Forequarter Amputation: An Intraoperative Masterclass in Advanced Oncologic Resection
Key Takeaway
Join us in the OR for a comprehensive masterclass on forequarter amputation. This guide covers meticulous preoperative planning, critical anatomical considerations, and a granular, step-by-step intraoperative execution. Learn precise techniques for managing complex oncologic resections, mitigating neurovascular risks, and ensuring optimal patient outcomes, including advanced flap reconstruction and postoperative care.
Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues and fellows, to a masterclass on one of the most challenging yet profoundly impactful procedures in orthopedic oncology: the Forequarter Amputation, historically referred to as an interscapulothoracic amputation. While the advent of modern neoadjuvant chemotherapy, advanced radiation protocols, and sophisticated limb-sparing reconstructive techniques have fortunately made this operation less common, it remains an absolutely critical tool in our armamentarium. It is reserved for highly select patients facing aggressive, locally advanced, or fungating tumors of the shoulder girdle where limb salvage is anatomically impossible or would definitively compromise oncologic control.
The patho-epidemiology of tumors requiring a forequarter amputation typically involves massive, high-grade sarcomas. Osteosarcoma and Ewing sarcoma of the proximal humerus or scapula that have broken through the cortex to form massive soft tissue extensions encasing the neurovascular bundle are classic indications. Similarly, advanced soft tissue sarcomas—such as undifferentiated pleomorphic sarcoma, synovial sarcoma, or malignant peripheral nerve sheath tumors (MPNST) originating in the axilla or brachial plexus—frequently dictate this radical approach. These tumors often exhibit a relentless local growth pattern, invading the intricate neurovascular and muscular networks of the thoracic outlet and axillary gateway.
Beyond primary sarcomas, we must also consider extensive locoregional recurrences and radiation-induced sarcomas. Patients who have previously undergone limb-sparing resections followed by adjuvant radiation may present years later with a secondary malignancy or a massive recurrence in a heavily scarred, irradiated bed. In these scenarios, the distorted anatomy, compromised vascularity, and aggressive tumor biology make further limb salvage an exercise in futility, predisposing the patient to positive margins, catastrophic flap failure, and ultimately, systemic dissemination. Here, the forequarter amputation offers the most reliable pathway to achieving R0 margins and local disease control.
The psychological impact of this procedure cannot be overstated, necessitating a robust, multidisciplinary approach. The sudden and complete loss of an upper extremity, along with the shoulder contour, induces profound body image alterations and functional deficits. As orthopedic oncologists, our duty extends beyond the operating theater; we must coordinate with psychiatrists, pain management specialists, and prosthetists well before the first incision. Whether the surgical intent is curative for a localized massive tumor or strictly palliative to manage a fungating, bleeding, and intractable lesion, aligning the patient's expectations with the surgical reality is the cornerstone of ethical oncologic practice.
Detailed Surgical Anatomy and Biomechanics
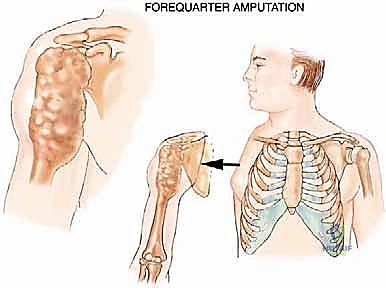
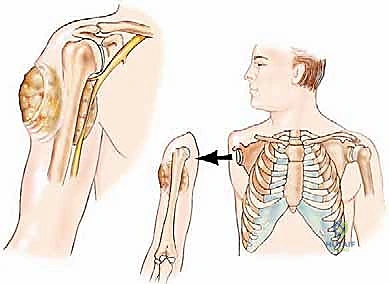
Understanding the intricate surgical anatomy of the shoulder girdle and its relationship to the chest wall is paramount for the safe execution of a forequarter amputation. The upper extremity is a highly mobile appendage suspended from the axial skeleton by a complex network of muscular slings and a single, critical bony articulation: the sternoclavicular joint. The clavicle acts as the sole osseous strut holding the scapula and arm away from the thorax. To achieve an en bloc resection, every single fascial, muscular, and neurovascular connection bridging the appendicular to the axial skeleton must be meticulously identified and transected.
