Modified Woodward Repair for Sprengel Deformity: An Intraoperative Masterclass
Key Takeaway
This masterclass details the Modified Woodward Repair for Sprengel deformity, guiding fellows through comprehensive preoperative planning, meticulous patient positioning, and granular intraoperative execution. We emphasize critical anatomical landmarks, neurovascular protection, and precise surgical techniques for scapular reduction and fixation. Extensive pearls, pitfalls, and postoperative management strategies are discussed to ensure optimal patient outcomes and functional improvement.
Welcome, fellows, to the operating theater. Today, we are addressing a highly challenging congenital anomaly: Sprengel deformity. This is not merely an exercise in repositioning a scapula; it is a complex reconstruction aimed at restoring biomechanical function, improving cosmesis, and, most importantly, ensuring the long-term well-being and functional independence of our young patients. This procedure demands meticulous preoperative planning, an intimate understanding of pediatric pathoanatomy, and flawless intraoperative execution. Let us begin this masterclass on the Modified Woodward Repair.
Comprehensive Introduction and Patho-Epidemiology
Sprengel deformity, first described by Eulenberg in 1863 and later popularized by Otto Sprengel in 1891, represents the most common congenital anomaly of the shoulder girdle. It is characterized by a congenitally high-riding, medially rotated, and often hypoplastic scapula. Embryologically, the scapula develops as a cervical appendage at approximately the fifth week of gestation. Normally, it descends from the cervical region to its final thoracic position (spanning the second to the seventh ribs) by the end of the third month of fetal development. In Sprengel deformity, this critical caudal migration is arrested. The exact etiology remains elusive, with prevailing theories suggesting an interruption of embryonic subclavian blood supply, cerebrospinal fluid abnormalities, or a sporadic genetic mutation leading to developmental arrest.
Epidemiologically, Sprengel deformity is a rare condition, though its exact incidence is difficult to quantify due to underreporting of mild, asymptomatic cases. It exhibits a distinct female predominance, with a female-to-male ratio of approximately 3:1. While the majority of cases are sporadic, autosomal dominant inheritance patterns with variable penetrance have been documented in familial clusters. The condition is unilaterally dominant, most frequently affecting the left shoulder, though bilateral involvement occurs in roughly 10% to 30% of documented cases. When bilateral, the deformity is often asymmetric, complicating both the clinical presentation and the surgical reconstruction strategy.
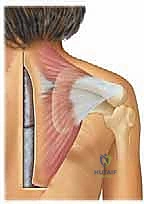
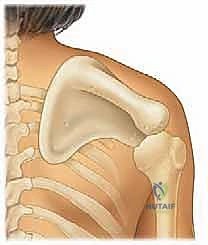
The pathoanatomy of the affected scapula is highly distinct. The bone is typically hypoplastic, exhibiting a decreased vertical diameter and an abnormally wide transverse diameter. The superior angle is frequently hooked or curved anteriorly, hooking over the clavicle or the upper ribs, which creates a significant mechanical block to normal kinematics. Furthermore, the scapula is rotated such that the glenoid faces inferiorly, drastically altering the resting mechanics of the glenohumeral joint. This abnormal position and altered morphology severely restrict both glenohumeral and scapulothoracic motion, particularly limiting forward elevation and active abduction. The periscapular musculature—specifically the trapezius, levator scapulae, and rhomboids—is typically hypoplastic, fibrotic, or partially absent, further compounding the functional deficit.
It is absolutely crucial for the orthopedic surgeon to recognize that Sprengel deformity rarely occurs in a vacuum. It is frequently a component of a broader spectrum of developmental anomalies. We routinely encounter associated conditions such as Klippel-Feil syndrome (cervical vertebral fusion), congenital scoliosis, cervical spina bifida, and diastematomyelia. Rib deformities, including fused or missing ribs, are common. The most notable anatomical association is the presence of an omovertebral bone—a fibrous, cartilaginous, or osseous band connecting the superomedial angle of the scapula to the spinous processes, lamina, or transverse processes of the lower cervical spine (typically C4 to C7). A comprehensive preoperative workup must also screen for visceral anomalies, particularly renal agenesis or tracheoesophageal fistulas, as part of the VACTERL association spectrum.
