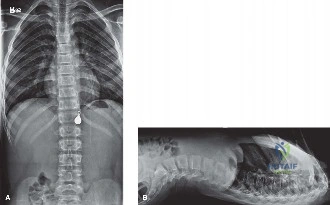
The pelvic inlet view provided further clarification of the posterior pelvic ring disruption, demonstrating posterior displacement and internal rotation of the right hemipelvis relative to the axial skeleton, alongside severe comminution within the sacral ala. The pelvic outlet view revealed vertical translation of the right hemipelvis, indicating a highly unstable, multi-planar disruption of the posterior pelvic ring architecture.
Given the high-energy mechanism, the complex radiographic presentation, and the profound neurological deficits, a high-resolution multidetector computed tomography scan of the pelvis with fine axial cuts and three-dimensional surface-shaded reconstructions was immediately obtained. The computed tomography scan is the gold standard for defining the osseous morphology of sacral fractures, assessing lumbopelvic stability, and evaluating the neural foramina for osseous encroachment.
The computed tomography scan provided critical three-dimensional information, revealing a highly complex, comminuted fracture involving the right sacral ala and extending centrally into the sacral body. The fracture pattern involved Denis Zone I, II, and III. Specifically, the fracture propagated through the right sacral neuroforamina at the S1, S2, and S3 levels, with significant osseous fragments retropulsed into the central sacral canal, severely compromising the space available for the cauda equina and the exiting sacral nerve roots.
Furthermore, the computed tomography scan demonstrated a transforaminal component on the left side, indicating a U-shaped or H-shaped spino-pelvic dissociation variant, though the primary displacement and comminution were concentrated on the right. The L5-S1 facet joints were evaluated for subluxation or fracture, which would further indicate lumbopelvic instability; the right L5-S1 facet joint demonstrated widening and capsular disruption.
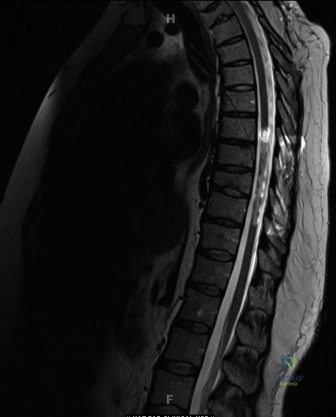
Due to the severe neurological deficits and the need to differentiate between neural compression by osseous fragments, acute hematoma, or nerve root avulsion, a magnetic resonance imaging scan of the lumbosacral spine and pelvis was obtained. Magnetic resonance imaging is paramount in the pediatric population to evaluate the cartilaginous components of the sacrum, the integrity of the ligamentous structures, and the exact status of the neural elements.
The magnetic resonance imaging sequences, including short tau inversion recovery and T2-weighted axial and sagittal images, confirmed severe compression of the S1 through S4 nerve roots bilaterally within the central canal and the right neural foramina by retropulsed bone and a localized epidural hematoma. Importantly, the magnetic resonance imaging demonstrated continuity of the nerve roots, ruling out complete anatomic avulsion (neurotmesis) and suggesting that the neurological deficit was primarily secondary to acute compression and contusion (neuropraxia and axonotmesis), thereby reinforcing the indication for urgent surgical decompression.
Differential Diagnosis
The presentation of a pediatric patient with high-energy pelvic trauma and acute lower extremity neurological deficits requires a systematic and rapid differentiation of several critical pathologies. The primary goal is to distinguish between osseous instability, central nervous system injury, and peripheral nerve root compromise, as the management algorithms and timing of intervention vary significantly.
