Ellis–Van Creveld’s Syndrome: Orthopedic Board Review

Comprehensive Introduction and Patho-Epidemiology
Genetic Basis and Molecular Pathophysiology
Ellis-van Creveld (EVC) syndrome, also known in the medical literature as chondroectodermal dysplasia, is a rare, autosomal recessive skeletal dysplasia characterized by a highly specific tetrad of clinical manifestations: disproportionate short stature, postaxial polydactyly, ectodermal dysplasia, and congenital cardiac defects. The genetic basis of this condition has been definitively mapped to mutations in the EVC1 and EVC2 genes, which are located in a head-to-head configuration on chromosome 4p16. These genes encode transmembrane proteins that localize to the basal body of the primary cilium, an organelle critical for cellular mechanotransduction and signal transduction in chondrocytes.
The primary cilium acts as the central hub for the Hedgehog signaling pathway, particularly the Sonic Hedgehog (Shh) and Indian Hedgehog (Ihh) cascades. In the developing growth plate, Ihh coordinates with parathyroid hormone-related peptide (PTHrP) in a feedback loop that dictates chondrocyte proliferation and differentiation. Mutations in EVC1 or EVC2 disrupt this critical signaling axis, leading to premature cessation of chondrocyte proliferation, accelerated hypertrophic differentiation, and ultimately, defective endochondral ossification. This molecular failure manifests macroscopically as the severe mesomelic and acromelic shortening characteristic of the syndrome.
Furthermore, the disruption of Shh signaling during early embryogenesis explains the pleiotropic nature of the syndrome. Shh is essential for establishing the zone of polarizing activity (ZPA) in the developing limb bud, which dictates the radioulnar (anterior-posterior) axis of the limb. Aberrant Shh signaling leads to the failure of proper digit segmentation and the development of postaxial polydactyly. Similarly, the ectodermal and cardiac anomalies stem from defective ciliary signaling in neural crest cells and embryonic mesoderm during the critical weeks of organogenesis.
Epidemiological Profile and Clinical Tetrad
While Ellis-van Creveld syndrome is exceedingly rare in the general population, with an estimated prevalence of 1 in 60,000 to 1 in 200,000 live births, it exhibits a striking founder effect within certain isolated genetic pools. The condition is most famously prevalent among the Old Order Amish population of Lancaster County, Pennsylvania, where the incidence is estimated to be as high as 1 in 5,000 births, with a carrier frequency approaching 1 in 14. This high prevalence is attributed to a single founder mutation introduced in the 18th century, combined with strict endogamy within the community.
The classic clinical tetrad is the hallmark of EVC. The chondrodysplasia presents as disproportionate dwarfism, predominantly affecting the distal segments of the limbs (mesomelic and acromelic shortening), while the axial skeleton remains relatively spared, aside from a narrow, bell-shaped thorax. Ectodermal manifestations include hypoplastic, dystrophic nails, sparse and brittle hair, and characteristic oral findings such as multiple frenula, malocclusion, and natal teeth. Postaxial polydactyly is nearly universally present in the hands and occasionally in the feet.

Congenital heart disease is present in approximately 50% to 60% of patients and is the primary determinant of life expectancy. As frequently tested on orthopedic board examinations, while a single atrium or atrial septal defect (ASD) is classically described, ventricular septal defects (VSD) are incredibly common and represent a critical source of morbidity. The presence of these cardiac anomalies necessitates rigorous preoperative cardiovascular optimization before any orthopedic intervention.
Systemic Manifestations and Orthopedic Relevance
Beyond the classic tetrad, EVC presents with a myriad of systemic challenges that profoundly influence orthopedic management. The narrow, bell-shaped thorax, caused by shortened ribs, frequently leads to restrictive lung disease and severe respiratory distress in the neonatal period. Up to 50% of mortalities in EVC occur in infancy due to cardiorespiratory failure. For the orthopedic surgeon, this translates to significant anesthetic risks, requiring meticulous preoperative pulmonary function testing and intraoperative ventilatory management.