Detailed Surgical Anatomy and Biomechanics
A profound understanding of the surgical anatomy of the shoulder girdle is the bedrock of a successful Woodward procedure. The scapulothoracic articulation is not a true synovial joint but rather a complex sliding interface between the concave anterior surface of the scapula and the convex posterior thoracic wall. In Sprengel deformity, this interface is disrupted by dense, fibrotic adhesions and the abnormal morphology of the scapula itself. The primary muscular stabilizers—the trapezius, rhomboid major and minor, and levator scapulae—originate from the axial skeleton and insert onto the scapula. In this deformity, these muscles are not only shortened and fibrotic but their vectors of pull are fundamentally altered, tethering the scapula in its elevated, medially rotated position.
The neurovascular anatomy in this region is unforgiving and demands the utmost respect. The spinal accessory nerve (Cranial Nerve XI) is the most critical structure at risk during the Woodward procedure. It exits the jugular foramen, courses through the posterior triangle of the neck, and runs deep to the anterior border of the trapezius, providing its primary motor innervation. In Sprengel deformity, the altered anatomy and dense fibrosis can distort the expected course of this nerve. Similarly, the dorsal scapular nerve (originating from the C5 root) and the dorsal scapular artery course deep to the levator scapulae and rhomboids, running parallel to the medial border of the scapula. Aggressive blind dissection in this medial gutter can lead to devastating denervation of the rhomboids and subsequent scapular winging.
The transverse cervical artery, a branch of the thyrocervical trunk, is another vital structure. It courses laterally across the posterior triangle of the neck, deep to the omohyoid and trapezius muscles, and divides into superficial and deep branches. The deep branch (often synonymous with the dorsal scapular artery) descends along the medial border of the scapula. During the resection of the omovertebral bone and the superomedial angle of the scapula, this artery is highly vulnerable. Inadvertent transection can lead to massive, difficult-to-control hemorrhage that retracts into the deep cervical fascial planes. Meticulous, extraperiosteal dissection and the preemptive identification of these vascular pedicles are mandatory.
Biomechanically, normal shoulder elevation relies on a synchronized scapulothoracic rhythm, typically described as a 2:1 ratio of glenohumeral to scapulothoracic motion. In a patient with Sprengel deformity, the tethering of the scapula by the omovertebral bone and fibrotic musculature obliterates the scapulothoracic contribution. Consequently, the patient relies entirely on glenohumeral motion, which is itself compromised by the inferior tilt of the glenoid. The Woodward procedure aims to restore a more physiological resting position of the glenoid and release the mechanical tethers, thereby recruiting the scapulothoracic articulation back into the kinematic chain. However, because the periscapular muscles are inherently dysplastic, normal biomechanics are rarely fully restored; the goal is a functional optimization rather than anatomical perfection.
Exhaustive Indications and Contraindications
Surgical intervention for Sprengel deformity is highly nuanced and must be tailored to the individual patient's functional deficit, cosmetic deformity, and physiological age. The primary indications for surgery are driven by the Cavendish grading system. Patients presenting with Cavendish Grade III (moderate deformity, shoulder elevated 2-5 cm, obvious asymmetry) and Grade IV (severe deformity, superior angle of the scapula near the occiput) are the principal candidates for surgical reconstruction. Functionally, a significant restriction in shoulder abduction and forward elevation—typically less than 90 to 100 degrees—constitutes a strong indication for release and repositioning to allow the child to perform activities of daily living, such as reaching overhead or combing their hair.