| Diagnostic Consideration | Clinical Presentation | Imaging Findings | Differentiating Factors |
|---|---|---|---|
| Complex Sacral Fracture with Neural Compression | Severe pelvic/lower back pain, asymmetric or symmetric lower extremity weakness (typically L5-S4 distribution), bowel/bladder dysfunction, saddle anesthesia. | CT shows sacral central/foraminal fractures (Denis Zone II/III). MRI confirms osseous or hematoma compression of nerve roots. | Direct correlation between the osseous injury level and the specific dermatomal/myotomal deficits. Mechanical instability is present. |
| Spinal Cord Injury Without Radiographic Abnormality | Motor and sensory deficits, potentially transient or progressive. Hyperreflexia or spasticity (if upper motor neuron) or flaccidity (during spinal shock). | Normal plain radiographs and CT scan. MRI reveals spinal cord edema, contusion, or hemorrhage without fracture or dislocation. | Absence of osseous injury on CT. Neurological level often higher than the lumbosacral junction. Common in pediatric cervical/thoracic spine due to ligamentous laxity. |
| Pelvic Ring Disruption without Sacral Fracture | Pelvic instability, pain with compression/distraction. Leg length discrepancy. Potential for severe hemodynamic instability. | Widening of the pubic symphysis, sacroiliac joint disruption (Tile B or C) without major sacral osseous comminution. | Neurological deficits are less common unless there is a traction injury to the lumbosacral plexus. Sparing of the central sacral canal. |
| Cauda Equina Syndrome secondary to Lumbar Trauma | Bilateral radicular pain, saddle anesthesia, asymmetric lower extremity paraplegia, loss of bowel/bladder control. | Lumbar burst fracture or massive acute disc herniation on CT/MRI compressing the thecal sac above the L5 level. | Pathology is located superior to the sacrum. Sacral osseous architecture is intact. |
| Lumbosacral Plexus Avulsion | Profound, flaccid paralysis of the affected lower extremity. Complete loss of sensation in the affected distribution. | MRI shows pre-ganglionic nerve root avulsion, pseudomeningoceles. Sacral fractures may or may not be present. | Irreversible neurological deficit. Decompression is not indicated as the nerve is anatomically transected from the spinal cord. |
In this specific case, the presence of a complex Denis Zone III sacral fracture with corresponding MRI evidence of neural compression by retropulsed bone and hematoma definitively confirmed the diagnosis of a complex sacral fracture with secondary cauda equina and sacral plexus compression.
Surgical Decision Making and Classification
The management of pediatric sacral fractures is complex and highly individualized, as the pediatric pelvic ring possesses significant remodeling potential and a robust periosteal hinge that often permits non-operative management of minimally displaced fractures. However, the presence of progressive or severe neurological deficits, open fractures, severe lumbopelvic instability, or significant pelvic deformity dictates surgical intervention.
The fracture in this patient was classified using the Denis classification system for sacral fractures. The Denis classification divides the sacrum into three zones based on the location of the fracture relative to the neural foramina. Zone I fractures involve the sacral ala lateral to the foramina and rarely cause neurological deficits. Zone II fractures involve the neural foramina and have a moderate risk of radiculopathy. Zone III fractures involve the central sacral canal and carry the highest risk of severe neurological deficits, including bowel and bladder dysfunction. This patient presented with a complex fracture involving Zones I, II, and III, with the Zone III component driving the profound neurological presentation.
Furthermore, the fracture pattern was evaluated for spino-pelvic dissociation. The presence of a transverse fracture line communicating with bilateral transforaminal or longitudinal fractures creates a functional dissociation between the axial skeleton (spine) and the pelvic ring. The Isler classification was utilized to assess lumbopelvic stability based on the fracture's relationship to the L5-S1 facet joint. A fracture line exiting lateral to the facet joint maintains lumbopelvic stability, while a fracture line exiting medial to or through the facet joint results in lumbopelvic instability. The computed tomography imaging in this case demonstrated a fracture line compromising the right L5-S1 facet articulation, indicating a highly unstable lumbopelvic junction requiring robust fixation.
The decision to proceed with operative intervention was absolute. The primary indication was the acute, severe neurological deficit involving bowel and bladder function (S2-S4 roots) and lower extremity motor function (S1-S2 roots) in the setting of documented mechanical compression. The surgical objectives were threefold:
1. Immediate decompression of the central sacral canal and the involved neural foramina to maximize the potential for neurological recovery.
2. Anatomic reduction of the sacral fracture and the pelvic ring to restore pelvic volume, alignment, and biomechanics.
3. Rigid internal fixation to stabilize the lumbopelvic junction, facilitate early mobilization, and prevent progressive deformity.
The timing of surgery in the setting of neurological deficit and pelvic trauma is a subject of ongoing academic debate. However, the consensus in the presence of acute cauda equina syndrome or progressive sacral root compression is emergent or urgent decompression, ideally within 24 to 48 hours of injury, once the patient is hemodynamically optimized and life-threatening concomitant injuries have been addressed.
Surgical Technique and Intervention
The patient was taken to the operating room and placed under general endotracheal anesthesia. Multimodality intraoperative neuromonitoring was established, including somatosensory evoked potentials, motor evoked potentials, and continuous spontaneous electromyography of the lower extremities and the external anal sphincter. The use of neuromonitoring is critical during sacral fracture reduction and fixation to detect and prevent iatrogenic injury to the lumbosacral plexus during manipulation and hardware placement.
The patient was carefully positioned prone on a radiolucent Jackson spinal table. All bony prominences were meticulously padded, and the abdomen was allowed to hang free to decrease intra-abdominal pressure and minimize epidural venous bleeding. The entire posterior spine, pelvis, and bilateral lower extremities were prepped and draped in a standard sterile fashion.

A posterior midline approach was utilized, centered over the lower lumbar spine and extending down to the sacrococcygeal junction. The paraspinal musculature was elevated subperiosteally to expose the L4 through S3 posterior osseous elements. Extreme care was taken during the exposure of the sacrum due to the severe comminution and the risk of plunging into the central canal.
The first phase of the procedure focused on neural decompression. A wide sacral laminectomy was performed from S1 through S3. The ligamentum flavum and epidural hematoma were carefully evacuated. The dural sac and the exiting sacral nerve roots were identified. The retropulsed osseous fragments from the central sacral body and the right neural foramina were carefully mobilized and removed using micro-curettes and Kerrison rongeurs. The S1, S2, and S3 nerve roots were traced bilaterally out to their respective foramina, ensuring complete circumferential decompression. Neuromonitoring signals remained stable throughout the decompression phase.
Following adequate decompression, the focus shifted to reduction and stabilization. Given the lumbopelvic dissociation and the patient's pediatric anatomy, a lumbopelvic fixation construct was selected to bypass the comminuted sacrum and transfer the load directly from the lumbar spine to the ilium.
Pedicle screws were placed bilaterally at the L4 and L5 levels under fluoroscopic guidance, utilizing standard anatomic trajectories. Iliac screws were then placed bilaterally. The starting point for the iliac screws was identified at the posterior superior iliac spine. A burr was used to create a cortical window, and a gearshift probe was advanced down the dense osseous corridor above the greater sciatic notch, aiming toward the anterior inferior iliac spine. The trajectory was confirmed in multiple planes using intraoperative fluoroscopy to ensure the screws remained entirely intraosseous and did not breach the sciatic notch or the acetabulum.
Once the pedicle and iliac anchors were secured, contoured titanium rods were seated into the screw heads. Reduction of the sacral kyphosis and the vertical translation of the right hemipelvis was achieved through a combination of manual traction on the lower extremities, cantilever reduction techniques using the rods, and direct manipulation of the iliac wings.
To directly address the sacral ala fracture and provide transverse stability to the pelvic ring, the placement of an iliosacral screw was evaluated. In the pediatric population, the upper sacral segments often exhibit significant cartilaginous components and potential sacral dysmorphism, making the safe osseous corridor for an iliosacral screw exceedingly narrow. Intraoperative three-dimensional fluoroscopy (O-arm) was utilized to map the S1 osseous corridor. A safe corridor was identified, and a single 6.5mm partially threaded cannulated screw was placed percutaneously from the right ilium, across the sacroiliac joint, and into the S1 vertebral body, achieving excellent compression across the fracture site.
Final intraoperative imaging confirmed anatomic reduction of the pelvic ring, optimal hardware placement without neuroforaminal breach, and restoration of lumbopelvic alignment. The surgical site was copiously irrigated. A subfascial closed suction drain was placed, and the wound was closed in multiple layers to ensure a watertight fascial closure and eliminate dead space, mitigating the risk of postoperative infection or seroma formation.
Post Operative Protocol and Rehabilitation
The immediate postoperative phase was conducted in the pediatric intensive care unit for continuous hemodynamic and neurological monitoring. The patient was kept strictly flat in bed for the first 24 hours to minimize the risk of dural leak and to allow the acute soft tissue swelling to subside.
Neurological examinations were performed serially. By postoperative day two, the patient demonstrated early signs of neurological recovery, with improved sensation to light touch in the S2 and S3 dermatomes and the return of a weak but detectable voluntary anal sphincter contraction. Motor function in the lower extremities remained stable but weak, with hip extension and plantarflexion showing slight improvement to a grade 3/5 by postoperative day five.
Deep vein thrombosis prophylaxis is a complex consideration in pediatric trauma patients. Given the high-energy mechanism, the pelvic fracture, and the prolonged immobility, mechanical prophylaxis with sequential compression devices was initiated immediately. Chemical prophylaxis with low molecular weight heparin was initiated on postoperative day two, following confirmation of stable postoperative hemoglobin and the absence of epidural hematoma expansion on a postoperative computed tomography scan.
The rehabilitation protocol was strictly phased to protect the fixation construct while maximizing functional recovery.
* Phase I (Weeks 0-6): The patient was restricted to non-weight-bearing status on the bilateral lower extremities. Mobilization was limited to bed-to-wheelchair transfers using a sliding board and strict log-rolling techniques. A comprehensive bowel and bladder management program was instituted, utilizing intermittent catheterization and a bowel regimen to prevent distension and autonomic dysreflexia. Passive and active-assisted range of motion exercises for the lower extremities were initiated to prevent contractures.
* Phase II (Weeks 6-12): Following radiographic confirmation of early callus formation and maintenance of hardware alignment at the six-week mark, the patient was advanced to touch-down weight-bearing. Intensive physical therapy focused on core strengthening, upper extremity conditioning, and progressive lower extremity motor re-education. Hydrotherapy was utilized to facilitate movement in a reduced-gravity environment.
* Phase III (Months 3-6): Weight-bearing was progressively advanced to full weight-bearing as tolerated, contingent upon radiographic evidence of solid osseous union. Gait training, proprioceptive exercises, and functional movement patterns were emphasized.
Long-term follow-up is critical in the pediatric population following severe pelvic trauma. The patient requires serial clinical and radiographic evaluations until skeletal maturity to monitor for potential complications, including physeal arrest of the triradiate cartilage (if involved), asymmetric pelvic growth leading to leg length discrepancy, and the development of post-traumatic scoliosis secondary to lumbopelvic hardware. Hardware removal is generally considered in pediatric patients once solid fusion is achieved, typically 12 to 18 months postoperatively, to prevent restriction of subsequent spinal growth.
Clinical Pearls and Pitfalls
The management of pediatric sacral fractures with neurological deficits is fraught with diagnostic and therapeutic challenges. The following clinical pearls and pitfalls are critical for the orthopedic trauma surgeon:
- Pitfall: Underestimating the Cartilaginous Anatomy. The pediatric pelvis contains a significant volume of unossified cartilage. Plain radiographs and standard computed tomography scans may severely underestimate the true extent of the displacement and the involvement of the sacroiliac joint or neurocentral synchondroses. Magnetic resonance imaging is an indispensable adjunct in these cases.
- Pearl: The Importance of the Sphincter Exam. In the acute trauma setting, a detailed lower extremity motor exam may be limited by pain. The digital rectal examination assessing voluntary tone and the bulbocavernosus reflex provides objective, critical data regarding the integrity of the S2-S4 roots and dictates the urgency of surgical intervention.
- Pitfall: Missed Lumbopelvic Instability. A sacral fracture is rarely an isolated injury in high-energy trauma. Failure to recognize a spino-pelvic dissociation (e.g., a U-type fracture extending through the L5-S1 facets) will lead to catastrophic failure if treated with isolated iliosacral screws. Lumbopelvic fixation is mandated for these highly unstable patterns.
- Pearl: Intraoperative 3D Imaging is Mandatory. The placement of iliosacral screws in a pediatric patient with a dysmorphic or severely comminuted sacrum carries a high risk of iatrogenic nerve injury. The use of intraoperative navigation or three-dimensional fluoroscopy (O-arm) significantly increases the safety and accuracy of screw placement in narrow osseous corridors.
- Pitfall: Neglecting the Soft Tissue Envelope. High-energy shear forces to the posterior pelvis frequently result in Morel-Lavallée lesions. Failure to identify and appropriately manage these degloving injuries prior to or during surgical incision significantly increases the risk of deep surgical site infection and hardware colonization.
- Pearl: Multidisciplinary Rehabilitation. Neurological recovery following severe sacral plexopathy is unpredictable and prolonged. Early integration of physical therapy, occupational therapy, urology, and neuro-rehabilitation specialists is paramount to optimizing the patient's long-term functional outcome and quality of life.
