Mastering Orthopaedic Trauma: Rehabilitation, External Fixation, and Infection Management
Key Takeaway
Postoperative rehabilitation in orthopaedic trauma requires a delicate balance between early joint mobilization and soft tissue preservation. The use of Ilizarov and spatial external fixators provides optimal biomechanical stability while allowing axial micromotion to promote osteogenesis. Managing postoperative complications, particularly MRSA infections, relies on aggressive debridement and strategic implant retention. Tailored weight-bearing protocols ensure optimal fracture union and functional recovery.
PRINCIPLES OF ORTHOPAEDIC TRAUMA REHABILITATION
The rehabilitation of the orthopaedic trauma patient is a dynamic, multiphase process that must begin immediately following surgical stabilization. The overarching goal is to restore maximal functional capacity while respecting the biological and biomechanical constraints of the healing fracture and surrounding soft tissue envelope. The timing and aggressiveness of rehabilitation are entirely dependent on the fracture pattern, the rigidity of the fixation construct, and the stability of the soft tissues.
Early Mobilization vs. Soft Tissue Stability
In the management of complex extremity trauma, a fundamental tension exists between the orthopaedic imperative to mobilize adjacent joints and the plastic surgical requirement to protect healing soft tissues. We advocate that adjacent joints should be mobilized as soon as possible to prevent arthrofibrosis and capsular contracture. However, in the setting of severe open fractures (e.g., Gustilo-Anderson Types IIIB and IIIC), the premature motion of musculotendinous units gliding over exposed or healing fracture surfaces can severely irritate the soft tissues, disrupt neoangiogenesis, and significantly decrease the local tissue resistance to infection.
To mitigate these risks, the surgical team must frequently incorporate the immobilization of adjacent joints using custom splints, functional braces, or dedicated foot/hand attachments integrated directly into external fixation systems. This prevents debilitating contractures (such as equinus contracture of the ankle) while protecting soft tissue flaps or grafts. Physical therapy protocols must be meticulously prescribed, initiating with active and active-assisted range-of-motion (ROM) exercises for joint mobilization strictly as soon as soft tissue healing permits.
Management of Neurological Deficits
High-energy trauma frequently results in concomitant peripheral nerve injuries, leading to transient or permanent neurological deficits and subsequent loss of active motor function. These deficits must be rigorously evaluated clinically and electrodiagnostically (EMG/NCS) when appropriate. Joints lacking active motor control are at an exceptionally high risk for rapid, rigid contracture formation. The appropriate joints must be continuously splinted in functional, neutral positions (e.g., the ankle at 90 degrees of dorsiflexion in the setting of a common peroneal nerve palsy) to avoid fixed deformities that would compromise future reconstructive efforts or tendon transfers.
Clinical Pearl: Always anticipate the "equinus drift" in lower extremity trauma. A patient with a tibial shaft fracture treated with an external fixator will naturally rest their foot in plantarflexion. Without a foot plate or dynamic splinting, a rigid Achilles contracture will develop within weeks, severely complicating the eventual return to weight-bearing.
THE ILIZAROV AND CIRCULAR EXTERNAL FIXATION SYSTEMS
Professor Gavriil Ilizarov revolutionized the treatment of complex orthopaedic pathologies—including highly comminuted fractures, recalcitrant nonunions, osteomyelitis, and severe limb deformities—through the development of his innovative modular circular external fixator utilizing tensioned transfixing wires.
Biomechanics and Frame Dynamics
The biomechanical genius of the Ilizarov system lies in its ability to manipulate the mechanical environment of the fracture site. In recent years, there has been a paradigm shift in external fixation design, moving toward hybrid and half-pin frames, and maintaining the external fixator as the definitive stabilization device until complete union of unstable fractures is achieved.
A critical biomechanical tradeoff exists in external fixation: the need for initial absolute rigidity to maintain anatomic fracture alignment and minimize the risk of infection in open fractures, versus the biological need for controlled axial micromotion to stimulate endochondral ossification and prevent atrophic nonunion.