The optimal age for surgical intervention is a subject of critical importance. The consensus in the pediatric orthopedic community is that the procedure should ideally be performed between the ages of 3 and 8 years. Operating on children younger than 3 years is technically challenging due to the small anatomical structures and the lack of sufficient ossification for secure muscle reattachment. Furthermore, the functional demands of a toddler rarely necessitate immediate intervention. Conversely, operating on children older than 8 to 10 years significantly increases the risk of brachial plexus traction injuries. In older children, the brachial plexus has adapted to the shortened, elevated position; pulling the scapula caudally stretches the plexus against the first rib and clavicle, potentially leading to devastating neuropraxia or axonotmesis.
Contraindications to the Woodward procedure must be strictly respected. Absolute contraindications include Cavendish Grade I and mild Grade II deformities where the functional limitation is negligible and the cosmetic appearance is acceptable to the family. Furthermore, patients with severe, life-limiting associated syndromes or profound cardiopulmonary compromise that precludes safe general anesthesia in the prone position are not candidates. Relative contraindications include older age (adolescents and adults), where the risk of neurological injury outweighs the potential functional gains. In such older patients, if surgery is demanded for severe cosmetic reasons, a simple resection of the superomedial border of the scapula without caudal repositioning may be offered as a safer, albeit limited, alternative.
| Category | Specific Criteria | Clinical Rationale |
|---|---|---|
| Primary Indications | Cavendish Grade III or IV | Obvious cosmetic deformity causing psychosocial distress; significant elevation (>2 cm). |
| Severe Functional Limitation | Active shoulder abduction or forward elevation restricted to < 90-100 degrees. | |
| Optimal Age Window (3-8 years) | Balances anatomical size for repair with the plasticity of the neurovascular structures. | |
| Presence of Omovertebral Bone | Mechanical block to motion that requires excision for any functional improvement. | |
| Absolute Contraindications | Cavendish Grade I or Mild II | Risks of major surgery outweigh the minimal functional or cosmetic benefits. |
| Severe Medical Comorbidities | Unsafe for prolonged prone positioning and general anesthesia (e.g., severe congenital cardiac defects). | |
| Relative Contraindications | Age > 8-10 years | Exponentially increased risk of brachial plexus stretch injury during caudal repositioning. |
| Unmanaged Cervical Spine Instability | Concomitant Klippel-Feil with instability must be addressed or protected prior to scapular manipulation. |
Pre-Operative Planning, Templating, and Patient Positioning
Successful execution of the Modified Woodward procedure begins long before the patient enters the operating room. Exhaustive preoperative planning is the foundation of a safe and effective outcome. The clinical evaluation must be meticulous. We obtain full-profile photographs from frontal, posterior, and bilateral lateral views to document the baseline shoulder asymmetry, neck webbing, and the degree of scapular elevation. Dynamic videos of the patient performing shoulder motion activities (abduction, forward elevation, internal/external rotation) are invaluable for postoperative comparison. A thorough neurological examination of the upper extremities is mandatory to establish a baseline, given the inherent risks to the brachial plexus during the procedure.
Imaging is multifaceted and non-negotiable. Standard radiographs, including AP and lateral views of the cervical spine, thoracic spine, and bilateral shoulders, are the first step. These define the bony deformities, measure the exact discrepancy in scapular height, and identify gross associated anomalies like fused ribs or hemivertebrae.
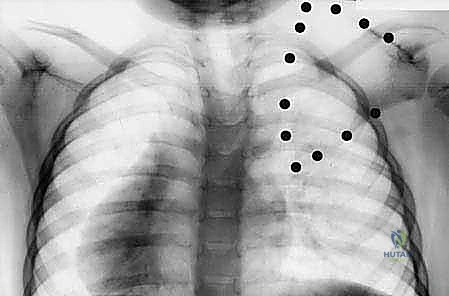
Here, you can clearly see the elevated right scapula on this AP radiograph, demonstrating a classic Cavendish Grade III deformity.
Another radiographic view demonstrating the significant elevation and the medial rotation of the inferior pole.

This radiograph also highlights the characteristic high-riding scapula and associated cervical anomalies.