Renal anomalies, including agenesis or dysplasia, and genitourinary malformations such as epispadias or cryptorchidism, are also occasionally observed. The orthopedic surgeon must maintain a high index of suspicion for these concomitant anomalies, as impaired renal function can alter the pharmacokinetics of perioperative medications, including antibiotics and analgesics, and complicate fluid management during major reconstructive procedures.
From a purely musculoskeletal perspective, the systemic nature of the collagen and chondrocyte dysfunction means that bone quality may be compromised, and healing times following osteotomies can be unpredictable. The combination of ligamentous laxity—particularly around the knee—and altered joint morphometry creates a challenging biomechanical environment. Surgical interventions must therefore be meticulously planned, recognizing that the underlying genetic defect continues to influence skeletal development and soft tissue integrity throughout the patient's growth trajectory.
Detailed Surgical Anatomy and Biomechanics
Upper Extremity and Hand Anomalies
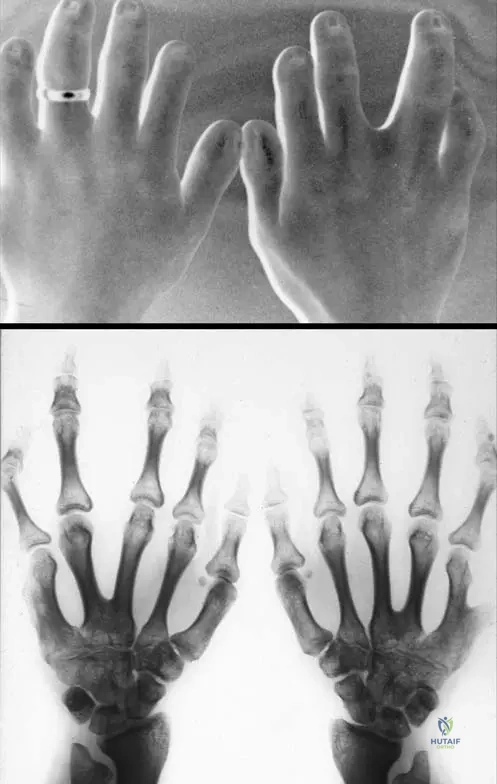
The upper extremity in Ellis-van Creveld syndrome is characterized by profound acromelic and mesomelic shortening, with the hand bearing the most complex and pathognomonic deformities. Postaxial polydactyly is present in nearly 100% of cases, typically manifesting as a fully formed, articulated sixth digit on the ulnar aspect of the hand (Type A polydactyly), though rudimentary soft tissue tags (Type B) can also occur. The supernumerary digit often shares an articulation with a widened or bifurcated fifth metacarpal head, complicating surgical excision.
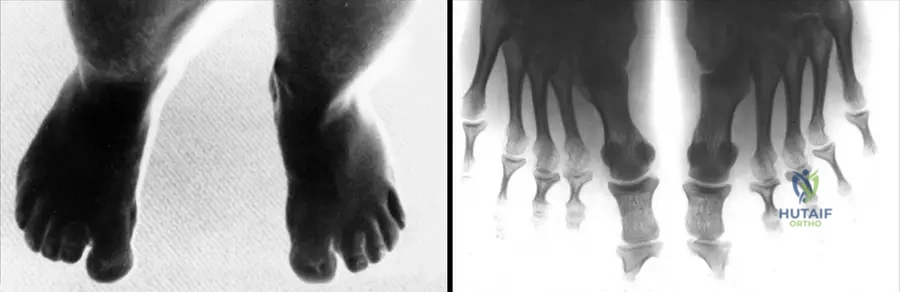
At the carpal level, EVC presents with highly specific synostoses. The fusion of the capitate and hamate is considered pathognomonic for the syndrome and is frequently accompanied by an extra carpal bone in the proximal row. This massive, fused carpal block fundamentally alters the kinematics of the wrist, limiting ulnar deviation and extension. Furthermore, the metacarpals and phalanges exhibit characteristic cone-shaped epiphyses, which lead to premature physeal closure and progressive brachydactyly.
The muscular anatomy of the hypothenar eminence is invariably anomalous. The abductor digiti minimi (ADM) and flexor digiti minimi often insert into the supernumerary digit. During surgical ablation of the extra digit, the orthopedic surgeon must meticulously identify these tendinous insertions and transfer them to the base of the proximal phalanx of the retained fifth digit. Failure to reconstruct the ulnar collateral ligament and transfer the intrinsic musculature will result in a functionally impaired, abducted, and weak little finger.
Lower Extremity and Knee Pathoanatomy
The lower extremity deformities in EVC are dominated by severe, progressive genu valgum, which is driven by highly specific pathoanatomy at the proximal tibia. Unlike idiopathic genu valgum, the deformity in EVC is characterized by a profound hypoplasia of the lateral tibial plateau. This creates a primary osseous depression that mimics a "reverse Blount's disease." The lateral femoral condyle may also be hypoplastic, exacerbating the valgus alignment.
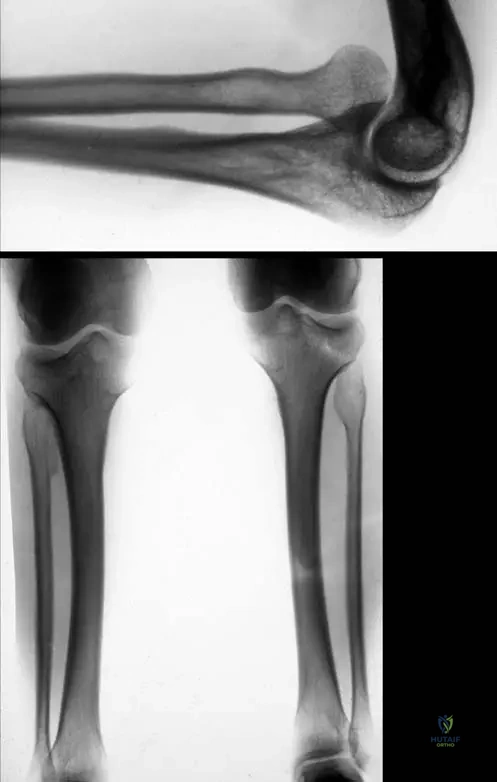
Biomechanically, this morphologic defect shifts the mechanical axis of the lower extremity far laterally, passing through or even lateral to the lateral compartment of the knee. This eccentric loading creates a vicious cycle of Hueter-Volkmann growth suppression: the increased compressive forces on the lateral physis further inhibit its growth, while the relative unloading of the medial physis accelerates medial growth, exponentially worsening the valgus deformity.
Soft tissue contractures inevitably follow the osseous deformity. The iliotibial band, lateral collateral ligament, and biceps femoris become adaptively shortened, while the medial collateral ligament becomes attenuated and incompetent. This leads to profound mediolateral instability. Furthermore, the valgus alignment drastically alters the Q-angle, leading to obligate lateral patellar tracking, recurrent patellar subluxation, and early-onset patellofemoral chondromalacia. Surgical correction must address both the osseous malalignment and the secondary soft tissue imbalances.
Thoracic and Axial Skeletal Considerations
While the appendicular skeleton bears the brunt of the dysplastic process, the axial skeleton, particularly the thorax, presents critical anatomical challenges. The ribs in EVC are characteristically short, horizontally oriented, and lack the normal downward slope. This creates a narrow, bell-shaped thoracic cage with severely restricted volume. The costochondral junctions may be irregular and bulbous, further limiting chest wall compliance.
Biomechanically, this thoracic dystrophy restricts the excursion of the diaphragm and limits the expansion of the pulmonary parenchyma. In the neonatal period, this manifests as life-threatening asphyxiating thoracic dystrophy, requiring prolonged mechanical ventilation. Even in patients who survive infancy, the restrictive lung disease persists, leading to chronic hypoventilation, cor pulmonale, and an elevated risk of perioperative respiratory failure during orthopedic procedures.
The spine itself is relatively spared compared to other chondrodysplasias like achondroplasia. However, exaggerated lumbar lordosis is common, often secondary to hip flexion contractures or as a compensatory mechanism for the altered center of gravity caused by the severe lower extremity malalignment. Spinal stenosis is not a classic feature of EVC, but routine clinical evaluation of the neuraxis remains a standard component of the comprehensive orthopedic assessment.
Exhaustive Indications and Contraindications
Criteria for Surgical Intervention
Surgical intervention in Ellis-van Creveld syndrome is primarily aimed at maximizing function, preventing early joint degeneration, and improving the patient's quality of life. In the upper extremity, the primary indication for surgery is the presence of postaxial polydactyly that interferes with normal hand function or presents a significant psychosocial burden. Excision and reconstruction are typically performed between 12 and 18 months of age, coinciding with the development of fine motor skills and pincer grasp.
In the lower extremity, surgical indications are dictated by the severity and progression of the genu valgum. Mild deformities may be observed, but progressive valgus that shifts the mechanical axis into the lateral compartment (Zone 2 or 3) is an absolute indication for intervention. In the growing child with open physes, guided growth (hemiepiphysiodesis) is indicated to harness remaining growth for gradual correction. In skeletally mature patients, or those with severe, rigid deformities and lateral plateau depression that cannot be corrected with guided growth alone, corrective osteotomies (high tibial or distal femoral) are indicated.
Patellofemoral instability, a frequent concomitant of the valgus deformity, is an indication for simultaneous extensor mechanism realignment. If the patella remains subluxated after the mechanical axis is restored to neutral, procedures such as medial patellofemoral ligament (MPFL) reconstruction or tibial tubercle osteotomy may be required to prevent rapid chondral wear and debilitating anterior knee pain.
Absolute and Relative Contraindications
The most critical contraindications to orthopedic surgery in EVC patients stem from their systemic manifestations. Unoptimized congenital heart disease—particularly severe VSD, ASD, or common atrium with evidence of pulmonary hypertension—is an absolute contraindication to elective orthopedic surgery. Cardiac status must be thoroughly evaluated by a pediatric cardiologist, and any necessary structural repairs should precede elective musculoskeletal interventions.
Severe, uncompensated restrictive lung disease secondary to thoracic dystrophy is another absolute contraindication. Patients with marginal respiratory reserve may not tolerate the stress of general anesthesia, prolonged supine positioning, or postoperative narcotic analgesia, risking fatal respiratory failure. Comprehensive pulmonary function testing and clearance by a pediatric pulmonologist are mandatory.
Relative contraindications include active local or systemic infection, severe osteopenia that would preclude adequate hardware fixation, and profound developmental delay that would prevent compliance with postoperative weight-bearing restrictions and rehabilitation protocols. In cases of extremely severe lateral tibial plateau hypoplasia, isolated guided growth may be relatively contraindicated, as it cannot restore the articular congruity; an intra-articular elevation osteotomy may be required instead.
Indications and Contraindications Summary
| Category | Indications for Surgery | Absolute Contraindications | Relative Contraindications |
|---|---|---|---|
| Upper Extremity | Type A/B postaxial polydactyly; functional impairment of grasp; severe cosmetic stigma (surgery at 12-18 mos). | Unoptimized cardiac defects (VSD/ASD); severe pulmonary hypertension. | Lack of rudimentary bone in supernumerary digit (alters approach); active local infection. |
| Lower Extremity (Growing) | Progressive genu valgum (Mechanical Axis in Zone 2/3); open physes; lateral patellar tracking. | Closed physes (for guided growth); uncorrected cardiorespiratory failure. | Severe intra-articular lateral plateau depression requiring structural elevation. |
| Lower Extremity (Mature) | Rigid genu valgum; mechanical axis deviation; early lateral compartment arthrosis. | Active joint infection; medically unstable for major anesthesia. | Severe osteopenia precluding fixation; inability to comply with rehab. |
Pre-Operative Planning, Templating, and Patient Positioning
Multidisciplinary Assessment and Cardiac Clearance
The preoperative evaluation of a patient with Ellis-van Creveld syndrome is a masterclass in multidisciplinary medical management. The orthopedic surgeon must act as the coordinator of a team that includes pediatric cardiology, pulmonology, genetics, and anesthesiology. A recent, comprehensive echocardiogram is mandatory to evaluate the status of the atrial and ventricular septa. If a VSD or ASD is present, the direction of the shunt and the presence of pulmonary hypertension must be documented.
Pulmonary assessment is equally critical. Chest radiographs are reviewed to assess the severity of the thoracic dystrophy. In older children, formal pulmonary function tests (PFTs) are obtained to quantify the degree of restrictive lung disease. The anesthesiology team must be involved early to plan for potential difficulties with intubation, the need for advanced airway adjuncts, and the strategy for postoperative pain management that minimizes respiratory depression, such as regional nerve blocks or epidural analgesia.
Genetic counseling should be offered to the family if not previously completed. Understanding the exact genetic mutation can sometimes provide prognostic information regarding the severity of the skeletal dysplasia. Furthermore, routine preoperative laboratory investigations must include a comprehensive metabolic panel to assess renal function, given the known association of EVC with renal dysplasia.
Radiographic Templating and Deformity Analysis
Meticulous radiographic templating is the cornerstone of successful deformity correction in EVC. For the upper extremity, standard PA and lateral radiographs of the hand and wrist are required to define the anatomy of the supernumerary digit, the presence of carpal synostoses (capitate-hamate), and the morphology of the metacarpals. This dictates the level of amputation and the need for collateral ligament reconstruction.
For the lower extremity, full-length, standing, weight-bearing anteroposterior (AP) to radiograph (long leg alignment films) are mandatory. The surgeon must calculate the Mechanical Axis Deviation (MAD), the mechanical Lateral Distal Femoral Angle (mLDFA), and the Medial Proximal Tibial Angle (MPTA). In EVC, the MPTA is typically significantly increased due to the lateral plateau hypoplasia, while the mLDFA may also be abnormal.
If an osteotomy is planned, the center of rotation of angulation (CORA) must be identified. In EVC, the CORA is often located at the joint line due to the articular depression, necessitating careful consideration of whether a metaphyseal osteotomy will induce an unacceptable secondary translation. Advanced imaging, such as a CT scan with 3D reconstruction, may be indicated to fully appreciate the complex 3D morphology of the hypoplastic lateral tibial plateau and to plan for potential intra-articular elevation procedures.
Anesthesia Considerations and Patient Positioning
Patient positioning and intraoperative setup require meticulous attention to detail. For upper extremity procedures, the patient is positioned supine with the arm extended on a radiolucent hand table. A well-padded pediatric tourniquet is applied to the proximal arm. Given the small size of the patients and the complex vascular anatomy, loupe magnification or an operating microscope should be available for the dissection of the neurovascular bundles during polydactyly reconstruction.
For lower extremity deformity correction, the patient is positioned supine on a completely radiolucent flat Jackson or OSI table to allow for unencumbered fluoroscopic imaging from the hip to the ankle. A bump is placed under the ipsilateral hip to control external rotation. Bilateral procedures are common, and the sterile field must encompass both legs up to the iliac crests to allow for intraoperative assessment of the mechanical axis using a cautery cord or alignment rod under fluoroscopy.
Anesthetic management must prioritize respiratory drive and hemodynamic stability. Due to the high prevalence of cardiac shunts, strict precautions against intravenous air bubbles must be maintained to prevent paradoxical emboli. Regional anesthesia, such as a supraclavicular block for the upper extremity or a combined spinal-epidural for the lower extremities, is highly favored to minimize the need for systemic opioids and to avoid the respiratory depression that can be catastrophic in patients with underlying thoracic dystrophy.
Step-by-Step Surgical Approach and Fixation Technique
Management of Postaxial Polydactyly
Surgical ablation of postaxial polydactyly in EVC is not a simple amputation; it is a complex reconstruction of the ulnar border of the hand. An elliptical or racquet-shaped incision is designed at the base of the supernumerary digit, ensuring adequate skin flaps for closure without tension. The dissection proceeds carefully to identify and isolate the neurovascular bundles supplying the extra digit. These are ligated and divided as proximally as possible to prevent painful neuroma formation.
The critical step involves the management of the hypothenar musculature. The abductor digiti minimi (ADM) frequently inserts aberrantly into the supernumerary digit. This tendon must be carefully detached, preserving its length and neurovascular pedicle. The supernumerary digit is then disarticulated at the metacarpophalangeal joint. If the fifth metacarpal head is bifurcated, a longitudinal osteotomy is performed to narrow the metacarpal neck and create a congruent articular surface for the remaining proximal phalanx.
Following the skeletal resection, the ulnar collateral ligament (UCL) of the retained fifth digit must be reconstructed to prevent radial deviation and instability during pinch. The capsule and periosteum from the resected digit are utilized to reinforce the UCL. Finally, the ADM tendon is transferred and sutured to the base of the proximal phalanx of the fifth digit and the extensor hood, restoring active abduction and stabilizing the ulnar border of the hand. The skin is closed with absorbable sutures, and a bulky, protective cast is applied.
Guided Growth for Genu Valgum
In the skeletally immature EVC patient with progressive genu valgum, hemiepiphysiodesis via tension band plating (guided growth) is the procedure of choice. Under fluoroscopic guidance, the medial physis of the distal femur, proximal tibia, or both (depending on the preoperative templating of the mLDFA and MPTA) is identified. A 2-3 cm longitudinal incision is made over the medial aspect of the target physis.
The subcutaneous tissues are bluntly dissected to expose the periosteum. It is critical to avoid violating the periosteum or the perichondrial ring, as this can lead to permanent physeal arrest. A guide pin is placed exactly at the level of the physis, and a two-hole extra-periosteal plate (e.g., an 8-plate) is positioned over the pin. The plate must be centered perfectly over the physis in both the AP and lateral planes to prevent the induction of a secondary sagittal plane deformity (genu recurvatum or procurvatum).
Cannulated screws are then inserted into the metaphysis and epiphysis. The screws should be parallel to the joint line and diverge slightly from each other to allow for maximum angular correction as the bone grows. Fluoroscopy is used to confirm that the screws do not violate the physis or the articular surface. The wound is closed in layers. This minimally invasive technique harnesses the patient's remaining growth to gradually correct the mechanical axis, requiring close radiographic follow-up every 3-4 months.
Corrective Osteotomy for Severe Lower Extremity Deformity
In skeletally mature patients, or those with severe, rigid deformities not amenable to guided growth, corrective osteotomies are required. For severe valgus driven by the tibia, a medial opening wedge high tibial osteotomy (HTO) or a lateral closing wedge HTO is performed. The lateral closing wedge is often preferred in EVC to avoid stretching the already compromised common peroneal nerve and to address the lateral tissue contractures.
An anterolateral incision is made, and the anterior compartment musculature is elevated. A fibular osteotomy is performed first at the junction of the middle and distal thirds to allow for tibial mobilization. Guide pins are placed under fluoroscopic guidance to define the wedge of bone to be resected from the proximal tibia, ensuring the hinge point remains intact on the medial cortex. The osteotomy is completed with an oscillating saw and osteotomes, and the wedge is removed.
The deformity is slowly corrected by closing the wedge, which shifts the mechanical axis medially. Rigid internal fixation is achieved using a pre-contoured locking plate. In cases where the lateral tibial plateau is severely hypoplastic, a complex intra-articular osteotomy (e.g., a hemi-plateau elevation) may be necessary. This involves elevating the depressed lateral plateau and supporting it with structural allograft or autograft, followed by rigid fixation with subchondral raft screws. The mechanical axis is verified intraoperatively with an alignment rod before final closure.