Although the classic Ilizarov circular frame is only approximately 25% as stiff axially as standard unilateral half-pin fixators, it demonstrates comparable stiffness in bending and shear forces. This unique property—axial compliance combined with bending and shear rigidity—creates the ideal mechanobiological milieu for secondary bone healing.
Several critical factors dictate the overall stability and stiffness of a circular frame:
* Wire Diameter and Tension: These are the most influential factors. Standard 1.8 mm wires tensioned to 110–130 kg provide the foundation of frame rigidity.
* Ring Parameters: The size (diameter), number, and anatomical location of the rings directly affect stiffness. Rings should be sized to allow a two-finger breadth (approx. 2 cm) clearance circumferentially to accommodate postoperative edema.
* Wire Divergence: Transfixing wires should ideally intersect at 90-degree angles to maximize multiplanar stability. Anatomical constraints (safe corridors) often limit this, but angles less than 60 degrees significantly compromise stability.
* Olive Wires: The strategic use of stopper (olive) wires provides interfragmentary compression and prevents translation of the bone on the wire.
* Loading Conditions: Distraction or compression loads applied at the fracture or nonunion site alter the tension within the construct.
Intrinsic biomechanical factors unique to each patient must also be calculated, including the patient's body weight, the degree of cortical continuity (comminution), and the integrity of the surrounding soft tissue hinge.
Indications and Advantages in High-Energy Trauma
The Ilizarov and modern spatial external fixators permit the stabilization of high-energy, highly comminuted fractures with minimal additional operative trauma to the already compromised soft tissues. By utilizing percutaneous wire and pin insertion, the critical periosteal and endosteal blood supply is meticulously preserved.
Early functional use of the limb, including progressive weight-bearing, is not only permitted but actively encouraged, as the axial loading stimulates callus formation through the dynamization of the frame. Furthermore, the Ilizarov technique frequently eliminates the need for massive internal hardware in contaminated fields, drastically reducing the risk of deep-seated osteomyelitis.
SURGICAL TECHNIQUE: APPLICATION OF CIRCULAR AND SPATIAL FRAMES
The application of a circular or spatial frame requires meticulous preoperative planning, precise anatomical knowledge of safe neurovascular corridors, and intraoperative adaptability.
Step-by-Step Frame Application
- Patient Positioning and Preparation: The patient is positioned supine on a radiolucent Jackson or fracture table. The limb is prepped and draped widely to allow unrestricted access for fluoroscopy and frame manipulation.
- Ring Sizing and Assembly: Pre-assembled frames or individual rings are selected based on preoperative templating. The rings must accommodate anticipated swelling.
- Reference Wire Placement: A reference wire is placed parallel to the joint line (e.g., the knee or ankle) in the metaphyseal segment. This wire dictates the orientation of the entire frame.
- Tensioning: The reference wire is secured to the ring and tensioned using a dynamometer to the appropriate load (typically 130 kg for adult tibial metaphysis).
- Orthogonal Fixation: Additional wires and half-pins are inserted through safe corridors. In the femur, fractures should be stabilized with at least six pins due to the massive deforming forces of the thigh musculature.
- Short Segment Fixation: When dealing with a short periarticular fragment at the hip or knee, the surgeon must use at least three pins in a multiplane configuration to achieve adequate fixation and prevent toggling.
- Final Tightening and Verification: Tighten all pin attachments, bolts, and wire fixation clamps. Add additional half-pins and longitudinal strut supports as necessary to maximize stability.
Surgical Warning: Thermal necrosis during wire or pin insertion is a leading cause of pin-tract infection and subsequent loosening. Always use sharp drill bits, drill sleeves, and copious cold saline irrigation during insertion. Never plunge the drill through the far cortex.

FIGURE 53-46 Application of spatial-frame external fixator with use of image intensification. The integration of real-time fluoroscopy ensures precise wire placement within safe anatomical corridors, avoiding critical neurovascular structures while maximizing cortical purchase.
