Arthroscopic Double-Row Rotator Cuff Repair: An Intraoperative Masterclass
Key Takeaway
Join us in the OR for an immersive masterclass on arthroscopic double-row rotator cuff repair. We'll meticulously cover comprehensive anatomy, preoperative planning, granular intraoperative steps, critical pearls and pitfalls, and essential postoperative management to ensure optimal patient outcomes and surgical excellence.
Welcome, fellows, to the operating theater. Today, we are performing an arthroscopic double-row rotator cuff repair, a procedure that has truly revolutionized how we manage symptomatic full-thickness rotator cuff tears. This is not merely an exercise in fixing a tendon; it is a meticulous biomechanical reconstruction aimed at restoring function, alleviating pain, and preventing irreversible glenohumeral degeneration. As orthopedic surgeons, our goal is to recreate the native footprint anatomy, maximizing the pressurized contact area between the tendon and the bone bed to optimize biological healing. Let us delve deeply into the profound nuances of this critical shoulder surgery.
Comprehensive Introduction and Patho-Epidemiology
The evolution of rotator cuff repair has transitioned from morbid open procedures involving deltoid detachment to sophisticated, minimally invasive arthroscopic techniques. The advent of the arthroscopic double-row, and specifically the suture-bridge or transosseous-equivalent construct, represents a paradigm shift in our biomechanical approach. Historically, single-row repairs, while clinically successful in many instances, often failed to restore the anatomical footprint, leading to point-loading at the suture-tendon interface and potential gap formation. The double-row technique mitigates this by utilizing a medial row of anchors at the articular margin and a lateral row at the greater tuberosity, effectively compressing the tendon against the decorticated bone bed.
Understanding the patho-epidemiology of rotator cuff disease is essential for appropriate surgical decision-making. Rotator cuff tears are predominantly a disease of aging, with prevalence increasing significantly after the fifth decade of life. The etiology is multifactorial, encompassing both intrinsic and extrinsic mechanisms. Intrinsic theories focus on age-related microvascular changes, particularly the "watershed zone" of hypovascularity near the supraspinatus insertion, as well as cellular apoptosis and collagen degeneration. Extrinsic theories highlight mechanical impingement from the coracoacromial arch, altered scapular kinematics, and repetitive microtrauma.

Furthermore, the chronicity of the tear dictates the biological quality of the tissue we are attempting to repair. Chronic, retracted tears lead to muscle atrophy and fatty infiltration, changes that are largely irreversible even after successful structural repair. The mechanical environment of a torn rotator cuff alters the delicate force couples of the shoulder, leading to superior migration of the humeral head, accelerated glenohumeral arthritis (cuff tear arthropathy), and profound functional disability. Recognizing these pathological cascades is what elevates a surgeon from a mere technician to a master clinician, allowing for precise patient selection and tailored surgical strategies.
Detailed Surgical Anatomy and Biomechanics
Before we make any incision, a profound understanding of the shoulder's intricate anatomy and the biomechanical interplay of its structures is paramount. The rotator cuff is a complex of four muscles originating from the scapula and inserting onto the tuberosities of the proximal humerus, acting synergistically to provide dynamic stability through concavity compression and to allow a wide, multi-planar range of motion.
The Rotator Cuff Muscles and Their Footprint
The supraspinatus is our primary focus in the majority of degenerative tears. Originating from the supraspinous fossa, it inserts onto the superior facet of the greater tuberosity. It is innervated by the suprascapular nerve (C5, C6). The native footprint is critical: it averages 25 mm in anterior-posterior width, with a medial-to-lateral footprint of approximately 12.1 mm at the midtendon. The infraspinatus, originating from the infraspinous fossa, inserts onto the middle facet. It averages 29 mm wide, with a mean medial-to-lateral width of 19 mm, and its tendon fibers intimately interdigitate with the posterior supraspinatus.
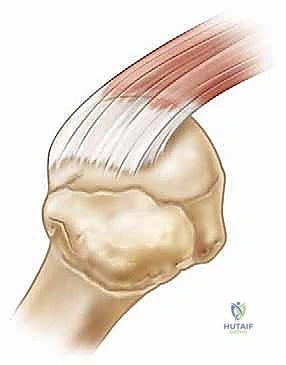
The teres minor and subscapularis complete the cuff. The teres minor inserts onto the inferior facet, innervated by the axillary nerve, and assists in external rotation. The subscapularis, the massive anterior stabilizer, inserts onto the lesser tuberosity and is innervated by the upper and lower subscapular nerves. From a neurovascular standpoint, the suprascapular nerve is at extreme risk during excessive medial dissection or retraction, particularly as it passes through the spinoglenoid notch. The axillary nerve courses inferiorly, making it vulnerable during inferior capsular releases or when establishing low anterior or posterior portals.
Biomechanics of the Rotator Cable and Crescent
Stephen Burkhart beautifully described the biomechanical concept of the "rotator cable" and the "crescent." The rotator cable is a thick bundle of fibers running perpendicular to the supraspinatus and infraspinatus tendons, extending from the coracohumeral ligament anteriorly to the inferior border of the infraspinatus posteriorly. This cable acts like a suspension bridge, stress-shielding the thinner, more distal "crescent" tissue that inserts directly onto the greater tuberosity.
In older patients, the crescent tissue often degenerates and tears, but if the cable remains intact, the shoulder may remain biomechanically compensated and asymptomatic. However, when a tear propagates through the cable, the suspension bridge fails, leading to massive retraction and loss of the transverse and coronal force couples. Our double-row repair aims to recreate this biomechanical stability. By placing medial anchors just lateral to the articular margin and spanning sutures laterally, we effectively recreate the compressive forces of the native footprint, minimizing micromotion and maximizing the surface area available for Sharpey's fiber integration.
Exhaustive Indications and Contraindications
The decision to proceed with an arthroscopic double-row rotator cuff repair requires a delicate balance of patient factors, anatomical considerations, and biological realities. Not every tear requires a double-row construct, and not every patient is a candidate for surgery. The goal is to select patients whose biological healing potential aligns with the mechanical superiority of the construct.
Generally, double-row or transosseous-equivalent repairs are highly indicated for medium to large full-thickness tears (typically 1.5 cm to 3 cm in anterior-posterior dimension) where there is adequate tendon mobility to reach the lateral footprint without excessive tension. In these cases, the double-row construct provides superior biomechanical strength, higher ultimate load to failure, and decreased gap formation compared to single-row repairs. It is also indicated in younger, highly active patients where maximizing the structural integrity of the repair is paramount for returning to high-demand activities.
Conversely, absolute contraindications include active joint infection, severe glenohumeral osteoarthritis (where an arthroplasty might be more appropriate), and profound medical comorbidities precluding surgery. Relative contraindications are heavily tied to tissue quality. Massive, immobile tears with severe fatty infiltration (Goutallier grade 3 or 4) or severe muscle atrophy are often biologically irreparable, even if mechanically approximated. In such cases, attempting a high-tension double-row repair will inevitably lead to failure, often a catastrophic Type II failure (medial row pull-through).
| Parameter | Indications for Double-Row Repair | Contraindications / Alternative Strategies |
|---|---|---|
| Tear Size | Medium to Large (1.5 cm - 3.0 cm) | Massive, fixed, retracted tears (>4 cm) lacking mobility |
| Tissue Quality | Good to excellent tendon thickness; minimal delamination | Severe degeneration, paper-thin tissue, severe delamination |
| Muscle Biology | Goutallier Grade 0, 1, or 2 (minimal fatty infiltration) | Goutallier Grade 3 or 4 (severe fatty infiltration/atrophy) |
| Patient Profile | Younger, active, high physical demands, compliant with rehab | Elderly, low demand, non-compliant, severe medical comorbidities |
| Bone Quality | Adequate tuberosity bone stock for multiple anchors | Severe osteopenia/osteoporosis (high risk of anchor pull-out) |
| Joint Status | Minimal to mild glenohumeral arthritis | Advanced cuff tear arthropathy (consider Reverse Shoulder Arthroplasty) |
Pre-Operative Planning, Templating, and Patient Positioning
Precise preoperative planning is non-negotiable for a successful arthroscopic rotator cuff repair. The blueprint for the surgery is drawn long before the patient enters the operating room, relying heavily on a meticulous physical examination and advanced imaging modalities.
Advanced Imaging and Classification
Magnetic Resonance Imaging (MRI) without contrast is our primary workhorse. We systematically evaluate the tear using established classification systems to predict reparability and outcomes. We utilize the Patte classification to assess the degree of tendon retraction in the coronal plane (Stage 1: proximal to footprint, Stage 2: level of humeral head, Stage 3: level of glenoid). The Thomazeau classification helps us quantify supraspinatus muscle atrophy by analyzing the occupation ratio of the muscle belly in the supraspinous fossa on the sagittal oblique T1 sequence.

Crucially, we assess fatty infiltration using the Goutallier classification on T1-weighted sagittal oblique images. A Goutallier grade of 3 (equal fat and muscle) or 4 (more fat than muscle) is a poor prognostic indicator for healing, often steering us away from heroic structural repairs toward partial repairs, superior capsular reconstruction, or tendon transfers. We also carefully evaluate the bone quality of the greater tuberosity; cystic changes or severe osteopenia will dictate our choice of anchors, often necessitating larger diameter or all-suture anchors to prevent pull-out during tensioning.
Anesthesia and Patient Positioning
Our standard protocol involves a regional interscalene nerve block combined with general anesthesia. The regional block provides profound preemptive analgesia, minimizes intraoperative volatile anesthetic requirements, and facilitates early postoperative comfort. Before positioning, an examination under anesthesia (EUA) is mandatory to assess passive range of motion. Fixed superior humeral migration or profound stiffness indicates secondary capsular contracture, alerting us to the need for a thorough capsular release prior to attempting tendon mobilization.
For double-row repairs, the lateral decubitus position is highly preferred over the beach chair position. The patient is secured in a vacuum beanbag, tilted slightly posteriorly to bring the glenoid parallel to the floor. An axillary roll is meticulously placed to protect the brachial plexus. The operative arm is suspended in a specialized traction device (e.g., Spider arm holder) with 5 to 10 pounds of traction, positioned at 30 to 45 degrees of abduction and 15 degrees of forward flexion. This specific vector distracts the glenohumeral joint and significantly expands the subacromial space, providing unparalleled visualization and working room for complex suture passing and anchor insertion.
Step-by-Step Surgical Approach and Fixation Technique
The execution of a double-row repair is a symphony of precise steps. My focus is on meticulous tissue handling, sequential mobilization, and biomechanically sound fixation.
Diagnostic Arthroscopy and Subacromial Preparation
We begin by establishing a standard posterior portal, 2 cm inferior and 1 cm medial to the posterolateral acromial corner. A systematic diagnostic arthroscopy of the glenohumeral joint is performed to evaluate the articular cartilage, labrum, and biceps tendon. If the biceps is significantly frayed or unstable, a tenodesis or tenotomy is performed, as leaving a pathological biceps can compromise the outcome of the cuff repair. We then establish an anterior-superior portal using an outside-in technique, ensuring it sits safely lateral to the coracoid to avoid the cephalic vein and musculocutaneous nerve.

The arthroscope is then redirected into the subacromial space, and a lateral working portal is established 3 cm distal to the lateral acromial edge. Using a motorized shaver and radiofrequency ablation wand, a radical bursectomy is performed. This is not merely for visualization; removing the hypertrophic, inflammatory bursa is critical for pain relief and creates a healthy biological environment. If a Type II or III acromion is causing mechanical impingement, a conservative acromioplasty is performed, taking care to preserve the coracoacromial ligament unless it is severely frayed, as it serves as a secondary restraint to superior humeral translation.
Tear Assessment and Meticulous Mobilization
With the subacromial space cleared, we formally assess the tear pattern (crescent, U-shaped, L-shaped, or massive contracted). For retracted tears, mobilization is the most critical and time-consuming step. We perform sequential releases to achieve a tension-free repair. First, an intra-articular release is performed, freeing the capsule from the undersurface of the tendon. Next, an extra-articular bursal release clears adhesions between the cuff and the deltoid fascia.

If the tendon remains retracted, we perform "interval slides." An anterior interval slide releases the coracohumeral ligament and the interval between the supraspinatus and subscapularis. A posterior interval slide releases the interval between the supraspinatus and infraspinatus. These releases must be performed meticulously with electrocautery, staying lateral to the glenoid rim to strictly avoid injury to the suprascapular nerve, which lies precariously close, roughly 1.5 cm medial to the superior glenoid rim.
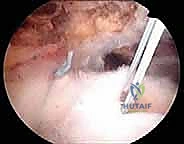
Footprint Preparation and Medial Row Fixation
Once adequate excursion is achieved, we prepare the greater tuberosity footprint. Using a motorized burr or a curette, we lightly decorticate the footprint to remove soft tissue and expose bleeding cancellous bone. We avoid excessive cortical resection, which can weaken anchor fixation. To enhance the biological milieu, we often use an awl to create "marrow vents" in the footprint, allowing bone marrow elements and mesenchymal stem cells to bathe the repair site.

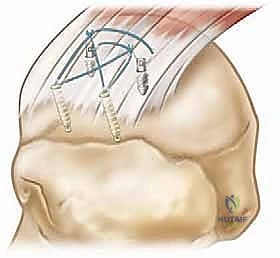
We then place our medial row anchors. These are inserted exactly at the articular margin, typically 1.5 to 2.0 cm apart. The trajectory is critical; anchors should be inserted at a 45-degree "deadman's angle" relative to the cortical surface to maximize pull-out strength. We typically use double- or triple-loaded suture anchors.

Suture Passing and Lateral Row Fixation
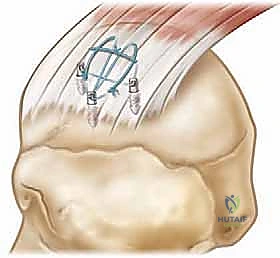
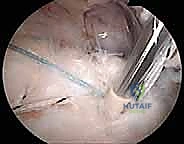
Suture passing requires a strategic approach. We utilize antegrade or retrograde suture passing devices (e.g., Suture Lasso or Scorpion) to pass the medial row sutures through the tendon. The bite should be taken approximately 12 to 15 mm medial to the lateral edge of the tendon to ensure a robust grasp of the healthy tendon tissue, incorporating the rotator cable if possible. We typically pass sutures in a mattress configuration to maximize tissue grip and prevent suture pull-through.
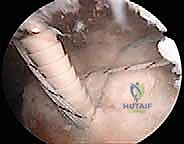
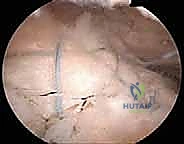
Once all medial sutures are passed, we do not tie them. Instead, we retrieve one suture limb from each medial anchor to create a crossed or parallel suture bridge configuration over the bursal surface of the tendon. These limbs are then loaded into knotless lateral row anchors.
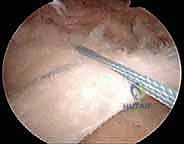
The lateral anchors are inserted into prepared bone sockets on the lateral aspect of the greater tuberosity, approximately 10 to 15 mm lateral to the edge of the footprint. As the lateral anchor is impacted into the bone, the suture bridge is tensioned, compressing the tendon firmly against the decorticated footprint. We must ensure the tension is adequate to eliminate gaps but not so tight as to strangulate the microvascular supply of the tendon.
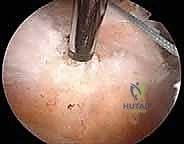
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, arthroscopic double-row rotator cuff repair carries inherent risks. A comprehensive understanding of potential complications, their incidence, and salvage strategies is mandatory for the operating surgeon. The most common complication is postoperative stiffness, while the most mechanically devastating is structural failure or retear.
Structural failure can occur at different interfaces. Type I failure occurs at the tendon-bone interface, often due to poor biological healing. Type II failure, unique to double-row and suture-bridge constructs, is a medial row failure where the tendon tears medial to the medial row of anchors. This is believed to be caused by over-tensioning of the medial row, stress shielding of the medial tendon, or progressive intrinsic degeneration. When a Type II failure occurs, the remaining tissue is often severely compromised, making revision repair exceedingly difficult.
Infection is rare but catastrophic. Cutibacterium acnes (formerly Propionibacterium acnes) is an indolent, slow-growing anaerobic bacterium native to the axillary flora that is increasingly recognized as a pathogen in shoulder surgery. It often presents atypically, with insidious pain and stiffness rather than overt signs of sepsis. Prevention requires meticulous skin preparation, often utilizing benzoyl peroxide preoperatively, and careful draping to isolate the axilla.
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage / Management Strategy |
|---|---|---|---|
| Postoperative Stiffness | 5% - 15% | Prolonged immobilization, poor pain control, intrinsic capsular fibrosis | Aggressive physical therapy, intra-articular corticosteroids; arthroscopic capsular release if refractory (>6 months). |
| Retear (Type I - Footprint) | 10% - 25% (size dependent) | Poor biology, non-compliance, trauma, inadequate fixation | Revision arthroscopic repair, patch augmentation, or conservative management if asymptomatic. |
| Retear (Type II - Medial) | 5% - 10% | Over-tensioning, stress shielding, severe tissue degeneration | Highly challenging. Consider superior capsular reconstruction (SCR), tendon transfer, or Reverse Arthroplasty if older. |
| Infection (C. acnes, S. aureus) | < 1% | Contamination from skin flora, immunocompromise, diabetes | Arthroscopic irrigation and debridement (I&D), removal of loose hardware, long-term targeted intravenous antibiotics. |
| Nerve Injury (Neurapraxia) | < 1% | Traction from positioning, errant portal placement, aggressive dissection | Usually transient. Observation, EMG/NCS at 6 weeks if no improvement. Avoid excessive traction (>10 lbs). |
| Anchor Pull-out / Osteolysis | 1% - 3% | Severe osteopenia, poor anchor trajectory, excessive tension | Revision with larger diameter anchors, all-suture anchors, or utilizing different fixation points (e.g., further distally). |
Phased Post-Operative Rehabilitation Protocols
The success of a double-row repair relies as much on the postoperative rehabilitation protocol as it does on the intraoperative technique. The biological integration of the tendon to the bone via Sharpey's fibers takes a minimum of 8 to 12 weeks. Therefore, our rehabilitation philosophy must strictly prioritize biological healing over rapid return of motion in the early phases.
Phase I: Protection and Restricted Motion (Weeks 0-6)
The primary goal is to protect the repair construct. The patient is immobilized in an abduction sling, which reduces tension on the supraspinatus repair. Only passive range of motion (PROM) is permitted, restricted to safe zones (typically forward flexion to 90 degrees, external rotation to 30 degrees in the scapular plane). Active motion of the elbow, wrist, and hand is encouraged to prevent distal stiffness. No active shoulder motion is allowed, as the contractile forces of the muscle can easily exceed the pull-out strength of the anchors in early healing.
Phase II: Active-Assisted and Active Motion (Weeks 6-10)
As the initial biological bond forms, we transition the patient out of the sling. We initiate active-assisted range of motion (AAROM) using pulleys and wands, gradually progressing to active range of motion (AROM) in all planes. The focus is on restoring normal scapulothoracic kinematics and eliminating compensatory shrugging mechanisms. Stretching is advanced to achieve full symmetric range of motion by the end of this phase.
Phase III: Early Strengthening (Weeks 10-16)
Once full, painless AROM is achieved, we introduce progressive resistance exercises. We begin with isometric exercises for the rotator cuff and periscapular musculature, advancing to isotonic exercises using light resistance bands and weights. The emphasis is on high-repetition, low-weight endurance training to re-educate the muscle fibers without overloading the maturing tendon-bone interface.
Phase IV: Advanced Strengthening and Return to Activity (Months 4-6+)
In the final phase, we focus on dynamic stabilization, proprioception, and sport-specific or work-specific functional training. Plyometric exercises and overhead lifting are gradually introduced. Return to heavy manual labor or contact sports is typically delayed until 6 months postoperatively, contingent upon symmetrical strength testing and a pain-free clinical examination.
Summary of Landmark Literature and Clinical Guidelines
The transition to double-row and transosseous-equivalent repairs is heavily supported by robust biomechanical literature, though the clinical superiority over single-row repairs remains a topic of nuanced debate in orthopedic academia.
Biomechanical studies by pioneers such as Mazzocca and Meier have unequivocally demonstrated that double-row constructs provide a significantly higher ultimate load to failure, greater initial fixation strength, and decreased gap formation under cyclic loading compared to single-row techniques. Furthermore, footprint contact area and pressure—critical factors for biological healing—are vastly maximized with the suture-bridge technique.
However, clinical outcome studies present a more complex picture. Landmark meta-analyses and randomized controlled trials (RCTs), such as those by Lapner and Hein, have shown that for small to medium tears (< 1.5 cm), there is often no statistically significant difference in clinical outcome scores (e.g., ASES, Constant scores) or retear rates between single and double-row repairs.
Conversely, for large and massive tears (> 3 cm), the clinical literature, including seminal work by Sugaya and Cole, strongly favors the double-row construct. In these larger tears, the double-row technique demonstrates significantly lower structural retear rates on postoperative MRI or ultrasound, correlating with better long-term strength and functional preservation. Therefore, current clinical guidelines recommend a tailored approach: single-row repairs may be sufficient for small, non-retracted tears, while double-row or transosseous-equivalent constructs are the gold standard for medium, large, and retracted tears to optimize both mechanical stability and biological healing.