Beyond plain films, a CT scan with 3D reconstruction is currently considered the gold standard for preoperative templating. It provides unparalleled visualization of the complex 3D bone deformity, specifically delineating the size, origin, and insertion of the omovertebral structure, which is often difficult to fully appreciate on 2D radiographs. Magnetic Resonance Imaging (MRI) of the neuroaxis is essential for evaluating muscle hypoplasia and, critically, ruling out intraspinal anomalies (such as diastematomyelia or tethered cord) which are highly prevalent in this patient population. Finally, renal and cardiac sonography are standard components of our protocol to rule out occult visceral anomalies associated with the VACTERL spectrum.
Intraoperative neuromonitoring is not an option; it is an absolute requirement. Given the proximity to the brachial plexus and the spinal accessory nerve, we utilize continuous somatosensory evoked potentials (SSEPs) and transcranial electrical motor evoked potentials (tEMPs). Baseline values are obtained immediately after the induction of anesthesia and positioning. This real-time feedback serves as our early warning system. If signal degradation occurs during the caudal pull of the scapula, the surgeon must immediately release the tension and reassess, potentially necessitating a prophylactic clavicular osteotomy to decompress the plexus.
Positioning the patient requires obsessive attention to detail. The patient is placed prone on the operating table. The head is carefully positioned in a neutral, forward-facing alignment, supported by a Mayfield headrest or a specialized gel donut. We strictly avoid excessive cervical rotation, flexion, or extension, which could strain the already anomalous cervical spine or compromise cerebral venous drainage. Longitudinal chest rolls are placed from the clavicles to the iliac crests to elevate the chest, allowing for free diaphragmatic excursion and minimizing intra-abdominal pressure, which in turn reduces epidural venous bleeding. The ipsilateral arm (the affected side) and shoulder girdle must be prepped and draped completely free, allowing for unrestricted manipulation and assessment of shoulder range of motion throughout the operation. All pressure points are meticulously padded.
Step-by-Step Surgical Approach and Fixation Technique
With the patient optimally positioned and neuromonitoring baselines established, we proceed with the surgical approach. The goal of the Modified Woodward procedure is to achieve significant caudal displacement of the scapula, correct its medial rotation, and enhance overall shoulder kinematics, all while meticulously preserving vital neurovascular structures. The operation involves a wide exposure, detachment of the scapular stabilizing muscles from their spinal origins, resection of the omovertebral bone and superomedial scapula, and finally, the caudal reattachment of the musculature.
Incision and Initial Dissection:
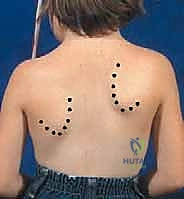
We begin with a generous, straight midline incision extending from the spinous process of C4 down to the spinous process of T9. This extensive length is critical; attempting this procedure through a limited incision severely compromises visualization and the ability to achieve an adequate caudal shift.
This image illustrates the ideal length and position of our incision, extending precisely from C4 to T9 along the spinous processes.
Once the skin is incised, we dissect through the subcutaneous tissue. We must undermine the skin and subcutaneous flaps laterally on the involved side, extending all the way to the medial border of the scapula and the lateral border of the trapezius. This creates large, full-thickness flaps that provide excellent visualization of the entire periscapular region. We then identify the fascial plane between the inferior border of the trapezius and the underlying latissimus dorsi muscle. Using blunt dissection, we separate the lateral border of the trapezius from the latissimus dorsi, establishing our primary surgical interval.
Muscular Detachment and Neurovascular Protection:
Focusing medially, we must detach the trapezius and rhomboid major and minor muscles from their origins on the spinous processes. We start distally at T9 and proceed superiorly. Using electrocautery, we carefully elevate these muscles extraperiosteally, working our way up to the level of C4. In Sprengel deformity, these muscles are frequently fibrotic, and their fascial layers blend abnormally, making identification challenging.
Here you see the wide exposure and the meticulous extraperiosteal detachment of the trapezius and rhomboid muscles from the spinous processes.
During this elevation, protecting the spinal accessory nerve and the dorsal scapular nerve is our highest priority. The spinal accessory nerve courses beneath the trapezius muscle, generally in line with the vertebral border of the scapula. Because of the severe fibrosis often present, visual identification can be treacherous. We rely heavily on our intraoperative neuromonitoring and meticulous, bloodless dissection techniques. We reflect the entire muscle flap laterally, exposing the underlying levator scapulae and the omovertebral structure.
Resection of the Omovertebral Bone and Scapular Osteotomy:
The levator scapulae muscle, originating from the superior medial aspect of the scapula, often overlies or encapsulates the omovertebral structure. We identify this structure—whether it be a fibrous band, a cartilaginous bridge, or a solid osseous connection—linking the scapula to the cervical spine. This entire tether must be excised completely. Using sharp dissection and rongeurs, we resect it extraperiosteally. During this step, the surgeon must be acutely aware of the transverse cervical artery lying deep to the levator scapulae; injury here causes brisk, problematic bleeding.
Once the omovertebral tether is removed, we divide the levator scapulae at its insertion. We then address the scapula itself. To remove the mechanical block to rotation and improve contour, we perform an extraperiosteal resection of the superomedial angle of the scapula. Using an oscillating saw or a large bone biter, we resect the bone along a line connecting the suprascapular notch to the base of the scapular spine. We then lift the scapula away from the chest wall, using a Cobb elevator to bluntly break any deep fibrotic adhesions in the subscapular space, ensuring the scapula is completely mobilized and free-floating.
The Woodward Maneuver and Fixation:
With the scapula completely mobilized, we perform the definitive Woodward maneuver. The assistant grasps the affected arm and the scapula, pulling it forcefully but steadily in a caudal and laterally rotated direction. The goal is to bring the scapular spine to the same horizontal level as the contralateral, normal side. As this downward traction is applied, the anesthesiologist and neurophysiologist must closely monitor the SSEP and MEP signals. If signals drop, traction must be released immediately.
This image demonstrates the critical caudal repositioning of the scapula and the reattachment of the muscular aponeurosis to the spinous processes at a more inferior level.
Once a safe, optimal position is achieved, we reattach the aponeurosis of the trapezius and rhomboids to the spinous processes at a much lower level. We utilize heavy, non-absorbable sutures (e.g., #2 Ethibond or FiberWire) passed through the spinous processes or the robust interspinous ligaments. The excess aponeurosis created by the downward shift is folded over in a "pants-over-vest" fashion to reinforce the repair and prevent dead space. A subfascial drain is placed, and the subcutaneous tissues and skin are closed in layers.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, the Modified Woodward procedure carries a distinct profile of potential complications. The most feared and devastating complication is brachial plexus palsy. This occurs due to traction on the plexus as the scapula is pulled caudally, or due to compression of the plexus between the clavicle and the first rib. The incidence of transient neuropraxia ranges from 2% to 5% in children under 8 years, but rises exponentially in older patients. If neuromonitoring signals are lost intraoperatively and do not recover upon release of traction, a prophylactic midshaft clavicular osteotomy (removing a 1-1.5 cm segment of the clavicle) must be performed immediately to decompress the thoracic outlet.
Scapular winging is another significant complication, occurring in roughly 5% to 10% of cases. This can be secondary to inadvertent injury to the spinal accessory nerve or the dorsal scapular nerve during the muscular dissection. Alternatively, it can result from mechanical failure of the muscle reattachment at the spinous processes. Management of winging depends on the etiology; nerve injuries are typically observed for spontaneous recovery over 6-12 months, while mechanical failures may require revision surgery and reinforcement of the muscular repair using allograft or synthetic mesh.