Hybrid Techniques: Combining Internal and External Fixation
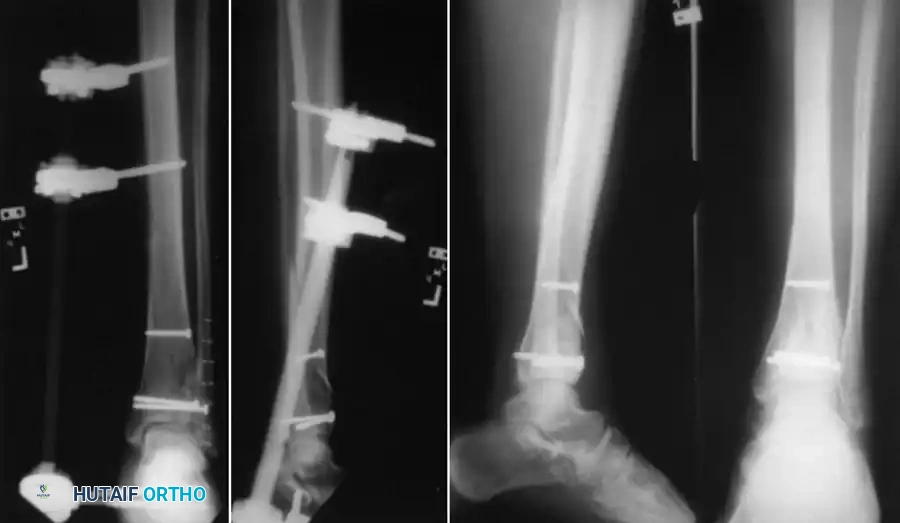
In complex intra-articular fractures (e.g., tibial plateau or pilon fractures), the joint surface must be anatomically reduced and rigidly fixed to prevent post-traumatic arthrosis. This is often achieved using limited internal fixation (independent lag screws) to reconstruct the articular block, followed by the application of a circular external fixator to neutralize the metaphyseal-diaphyseal dissociation.
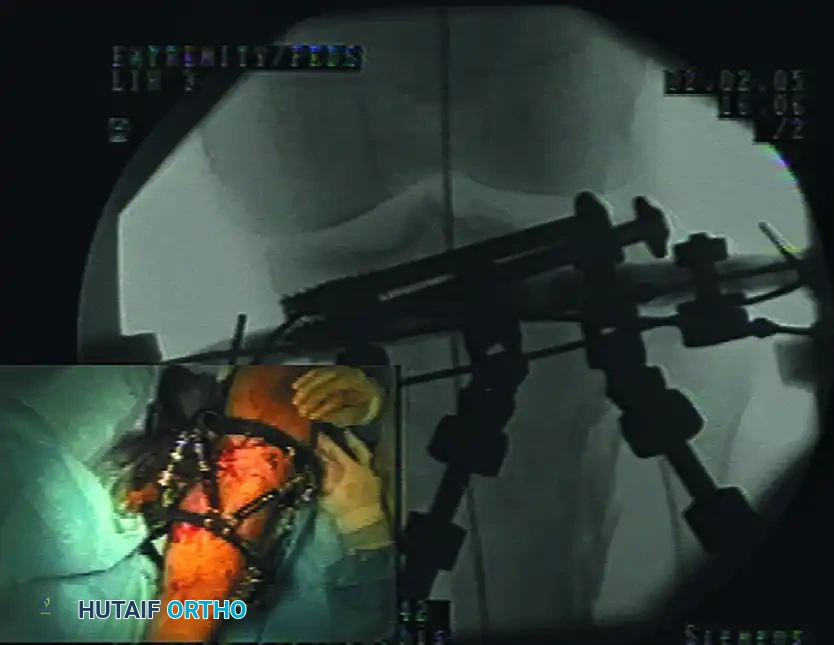
FIGURE 53-45 Screw fixation of articular fragment combined with external fixation. This hybrid approach minimizes soft tissue stripping while providing absolute stability to the articular surface and relative stability to the diaphysis.