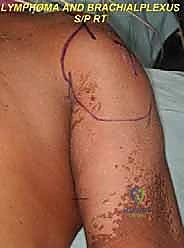
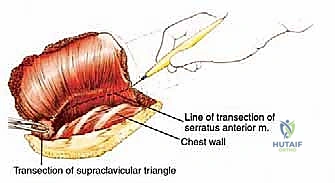
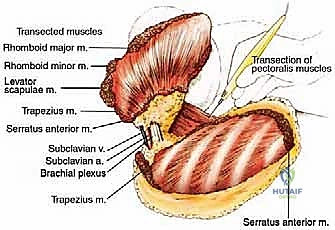
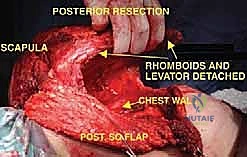
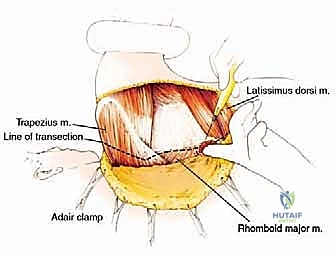
The muscular anatomy can be conceptualized into anterior, posterior, and medial suspensory groups. Posteriorly and medially, the trapezius (originating from the occiput to T12 and inserting on the lateral clavicle, acromion, and scapular spine) forms the superficial layer. Deep to this, the levator scapulae and the rhomboid major and minor tether the medial border of the scapula to the cervical and thoracic spine. The latissimus dorsi, a massive sheet of muscle, must be transected low on the back to release the inferior angle of the scapula and the humerus. Finally, the serratus anterior, which anchors the medial scapular border to the anterolateral ribs (1-8), must be divided close to its costal origins, taking care not to breach the parietal pleura.
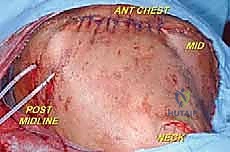
Anteriorly, the musculature forms the boundaries of the axilla and protects the vital neurovascular structures. The pectoralis major (clavicular and sternocostal heads) and the underlying pectoralis minor (inserting onto the coracoid process) must be divided to expose the axillary contents. The subclavius muscle, lying deep to the clavicle, must also be released during the clavicular osteotomy. Understanding the fascial planes here—specifically the clavipectoral fascia—is vital, as tumors originating in the axilla can track along these planes toward the thoracic inlet, directly influencing our margin planning and the necessary level of muscular transection.
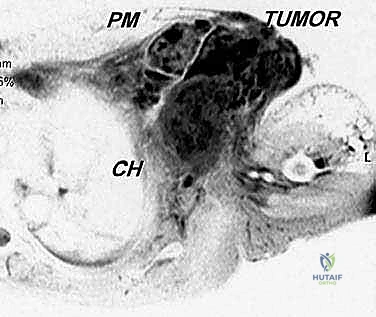
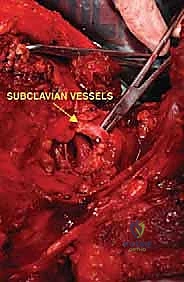
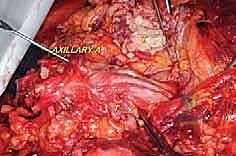
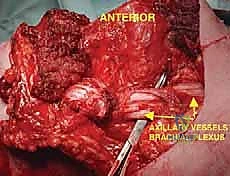
The neurovascular anatomy—what we term the "Axillary Gateway"—represents the most hazardous portion of the dissection. The subclavian artery transitions into the axillary artery at the lateral border of the first rib, passing posterior to the scalenus anterior muscle. The brachial plexus (C5-T1) emerges between the anterior and middle scalene muscles, with its trunks, divisions, and cords intimately wrapping around the axillary artery. The axillary vein lies anterior and slightly inferior to the artery. During a forequarter amputation, proximal control of these subclavian/axillary vessels is the absolute highest priority. Retraction of a transected, unligated subclavian artery beneath the clavicle and first rib into the thoracic cavity is a catastrophic, rapidly fatal event known as "subclavian escape."