Cosmetic dissatisfaction and loss of correction are frustrating long-term complications. The long midline incision is highly prone to widening, hypertrophy, and keloid formation, particularly in the tension-rich environment of the back. Furthermore, as the child grows, there can be a relative loss of the initial caudal correction, with the scapula appearing to "migrate" back upwards. This is often due to the inherent growth potential of the hypoplastic bone and recurrent fibrosis. Salvage management for severe loss of correction is exceedingly difficult; revision Woodward procedures carry an exceptionally high risk of nerve injury and are generally discouraged unless functional loss is profound.
| Complication | Estimated Incidence | Prevention and Salvage Management |
|---|---|---|
| Brachial Plexus Palsy | 2% - 5% (Higher in older children) | Prevention: Continuous SSEP/MEP monitoring; limit caudal pull in older patients. Salvage: Immediate release of traction; concurrent clavicular osteotomy if signals do not return. |
| Scapular Winging | 5% - 10% | Prevention: Extraperiosteal dissection; meticulous identification/protection of CN XI. Salvage: EMG to confirm nerve status; revision repair of muscle avulsions if mechanically failed. |
| Keloid / Hypertrophic Scar | 15% - 25% | Prevention: Subcuticular closure; minimal tension on skin edges. Salvage: Silicone sheeting, intralesional corticosteroid injections, dermatologic consultation. |
| Hematoma / Seroma | 3% - 5% | Prevention: Meticulous hemostasis of transverse cervical artery; use of closed suction drains. Salvage: Bedside aspiration or formal surgical evacuation if expanding or infected. |
| Loss of Correction | 10% - 20% (Long-term) | Prevention: Secure "pants-over-vest" fascial closure; adequate release of all deep subscapular tethers. Salvage: Physical therapy to maximize remaining ROM; revision surgery rarely indicated. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following a Woodward procedure is as critical as the surgery itself. The protocol must balance the need for secure tissue healing of the reattached musculature with the imperative to prevent debilitating stiffness. Rehabilitation is divided into four distinct, progressive phases.
Phase I (Weeks 0 to 3) focuses on tissue protection and pain management. The patient is immobilized in a custom sling or a Velpeau bandage, holding the arm in adduction and internal rotation. The primary goal is to protect the muscular reattachment at the spinous processes. Active and passive range of motion of the shoulder is strictly prohibited. However, to prevent distal stiffness, patients are encouraged to perform active range of motion exercises for the elbow, wrist, and hand multiple times a day. Pain is managed with a multimodal analgesic regimen, and the surgical drain is typically removed on postoperative day 1 or 2 when output is minimal.
Phase II (Weeks 3 to 6) marks the initiation of gentle shoulder mobilization. The sling is gradually discontinued, initially worn only at night or in crowded environments. The physical therapist introduces passive range of motion (PROM) and active-assisted range of motion (AAROM) exercises. The focus is primarily on restoring forward elevation and external rotation in the scapular plane. Aggressive stretching or forced manipulation is avoided to prevent avulsion of the healing trapezius and rhomboids. Scapular mobilization techniques are employed to prevent the reformation of dense subscapular adhesions.
Phase III (Weeks 6 to 12) transitions the patient towards active functional use. Active range of motion (AROM) is fully integrated. The emphasis shifts to strengthening the periscapular stabilizers, particularly the serratus anterior and the lower trapezius, to optimize the newly established scapulothoracic rhythm. Light resistance band exercises and isometric strengthening are introduced. The patient is encouraged to begin using the affected arm for light activities of daily living, such as eating and dressing.
Phase IV (3 Months and Beyond) is the return to unrestricted activity. By this stage, the muscular repair is solidly healed. Patients undergo advanced strengthening and proprioceptive training. They are cleared for all recreational activities and sports, though contact sports may be delayed until 6 months postoperatively depending on the surgeon's discretion and the patient's functional recovery. Long-term follow-up is essential, with annual clinical and radiographic evaluations to monitor for any loss of correction or asymmetric growth during the adolescent growth spurt.
Summary of Landmark Literature and Clinical Guidelines
The evolution of surgical treatment for Sprengel deformity is deeply rooted in a few landmark publications that continue to guide our clinical decision