POSTOPERATIVE COMPLICATIONS: SURGICAL SITE INFECTIONS AND MRSA
Surgical site infections (SSIs) remain one of the most devastating complications in orthopaedic trauma. The incidence is significantly elevated in trauma patients compared to elective orthopaedic populations.
Epidemiology and Risk Factors
The reported rate of Methicillin-Resistant Staphylococcus aureus (MRSA) infections in trauma patients is approximately 11%, which is nearly double the rate reported in general orthopaedic patients (4% to 5.6%).
Extensive epidemiological studies have identified several critical risk factors for the development of deep MRSA infections at the surgical site:
* Known MRSA carrier status at the time of admission.
* Presence of a hip fracture.
* Advancing age (associated with an almost 2% increase in relative risk per year).
* Peripheral vascular disease.
* Chronic obstructive pulmonary disease (COPD).
* Admission to an intensive care unit (ICU).
* The presence of an open traumatic wound.
Diagnostic Biomarkers: The Role of C-Reactive Protein (CRP)
Early diagnosis of postoperative infection is paramount. The measurement of serum C-reactive protein (CRP) levels has proven highly valuable in the diagnosis of infection following the internal or external fixation of fractures.
The normal postoperative kinetics of CRP follow a predictable curve:
1. Elevation: CRP levels increase immediately after surgery.
2. Peak: Levels typically peak on the second postoperative day.
3. Decline: In patients without infection, CRP levels steadily decrease thereafter.
4. Secondary Rise (Pathologic): In patients developing an SSI, a secondary elevation in CRP levels is noted, typically beginning on the fourth day after surgery.
Clinical Pearl: A CRP value of $\ge$ 96 mg/L on the fourth postoperative day is highly predictive of a deep surgical site infection. Surgeons should maintain a low threshold for joint aspiration or surgical exploration if this secondary spike is observed alongside clinical signs of erythema or disproportionate pain.
Management Strategies: Implant Retention vs. Removal
Deep orthopaedic infections must be treated aggressively with repeat, meticulous surgical debridements and appropriate culture-directed systemic antibiotic coverage (usually intravenous, such as Vancomycin or Daptomycin for MRSA).
When an infection occurs in the presence of a skeletal fixation device (plate, intramedullary nail, or external fixator pins), the surgeon faces a complex biological tradeoff between the necessity of bony stability and the detrimental foreign body response.
* The Biofilm Dilemma: Stability is absolutely necessary to allow the host immune system and antibiotics to eliminate the infection and permit fracture union. However, bacteria (especially S. aureus) rapidly form a glycocalyx biofilm on orthopaedic implants, rendering them highly resistant to immune clearance and systemic antibiotics, resulting in persistent infection.
* Implant Removal: If an implant is no longer mechanically needed to maintain bony stability (e.g., the fracture is clinically and radiographically united), it should be removed immediately to eradicate the biofilm nidus.
* Implant Retention: Implants that are still mechanically required for stability should generally be retained until bony union occurs. This concept of "union with infection" is well-documented. A landmark study of 121 patients with early postoperative infections after internal fixation reported a fracture union rate of 71% utilizing a protocol of aggressive operative debridement, retention of stable hardware, and culture-specific suppressive antibiotic treatment.
* Implant Exchange: If the internal hardware is loose or failing in the face of infection, it must be removed. The fracture should then be stabilized by replacing the internal device with another form of fixation, most commonly removing a plate or nail and applying a spanning external fixator.
POSTOPERATIVE WEIGHT-BEARING AND FUNCTIONAL PROTOCOLS
The progression of postoperative weight-bearing is not a standardized timeline; rather, it is a highly individualized prescription that must be continuously adjusted. Weight-bearing should be limited and progressed depending on:
1. The mechanical stability of the surgical fixation.
2. The type of fixation used and its inherent fatigue life (e.g., plates may fail under cyclical loading before bone heals).
3. The systemic condition and compliance of the patient.
Fracture-Specific Weight-Bearing Guidelines
Progression of weight-bearing must be monitored radiographically, looking for evidence of hardware stability and progressive bone regeneration (callus formation).