Complications, Incidence Rates, and Salvage Management
Intraoperative and Early Postoperative Complications
Orthopedic interventions in Ellis-van Creveld syndrome carry a unique risk profile due to the underlying genetic and systemic anomalies. Intraoperatively, the most feared complication is a cardiorespiratory event secondary to the patient's underlying VSD/ASD or thoracic dystrophy. Hypoxia, hypercarbia, or fluid shifts can precipitate pulmonary hypertensive crises. Continuous invasive hemodynamic monitoring and close communication with the anesthesia team are paramount.
Surgically, neurovascular injury is a significant risk during deformity correction. The common peroneal nerve is particularly vulnerable during the correction of severe valgus deformities, either from direct traction during an opening wedge medial osteotomy or from direct injury during a lateral closing wedge procedure. The incidence of transient peroneal nerve palsy in major valgus correction can approach 5-10%. Prophylactic peroneal nerve decompression at the fibular head should be considered in severe cases.
Early postoperative complications include wound dehiscence and infection. The compromised soft tissue envelope, particularly over the medial proximal tibia, is susceptible to breakdown. Meticulous surgical technique, layered closure without tension, and the use of incisional negative pressure wound therapy can mitigate this risk. If a deep surgical site infection occurs, aggressive surgical debridement and culture-directed intravenous antibiotics are mandatory to prevent catastrophic hardware infection and osteomyelitis.
Late Deformity Recurrence and Hardware Issues
In the growing child treated with guided growth, the most common late complication is the "rebound phenomenon." Because the underlying genetic defect in the EVC genes continues to drive abnormal physeal growth, the valgus deformity frequently recurs once the tension band plates are removed. The recurrence rate in syndromic genu valgum can exceed 50%. Consequently, surgeons often overcorrect the mechanical axis slightly into varus before hardware removal, or opt to leave the hardware in place, monitoring closely for overcorrection.
Delayed union or nonunion is a significant risk following corrective osteotomies. The abnormal collagen architecture and defective endochondral ossification inherent to EVC can impair the normal biology of fracture healing. The incidence of delayed union in syndromic osteotomies is notably higher than in idiopathic cases. Rigid locking plate fixation and the liberal use of osteoinductive bone grafts (e.g., autologous iliac crest, BMP-2) are recommended to stimulate union.
Hardware failure, including screw breakage or plate pullout, can occur if the osteotomy fails to heal or if the patient is non-compliant with weight-bearing restrictions. In the event of a nonunion with hardware failure, salvage management requires revision surgery. This typically involves removal of the broken hardware, aggressive takedown of the nonunion site, re-cutting the bone ends to healthy bleeding tissue, placement of structural bone graft, and revision fixation, often utilizing a multi-planar external fixator (e.g., Ilizarov or Taylor Spatial Frame) to provide absolute stability and allow for fine-tuning of the alignment.
Complications and Salvage Management Summary
| Complication | Estimated Incidence | Etiology/Risk Factors | Salvage Management / Prevention |
|---|---|---|---|
| Cardiorespiratory Crisis | 5-15% (Systemic) | Underlying VSD/ASD; restrictive lung disease; fluid overload. | Pre-op optimization; invasive monitoring; avoid hypoxia/hypercarbia. |
| Peroneal Nerve Palsy | 5-10% | Traction from valgus correction; direct injury during lateral approach. | Prophylactic nerve decompression; limit acute correction; AFO for drop foot. |
| Rebound Deformity | >50% (Guided Growth) | Persistent genetic physeal dysfunction post-hardware removal. | Slight overcorrection into varus prior to removal; serial observation; repeat plating. |
| Nonunion / Delayed Union | 10-20% (Osteotomies) | Defective endochondral ossification; inadequate fixation stability. | Rigid locking plates; use of autograft/BMP; revision with external fixation if failed. |
| Wound Dehiscence | 5-8% | Poor soft tissue envelope; excessive tension on closure. | Tension-free closure; incisional VAC; prompt debridement and flap coverage if deep. |
Phased Post-Operative Rehabilitation Protocols
Immediate Postoperative Phase