Exhaustive Indications and Contraindications
This is not a procedure we undertake lightly; it is a mutilating, life-altering amputation. The decision to proceed with a forequarter amputation requires a rigorous tumor board discussion, balancing the oncologic necessity of achieving negative margins against the profound functional and psychological costs. Our primary goals are either curative en bloc resection of a massive malignancy or palliation of intractable pain, hemorrhage, and sepsis caused by a fungating tumor unresponsive to systemic therapies.
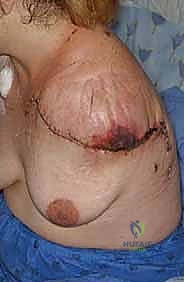
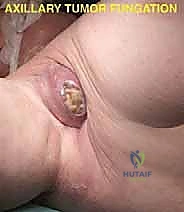
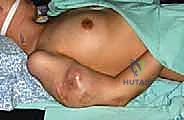
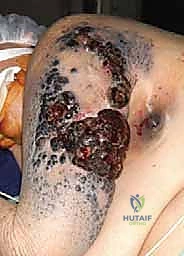
Specific clinical indications include extensive soft tissue sarcomas of the proximal arm or axilla that demonstrably encase the brachial plexus and axillary vessels. When a tumor spans the shoulder joint, involves the scapula, and obliterates the neurovascular bundle, limb salvage is anatomically impossible without leaving gross residual disease. Similarly, advanced primary bone sarcomas (like massive osteosarcomas or chondrosarcomas of the proximal humerus) that have escaped the osseous compartment to invade the axilla and chest wall musculature demand this radical approach. Furthermore, patients presenting with severe tumor fungation—where the mass has ulcerated through the skin, leading to chronic infection, malodor, and intractable neurogenic pain—are prime candidates for palliative forequarter amputation to restore dignity and quality of life.

Contraindications must be carefully evaluated to prevent performing a massive surgery that yields no oncologic or palliative benefit. Absolute contraindications include extensive tumor invasion into the deep paraspinal musculature, the epidural space, or massive involvement of the intrathoracic cavity that precludes a negative margin even with combined chest wall resection. Furthermore, patients with severe cardiopulmonary comorbidities who cannot tolerate the immense physiologic stress, blood loss, and fluid shifts associated with this surgery are not candidates. Disseminated metastatic disease is a relative contraindication; however, in cases of severe fungation and hemorrhage, a palliative amputation may still be justified to manage local symptoms, provided the patient's life expectancy warrants the recovery period.
The "gray zone" of indications involves tumors that abut the chest wall or extend high into the posterior triangle of the neck. In these highly selected cases, a forequarter amputation can be combined with a radical neck dissection or an en bloc chest wall resection (ribs 1-3). This requires a coordinated effort with thoracic and head-and-neck surgeons. While morbidity is significantly increased, it offers a potential chance for cure in otherwise unresectable locoregional disease.
| Category | Specific Criteria | Clinical Rationale |
|---|---|---|
| Absolute Indications | Massive soft tissue sarcoma encasing the axillary neurovascular bundle. | Limb salvage impossible; high risk of systemic spread if margins are compromised. |
| Absolute Indications | Advanced bone sarcoma of proximal humerus/scapula with massive extraosseous extension. | En bloc resection required to achieve R0 margins; joint contamination precludes salvage. |
| Palliative Indications | Fungating, bleeding, or infected tumor causing intractable pain. | Improves quality of life, removes source of sepsis/hemorrhage, restores patient dignity. |
| Relative Contraindications | Widespread metastatic disease (unless palliative for severe local symptoms). | Systemic disease dictates prognosis; major surgery may delay palliative chemotherapy. |
| Absolute Contraindications | Unresectable extension into the epidural space, spinal cord, or massive intrathoracic invasion. | Surgery will be intralesional; unacceptable morbidity without oncologic benefit. |
| Absolute Contraindications | Severe cardiopulmonary failure or inability to tolerate massive fluid shifts. | High risk of intraoperative mortality; physiologic reserve insufficient for recovery. |
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning is the absolute cornerstone of a successful forequarter amputation. The goal is to eliminate intraoperative surprises, meticulously map the tumor's extent, and design soft tissue flaps that will provide adequate, tension-free coverage of the massive chest wall defect while strictly adhering to oncologic margins. We rely heavily on a combination of advanced imaging modalities to achieve this three-dimensional understanding.
Magnetic Resonance Imaging (MRI) with intravenous contrast is the gold standard for evaluating soft tissue extension, neurovascular encasement, and intramedullary bone involvement. We carefully assess the relationship of the tumor to the brachial plexus, the scalene muscles, and the chest wall. Computed Tomography (CT) of the chest is mandatory, not only for pulmonary staging but also to evaluate cortical bone destruction of the scapula and ribs, and to identify any direct plural or pulmonary invasion. In complex cases, 3D reconstructions can be invaluable for templating the exact level of the clavicular osteotomy and anticipating chest wall resections.
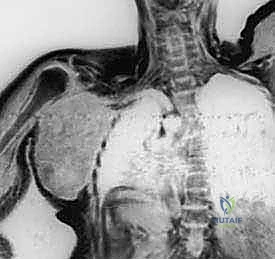
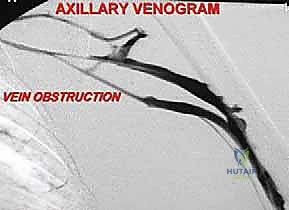
Vascular imaging is critical. CT angiography or conventional angiography provides a precise roadmap of the subclavian and axillary vessels. It helps identify anatomical variants (such as a high-riding subclavian artery or anomalous branches) and confirms the patency or occlusion of the vessels by the tumor. Venography, or increasingly MR venography, is highly sensitive for assessing venous thrombosis caused by tumor compression—a reliable surrogate marker for encasement of the adjacent brachial plexus. This vascular mapping dictates whether we can safely ligate the vessels below the clavicle or if we must perform a higher, supraclavicular dissection.
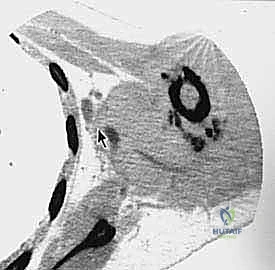
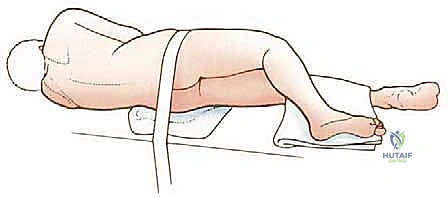
Patient positioning and anesthesia preparation require meticulous attention. The patient is typically placed in a lateral decubitus or a "floppy lateral" position, stabilized with a beanbag, allowing access to both the anterior chest and the posterior scapular region. The entire forequarter, neck, hemithorax, and back must be prepped and draped free to allow manipulation of the limb. Anesthesia must be prepared for rapid, massive blood loss. Large-bore peripheral IVs, a central venous line (placed on the contralateral side), and an arterial line are mandatory. We often employ controlled hypotensive anesthesia during the initial dissection to minimize blood loss, normalizing pressure only after the major vessels are secured and before final hemostasis.
Step-by-Step Surgical Approach and Flap Fixation Technique
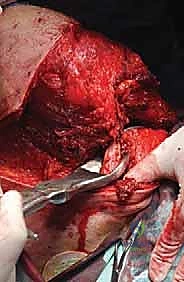
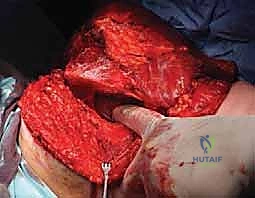
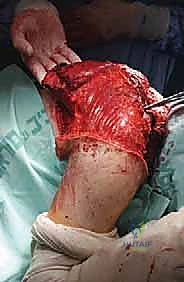
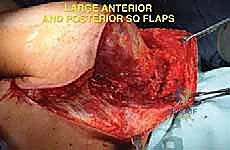
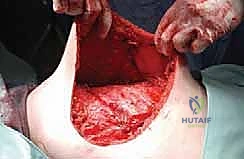
The surgical execution of a forequarter amputation is a highly choreographed sequence that demands anatomical precision and oncologic discipline. The procedure is generally divided into anterior dissection for neurovascular control, posterior dissection for muscular release, and final specimen delivery followed by complex wound closure.
Incision and Anterior Dissection
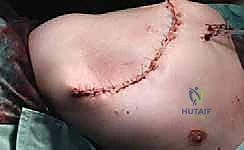
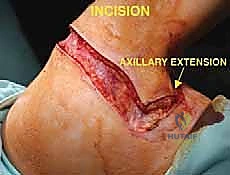
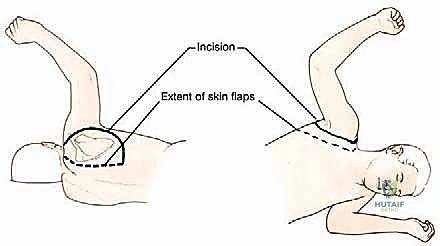
The classic incision is racquet-shaped, though it must be highly customized based on the tumor's location to ensure wide margins. The anterior limb typically begins at the medial third of the clavicle, extending laterally along the deltopectoral groove, and sweeping inferiorly across the axilla to join the posterior limb. The posterior limb drops from the lateral clavicle, following the vertebral border of the scapula down to its inferior angle, before curving anteriorly to meet the axillary incision.
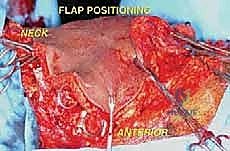
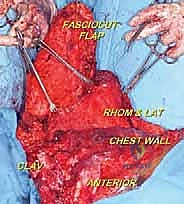
The first major structural step is the clavicular osteotomy. The periosteum of the medial third of the clavicle is incised and carefully elevated. A Gigli saw or an oscillating saw is used to divide the clavicle. The subclavius muscle is then sharply divided. This maneuver allows the shoulder girdle to fall laterally, opening the "book" of the thoracic inlet and providing direct, unhindered access to the subclavian vessels and brachial plexus.
Following the osteotomy, the pectoralis major is transected near its clavicular and sternal origins, depending on the required margin. The clavipectoral fascia is incised, exposing the pectoralis minor, which is similarly divided. At this stage, the axillary sheath is fully exposed. Careful blunt and sharp dissection is utilized to isolate the neurovascular bundle, ensuring no tumor capsule is breached during this critical exposure phase.
Neurovascular Ligation and Control
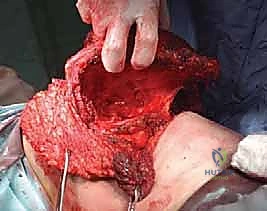
Vascular control is the most critical and potentially hazardous phase of the operation. There is historical debate regarding whether to ligate the artery or the vein first. Ligation of the artery first minimizes blood loss and reduces the risk of tumor engorgement. However, in highly friable or massive tumors, early ligation of the vein may be preferred to prevent systemic tumor embolization during manipulation. In practice, we isolate both, ligate the artery first to drain the limb of blood, and immediately follow with the vein.
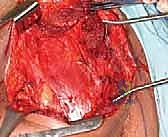
The subclavian/axillary artery and vein are individually skeletonized. We utilize heavy, non-absorbable sutures (e.g., #1 Silk) for double ligation, supplemented by a distal transfixion suture (stick tie) on the proximal stump to definitively prevent slippage. The vessels are then divided. Extreme care must be taken to ensure the proximal stumps are long enough to prevent retraction beneath the first rib, which would necessitate an emergent thoracotomy to control bleeding.
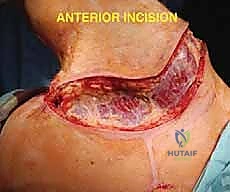
Next, the brachial plexus is addressed. The nerve roots or trunks are isolated as proximally as oncologically necessary. To minimize the risk of devastating postoperative neuroma formation, the nerves are placed under gentle distal traction, sharply transected with a fresh scalpel blade, and allowed to retract deep into the interscalene musculature. We often inject the nerve stumps with a long-acting local anesthetic to aid in immediate postoperative pain control. Additional collateral vessels, such as the transverse cervical and suprascapular arteries, are identified and ligated at this stage
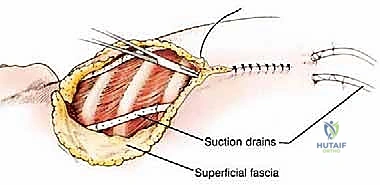
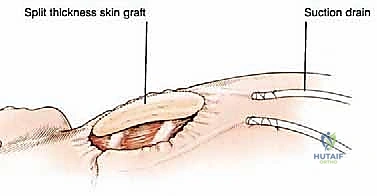
Clinical & Radiographic Imaging Archive