- Diaphyseal Fractures (Intramedullary Nails): For most diaphyseal fractures without intra-articular extension treated with locked, load-sharing intramedullary nails, we usually allow immediate touch-down weight bearing (TDWB) or even weight-bearing as tolerated (WBAT), depending on the degree of cortical contact and comminution.
- Intra-articular Fractures: Conversely, for complex intra-articular fractures (e.g., tibial plateau, pilon, or distal femur fractures) treated with plates and screws, weight-bearing is strictly prohibited (Non-Weight Bearing - NWB) for a minimum of 10 to 12 weeks (3 months) to prevent articular subsidence. However, early active and passive ROM of the joint is heavily encouraged to nourish the articular cartilage and prevent stiffness.
Long-Term Functional and Vocational Rehabilitation
Range-of-motion and progressive strengthening exercises must be closely monitored and directed by both the attending orthopaedic physician and a specialized physical therapist. However, the ultimate success of the rehabilitation protocol relies heavily on patient compliance. The patient must be thoroughly educated and instructed regarding their personal responsibility in achieving the maximal functional return of the extremity.
Furthermore, orthopaedic trauma often results in life-altering disability. Vocational rehabilitation counseling should be initiated early in the postoperative course. Multidisciplinary support ensures that the patient is not only physically rehabilitated but also provided with the necessary resources and adaptations to enable a productive, meaningful return to society and the workforce.
📚 Medical References
- Agee JM: External fi xation: technical advances based upon multiplanar ligamentotaxis, Orthop Clin North Am 24:265, 1993.
- Anderson LD, Hutchins WC: Fractures of the tibia and fi bula treated with casts and transfi xing pins, South Med J 59:1026, 1966.
- Anderson LD, Hutchins WC, Wright PE, et al: Fractures of the tibia and fi bula treated by casts and transfi xing pins, Clin Orthop Relat Res 105:179, 1974.
- Anderson R: Subtrochanteric and high shaft fractures of the femur—closed reduction based on a new anatomic approach to treatment, South Surg 8:159, 1939.
- Behrens F: External skeletal fi xation, Instr Course Lect 30:112, 1981.
- Behrens F: External fi xation: special indications and techniques, Instr Course Lect 39:173, 1990.
- Behrens F, Johnson W: Unilateral external fi xation: methods to increase and reduce frame stiffness, Clin Orthop Relat Res 241:48, 1989.
- Behrens F, Johnson WD, Koch TW, et al: Bending stiffness of unilateral and bilateral external fi xator frames, Clin Orthop Relat Res 178:103, 1983.
- Bosworth DM: Skeletal distraction, Surg Gynecol Obstet 52:893, 1931.
- Burgess AR: External fi xation in the multiply injured patient, Instr Course Lect 39:229, 1990.
- Caja VL, Larsson S, Kim W, et al: Mechanical performance of the Monticelli-Spinelli external fi xation system, Clin Orthop Relat Res 309:257, 1994.
- Charnley JC: Positive pressure in arthrodesis of the knee joint, J Bone Joint Surg 30B:478, 1948.
- Costa P, Giancecchi F, Cavazzuti A, et al: Internal and external fi xation in complex diaphyseal and metaphyseal fractures of the humerus, Ital J Orthop Traumatol 17:87, 1991.
- Dubravko H, Szrko R, Tomislav T, et al: External fi xation in war trauma management of the extremities—experience from the war in Croatia, J Trauma 37:831, 1994.
- Evanoff M, Strong ML, MacIntosh R: External fi xation maintained until fracture consolidation in the skeletally immature, J Pediatr Orthop 13:98, 1993.
- Faure C, Merloz PH: Transfi xation: atlas of anatomical sections for the external fi xation of limbs, Berlin, 1987, Springer-Verlag. Gaudinez RF, Mallik AR, Szporn M: Hybrid external fi xation of comminuted tibial plateau fractures, Clin Orthop Relat Res 328:203, 1996.
- Green DP: Pins and plaster treatment of comminuted fractures of the distal end of the radius, J Bone Joint Surg 57A:304, 1975.
- Green SA: Complications of pin and wire external fi xation, Instr Course Lect 39:219, 1990.
- Green SA: Pin and wire technique for external fi xation, Instr Course Lect 44:487, 1995.
- Has B, Javanovic S, Wertheimer B, et al: External fi xation as a primary and defi nitive treatment of open limb fractures, Injury 26:245, 1995.
- Helland P, Boe A, Molster AO, et al: Open tibial fractures treated with the Ex-fi -re external fi xation system, Clin Orthop Relat Res 326:209, 1996.
- Hoffmann R: Closed osteosynthesis with special reference to war surgery, Acta Chir Scand 86:235, 1942.
- Hoffmann R: Osteotaxis, Acta Chir Scand 107:72, 1954.
- Ilizarov GA, Soibelman LM: Some clinical and experimental data concerning bloodless lengthening of lower extremities, Exp Kirug Ortop 4:27, 1969.
- Karlstrom G, Olerud S: External fi xation of severe open tibial fractures with the Hoffmann frame, Clin Orthop 180:68, 1983.
- Kershaw CJ, Cunningham JL, Kenwright J: Tibial external fi xation, weight-bearing, and fracture movement, Clin Orthop Relat Res 293:28, 1993.
- Kim NH, Hahn SB, Park HW, et al: The Orthofi x external fi xator for fractures of long bones, Int Orthop 18:42, 1994.
- Krettek C, Haas N, Tscherne H: The role of supplemental lagscrew fi xation for open fractures of the tibial shaft treated with external fi xation, J Bone Joint Surg 73A:893, 1991.
- Kumar A, Whittle AP: Treatment of complex (Schatzker type VI) fractures of the tibial plateau with circular wire external fi xation: retrospective case review, J Orthop Trauma 14:339, 2000.
- Labeeu F, Pasuch M, Toussaint P, et al: External fi xation in war traumatology: report from the Rwandese war (Oct 1, 1990 to Aug 1, 1993), J Trauma 40(suppl 3):223, 1996.
- Lambotte A: The operative treatment of fractures: report of Fractures Committee, Br Med J 2:1530, 1912.
- Marsh JL, Bonar S, Nepola JV, et al: Use of an articulated external fi xator for fractures of the tibial plafond, J Bone Joint Surg 77A:1498, 1995.
- Marsh JL, Smith ST, Do TT: External fi xation and limited internal fi xation for complex fractures of the tibial plateau, J Bone Joint Surg 77A:661, 1995.
- Mears DC: External skeletal fi xation, Instr Course Lect 30:162, 1981.
- Mohr VD, Eickhoff U, Haaker R, et al: External fi xation of open femoral shaft fractures, J Trauma 38:648, 1995.
- Olerud S: Treatment of fractures by the Vidal-Adery method, Acta Orthop Scand 44:516, 1973.
- Oni OO, Capper M, Soutis C: A fi nite element analysis of the effect of pin distribution on the rigidity of a unilateral external fi xation system, Injury 24:525, 1993.
- Remiger AR: Mechanical properties of the pinless external fi xator on human tibiae, Injury 23(suppl 3):28, 1992.
- Remiger AR, Magerl F: The pinless external fi xator—relevance of experimental results in clinical applications, Injury 25(suppl 3):15, 1994.
- Ries MD, Meinhard BP: Medial external fi xation with lateral plate internal fi xation in metaphyseal tibia fractures, Clin Orthop Relat Res 256:215, 1990.
- Shaw DL, Lawton JO: External fi xation for tibial fractures: clinical results and cost effectiveness, J R Coll Surg Edinb 40:344, 1995.
- Sisk TD: External fi xation: historic review, advantages, disadvantages, complications, and indications, Clin Orthop Relat Res 180:15, 1983.
- Sisk TD: General principles and techniques of external skeletal fi xation, Clin Orthop Relat Res 180:96, 1983.
- Stader O: Treating fractures of the long bones with the reduction splint, North Am Vet 20:55, 1939.
- Stamer DT, Schneck R, Staggers B, et al:
