Approach to the Lumbar Spine: Mastering Techniques & Anatomy

Key Takeaway
In this comprehensive guide, we discuss everything you need to know about Approach to the Lumbar Spine: Mastering Techniques & Anatomy. The approach to the lumbar spine utilizes both anterior and posterior surgical techniques to treat various pathologies. Posterior approaches, including minimally invasive methods, are frequently used to access posterior spinal elements, the spinal cord, and intervertebral discs. Anterior approaches, such as transperitoneal and retroperitoneal, target conditions like vertebral body infection, fracture, or tumors affecting the spine's anterior elements.
Comprehensive Introduction and Patho-Epidemiology
The anatomy of the human spine represents a marvel of evolutionary biomechanics, transitioning from a quadrupedal suspension bridge to a bipedal weight-bearing column. This axial skeleton varies significantly from region to region to accommodate distinct functional demands. The cervical spine is relatively light, small, and highly flexible, prioritizing range of motion for the cranium. Conversely, the thoracic spine is larger and relatively immobile, tethered by its articulation with the rib cage to protect vital cardiopulmonary structures. The lumbar spine, particularly its lower segments, possesses greater mobility than the thoracic spine but less than the cervical spine. It is tasked with the monumental biomechanical burden of supporting the entire upper body while facilitating truncal flexion, extension, and rotation.
Because the cervical and lumbar regions are the most mobile portions of the axial skeleton, they are subjected to the highest sheer and compressive forces. Consequently, pathology is overwhelmingly concentrated in these areas. Degenerative disc disease, spinal stenosis, spondylolisthesis, and disc herniations are endemic in the aging population, making the lumbar spine the most frequently operated region in orthopedic spine surgery. Understanding the patho-epidemiology of these conditions is paramount for the operating surgeon. The degenerative cascade, initially described by Kirkaldy-Willis, outlines the progression from localized synovial joint dysfunction to segmental instability, and finally to restabilization via osteophyte formation and hypertrophy, often at the expense of neural volume.
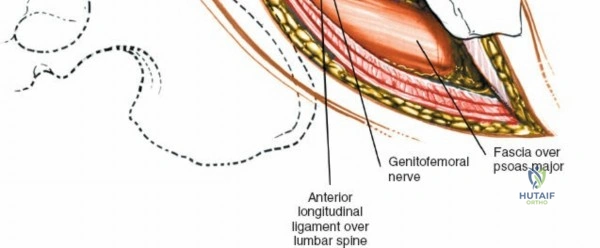
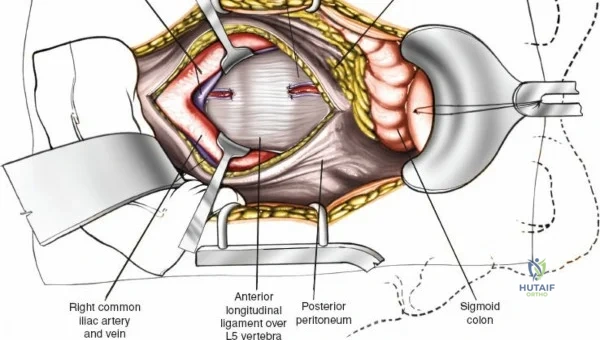
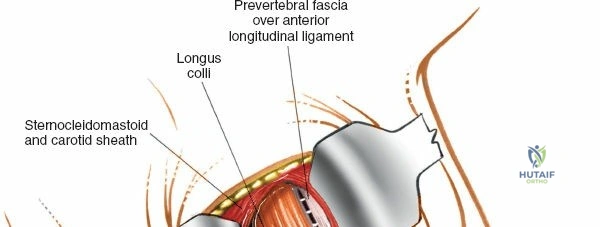
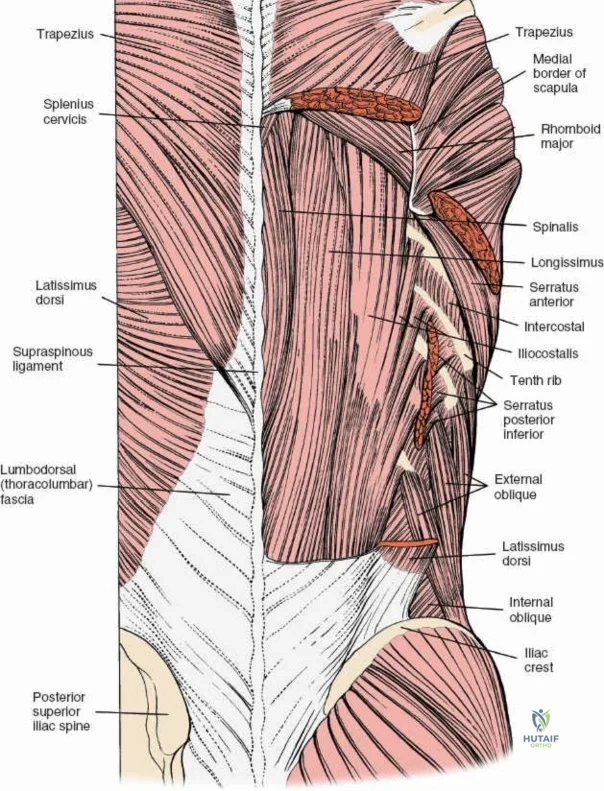
To effectively treat pathology of both the anterior and posterior elements of the spinal column, the orthopedic surgeon must be intimately familiar with a variety of surgical approaches. Pathologies such as vertebral body osteomyelitis, burst fractures, and primary or metastatic tumors often dictate an anterior approach to achieve adequate corpectomy and anterior column reconstruction. However, posterior approaches remain the workhorse of the spine surgeon's armamentarium. The midline posterior approach is the most ubiquitous, permitting unparalleled access to the posterior spinal elements (spinous processes, laminae, facet joints, and pedicles), as well as the spinal canal, cauda equina, and intervertebral discs.
Furthermore, when segmental instability or deformity necessitates arthrodesis, the posterior approach facilitates robust posterolateral and interbody fusion techniques. While the iliac crest remains the gold standard for autologous bone graft harvesting (detailed in pelvic surgical texts), modern posterior approaches often utilize local laminectomy bone, allografts, and orthobiologics to achieve solid arthrodesis. Mastery of the posterior approach to the lumbar spine is an absolute prerequisite for any orthopedic surgeon, forming the foundational exposure upon which both simple decompressions and complex deformity corrections are built.
Detailed Surgical Anatomy and Biomechanics
Osteology and Articular Anatomy
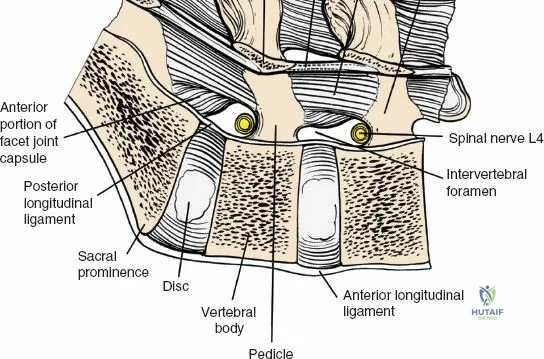
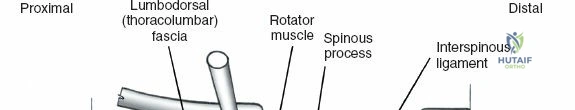
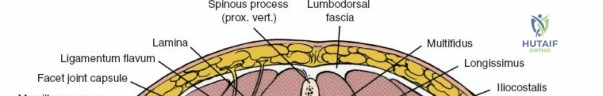
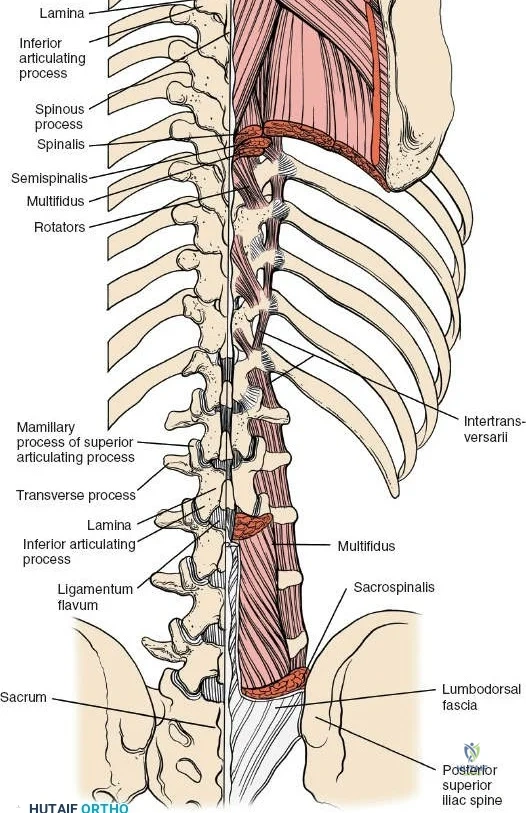
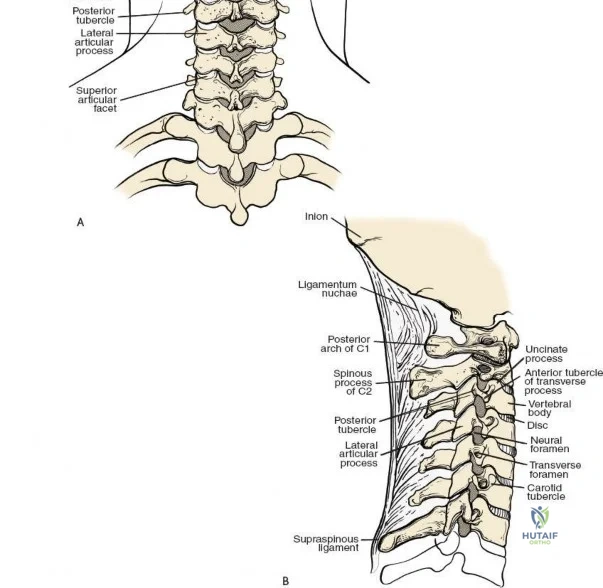
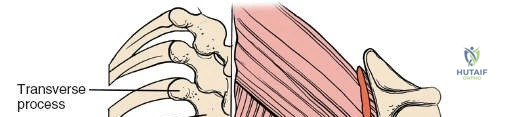
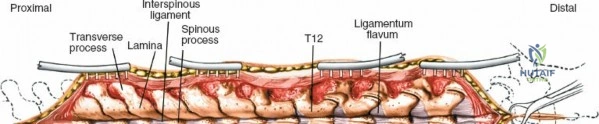
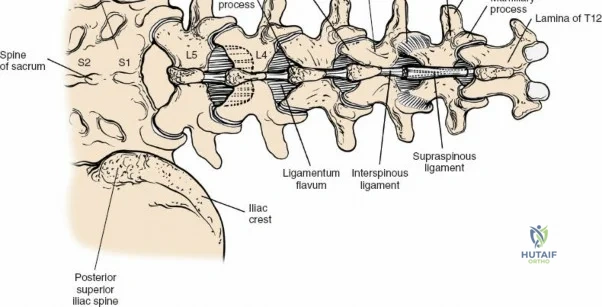
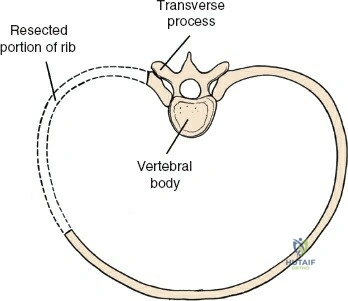
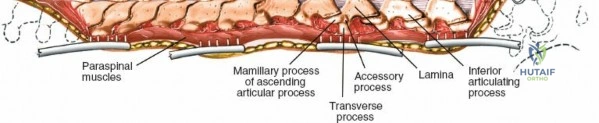
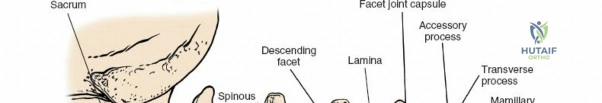
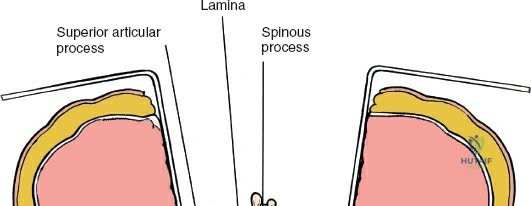
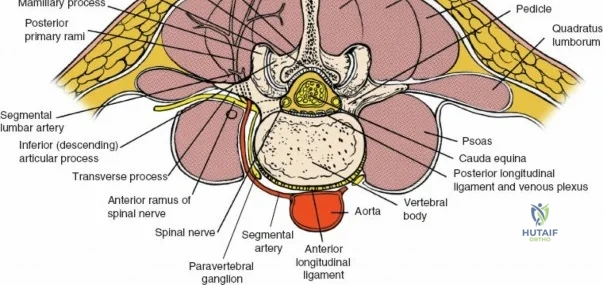
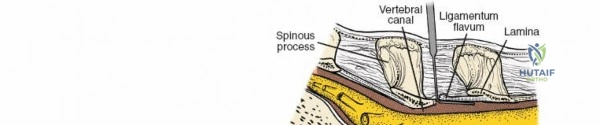
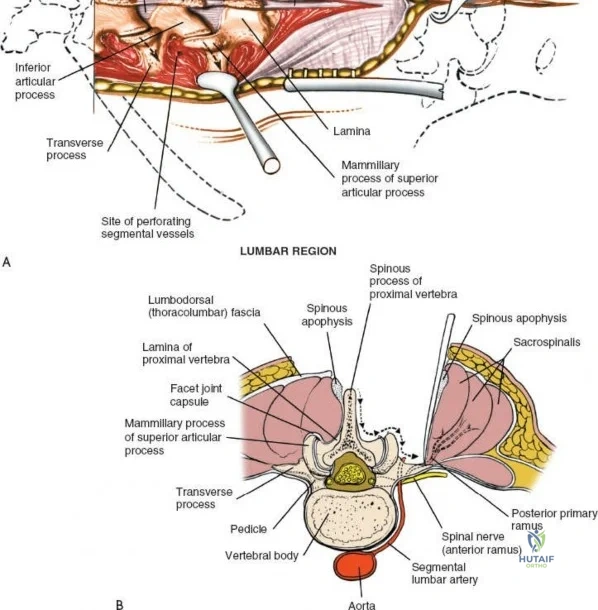
The lumbar vertebrae (L1-L5) are characterized by massive, kidney-shaped vertebral bodies designed to withstand immense axial loads. Posteriorly, the neural arch is formed by the stout pedicles, which project directly posteriorly from the upper half of the vertebral body, and the broad, thick laminae that converge in the midline to form the spinous process. The pedicle is a critical anatomical landmark, serving as the primary corridor for transpedicular screw fixation. The pars interarticularis, the region of the lamina between the superior and inferior articular processes, is uniquely susceptible to fatigue fractures (spondylolysis), particularly in adolescent athletes subjected to repetitive hyperextension.
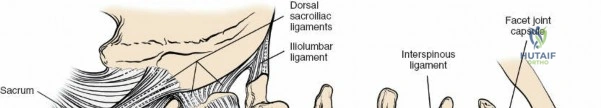
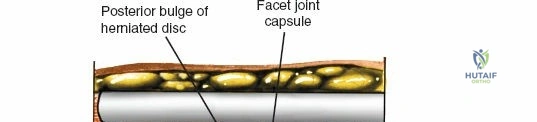
The facet (zygapophyseal) joints are true synovial joints formed by the articulation of the inferior articular process of the cephalad vertebra and the superior articular process of the caudal vertebra. In the lumbar spine, these joints are oriented primarily in the sagittal plane, permitting flexion and extension while highly restricting axial rotation. Hypertrophy of the superior articular process, often a sequela of disc space collapse and facet overriding, is a primary driver of lateral recess and foraminal stenosis, directly impinging upon the traversing and exiting nerve roots, respectively.
Ligamentous and Muscular Support
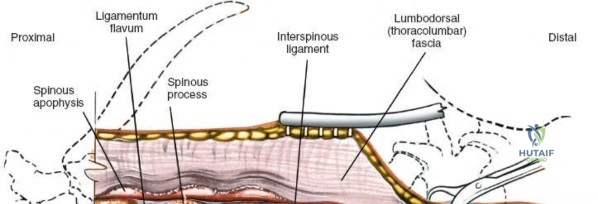
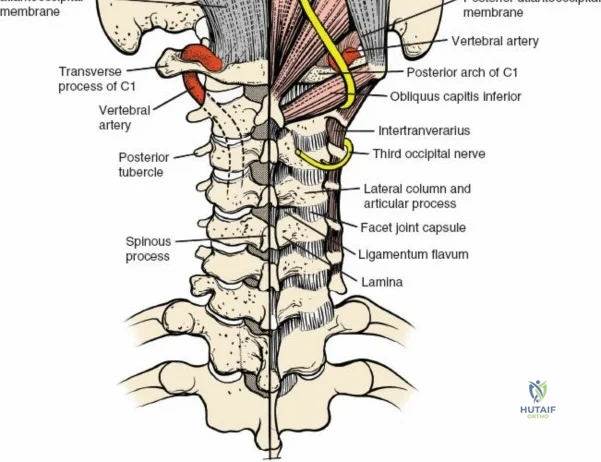
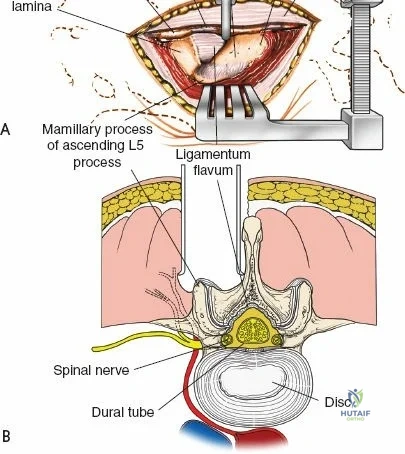
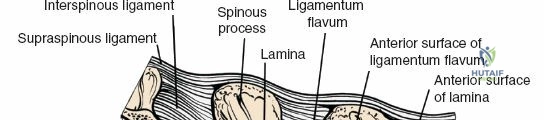
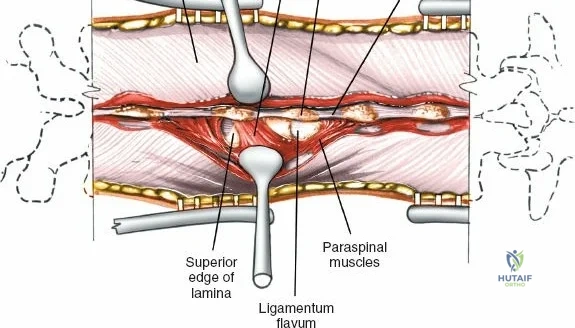
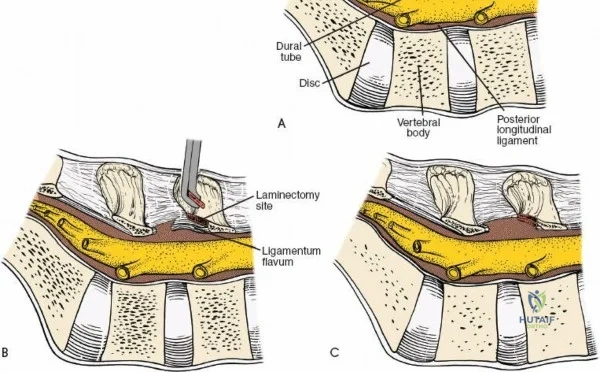
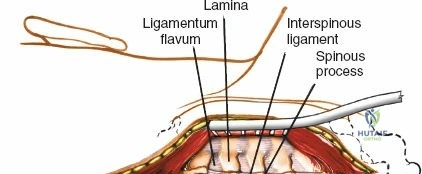
The posterior tension band of the lumbar spine is a complex interplay of robust ligaments and dynamic muscular stabilizers. The supraspinous ligament connects the apices of the spinous processes, while the interspinous ligament connects adjoining spinous processes. Deep to these lies the ligamentum flavum, a highly elastic, yellowish ligament that bridges the interlaminar space. The ligamentum flavum attaches to the anterior surface of the cephalad lamina and the superior edge of the caudal lamina. With age and disc degeneration, the ligamentum flavum hypertrophies and buckles anteriorly into the spinal canal during extension, contributing significantly to central canal stenosis.
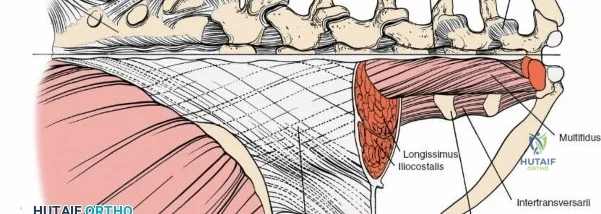
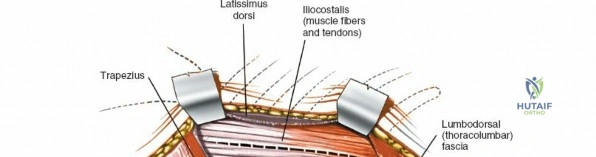
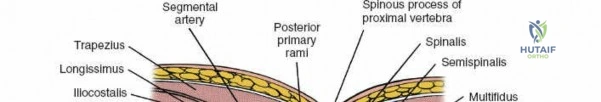
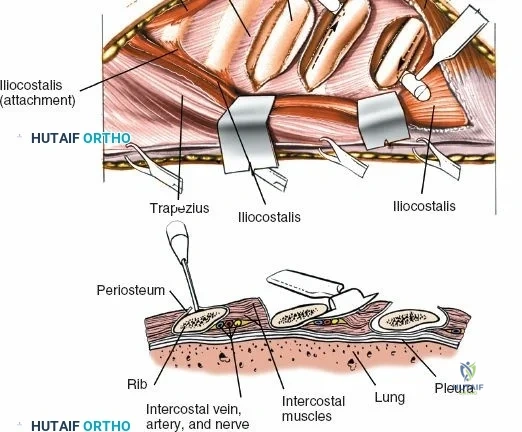
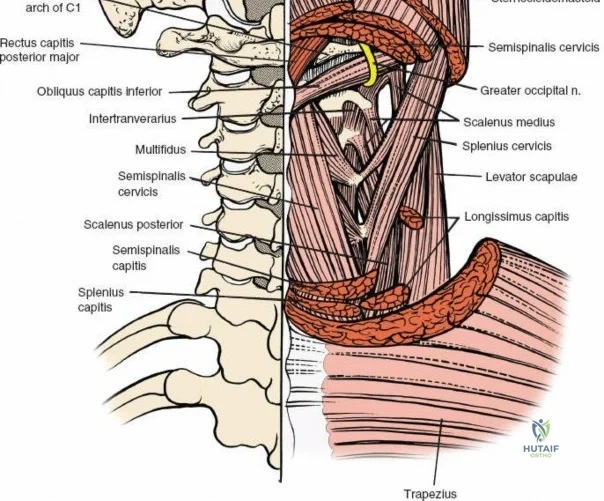
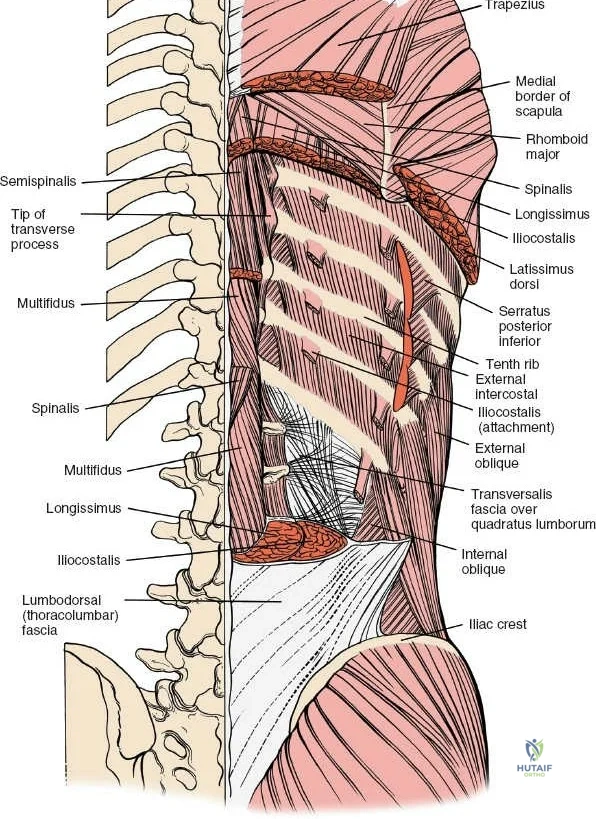
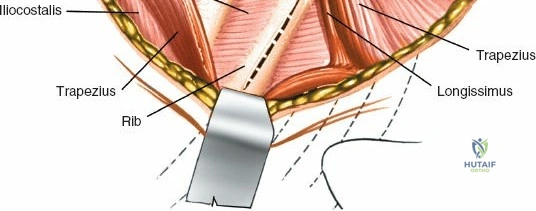
The muscular envelope of the posterior lumbar spine is primarily composed of the erector spinae group (iliocostalis, longissimus, spinalis) and the deeper transversospinalis group, notably the multifidus. The multifidus is a critical dynamic stabilizer, originating from the mammillary processes of the facet joints and inserting onto the spinous processes of superior vertebrae. Subperiosteal stripping and prolonged retraction of these muscles during standard open posterior approaches can lead to ischemic necrosis, denervation, and subsequent multifidus atrophy, which correlates strongly with failed back surgery syndrome and chronic postoperative axial pain.
Neurovascular Anatomy
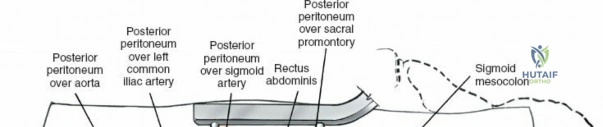
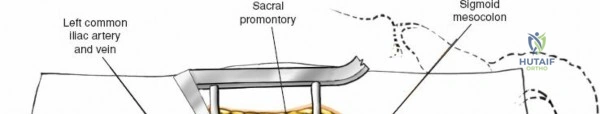
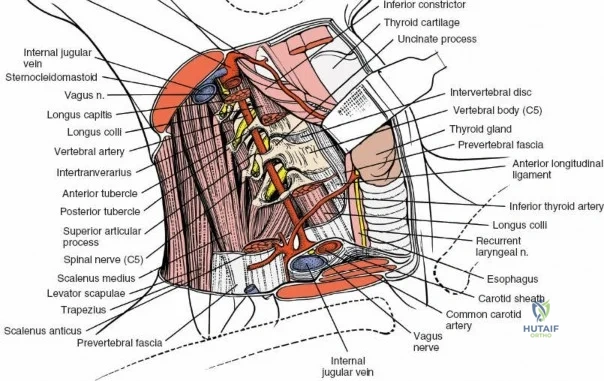
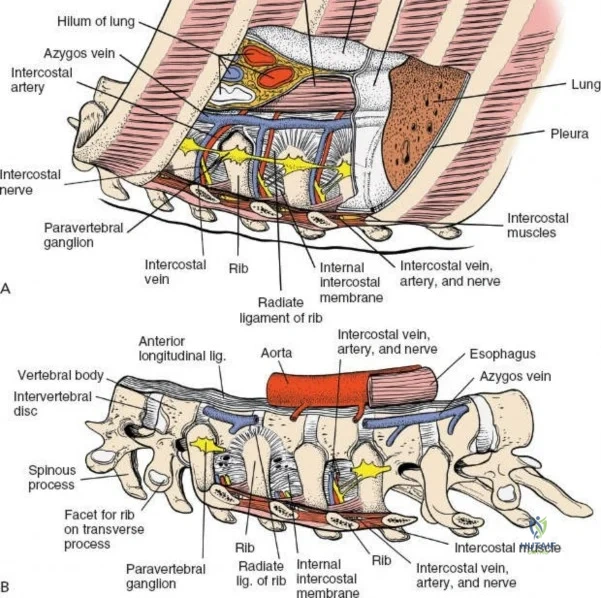
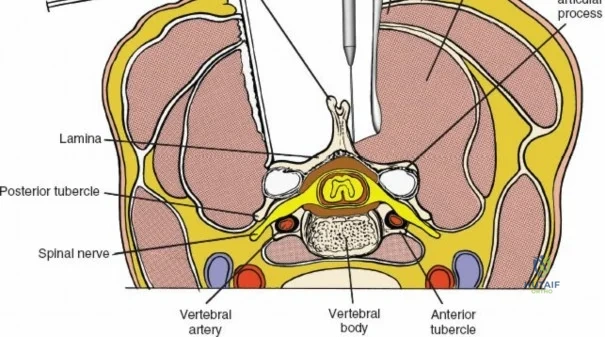
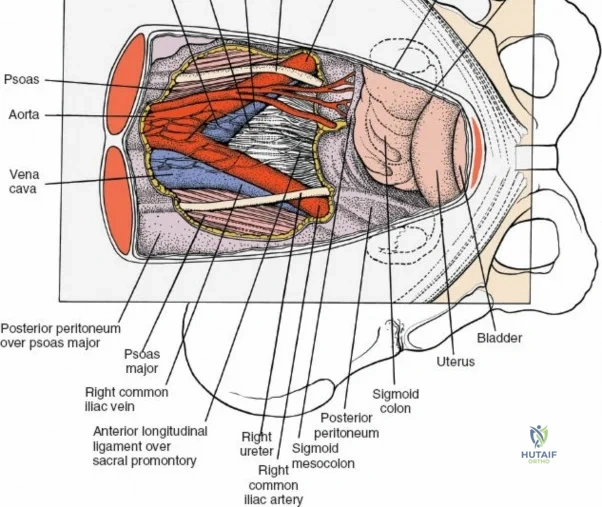
The neural anatomy of the lumbar spine dictates the precision required during decompression. The spinal cord typically terminates at the conus medullaris at the L1-L2 level. Caudal to this, the thecal sac contains the cauda equina, a collection of ventral and dorsal nerve roots floating in cerebrospinal fluid. Each lumbar nerve root exits the spinal canal through the intervertebral foramen below its corresponding pedicle (e.g., the L4 nerve root exits the L4-L5 foramen below the L4 pedicle). The surgeon must clearly distinguish between the traversing root (which is compressed by paracentral disc herniations) and the exiting root (which is compressed by far lateral disc herniations or foraminal stenosis).
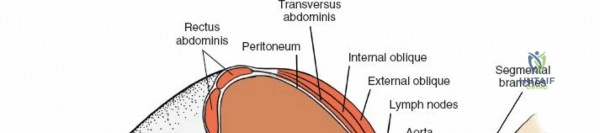
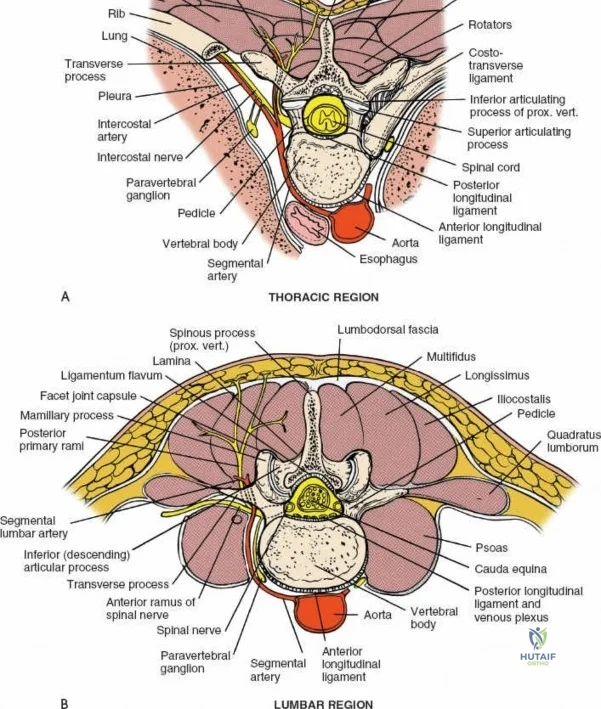
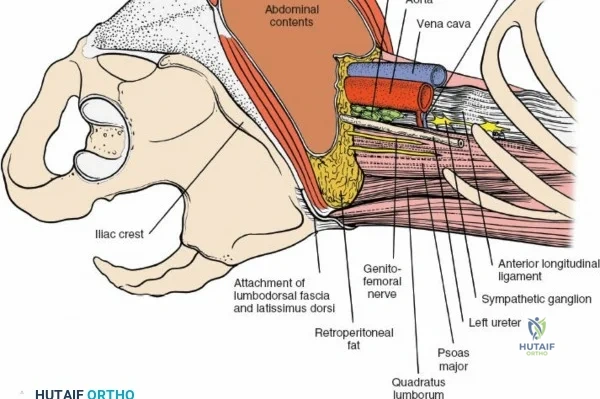
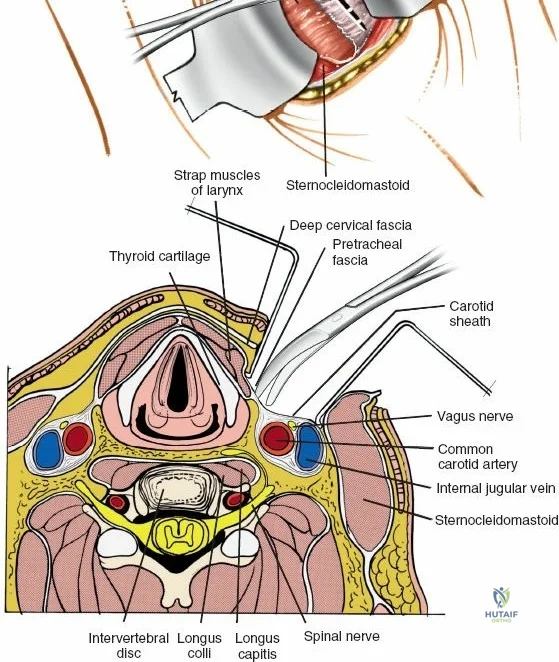
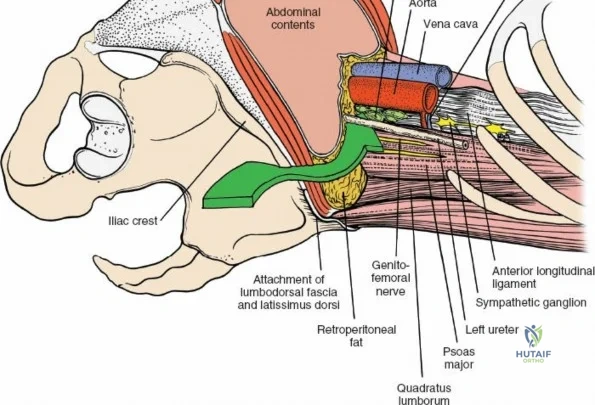
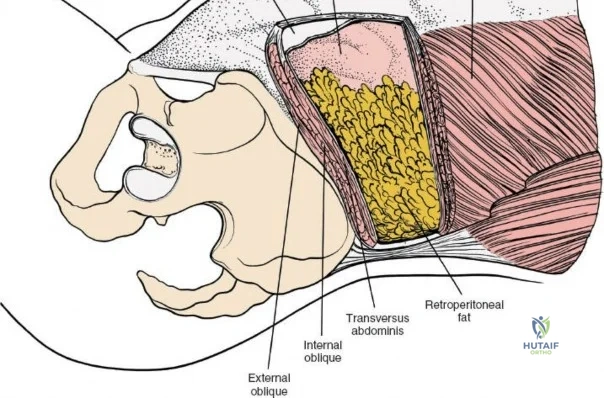
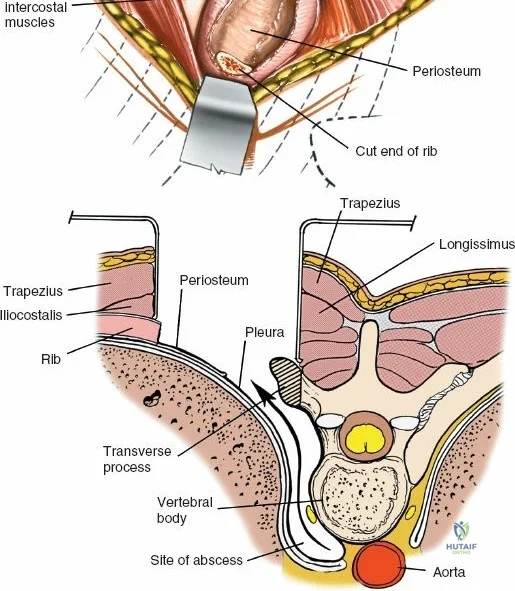
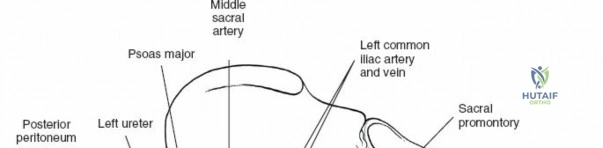
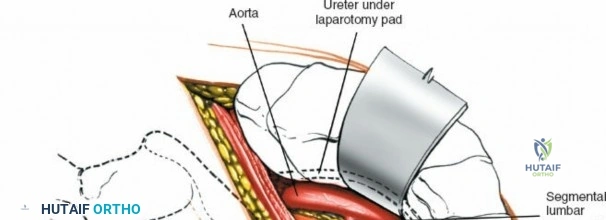
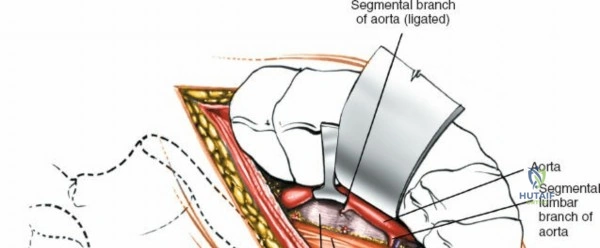
Vascularly, the posterior approach encounters the segmental lumbar arteries, which branch from the aorta and course around the vertebral bodies. Their dorsal branches supply the paraspinal musculature and facet joints. These vessels frequently bleed vigorously as dissection is carried laterally over the transverse processes. Within the spinal canal, Batson's venous plexus—a valveless, complex network of epidural veins—surrounds the thecal sac. This plexus communicates freely with the inferior vena cava. Increased intra-abdominal pressure engorges these veins, transforming a routine microdiscectomy into a hemorrhagic challenge. Meticulous patient positioning is therefore paramount to decompress the abdomen and collapse Batson's plexus.
Exhaustive Indications and Contraindications
The decision to proceed with a posterior approach to the lumbar spine must be predicated on a rigorous correlation of the patient's clinical presentation, physical examination findings, and advanced imaging. The posterior approach is highly versatile, but patient selection remains the ultimate determinant of surgical success.
Indications for Posterior Lumbar Surgery
Surgical intervention is generally indicated when conservative management (physical therapy, NSAIDs, epidural steroid injections) has failed after a period of 6 to 12 weeks, or immediately in the presence of progressive neurologic deficit or cauda equina syndrome.
- Herniated Nucleus Pulposus (HNP): Symptomatic radiculopathy correlating with an extruded or protruded disc fragment impinging on a nerve root.
- Lumbar Spinal Stenosis: Neurogenic claudication or severe radiculopathy secondary to central canal, lateral recess, or foraminal narrowing caused by facet hypertrophy, ligamentum flavum thickening, and disc bulging.
- Degenerative Spondylolisthesis: Forward translation of one vertebra over another, typically at L4-L5, with associated stenosis and mechanical back pain, often requiring decompression and instrumented fusion.
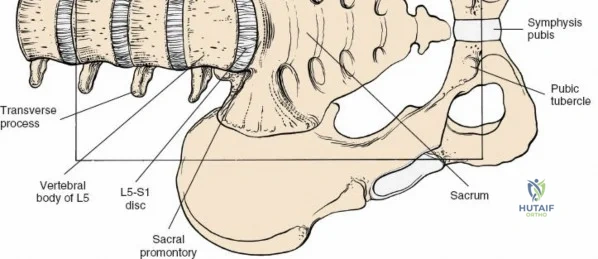
- Isthmic Spondylolisthesis: Pars interarticularis defects leading to slippage, most common at L5-S1.
- Trauma: Posterior column fractures, chance fractures, or burst fractures requiring posterior tension band restoration and stabilization.
- Infection and Neoplasm: Epidural abscess evacuation, or debulking/resection of primary or metastatic posterior element tumors.
Contraindications to the Posterior Approach
Contraindications are generally relative but require careful preoperative optimization and surgical planning. Active superficial skin infections or deep tissue infections at the planned surgical site represent an absolute contraindication to elective procedures.
Relative contraindications include profound medical comorbidities (severe cardiopulmonary disease) that preclude prone positioning or general anesthesia. Furthermore, certain anterior pathologies, such as severe rigid kyphotic deformities or massive anterior column osteomyelitis with extensive vertebral body destruction, may not be adequately addressed via a posterior-only approach and may necessitate an anterior or combined anterior-posterior strategy.
| Category | Indications | Contraindications |
|---|---|---|
| Degenerative | Spinal stenosis, Herniated disc, Degenerative spondylolisthesis | Asymptomatic imaging findings, Predominantly axial pain without instability |
| Trauma | Flexion-distraction injuries, Unstable burst fractures, Fracture-dislocations | Stable compression fractures (non-neurologic) |
| Deformity | Adult degenerative scoliosis, Flatback syndrome | Rigid, severe anterior column defects requiring anterior release |
| Oncology/Infection | Epidural compression, Posterior element tumors, Epidural abscess | Overlying active cellulitis/soft tissue infection |
Pre-Operative Planning, Templating, and Patient Positioning
Imaging and Pre-Operative Templating
Meticulous preoperative planning is the cornerstone of safe and effective spine surgery. Standard weight-bearing anteroposterior (AP) and lateral radiographs of the lumbar spine are mandatory to assess overall alignment, lordosis, and gross instability. Dynamic flexion-extension radiographs are critical for unmasking occult segmental instability, particularly in the setting of spondylolisthesis.
Magnetic Resonance Imaging (MRI) without contrast is the gold standard for evaluating neural element compression, disc hydration, and soft tissue pathology. In cases of prior lumbar surgery, gadolinium contrast should be utilized to differentiate epidural scar tissue (which enhances) from recurrent disc herniation (which typically does not enhance centrally). Computed Tomography (CT) is invaluable for assessing bony anatomy, facet joint arthropathy, pars defects, and for preoperative templating of pedicle screw trajectories and diameters.
Patient Positioning and Operating Room Setup
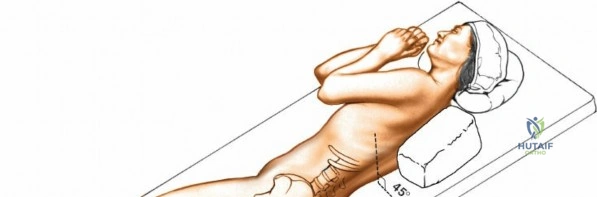
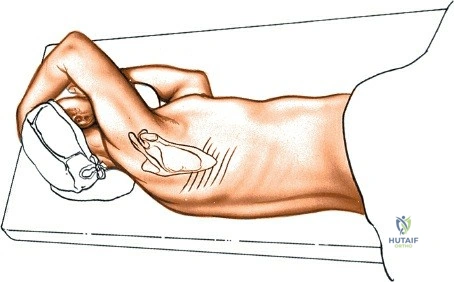
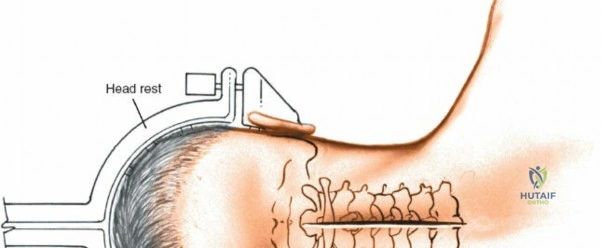
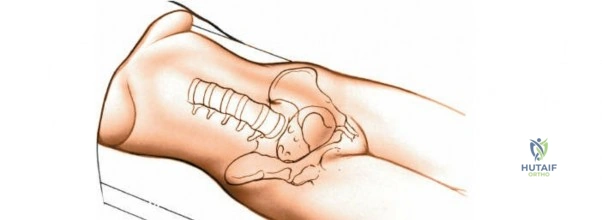
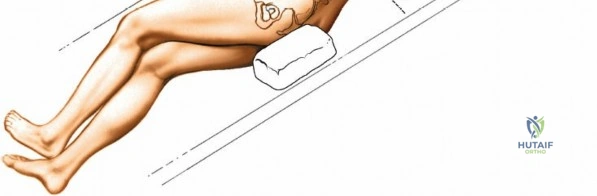
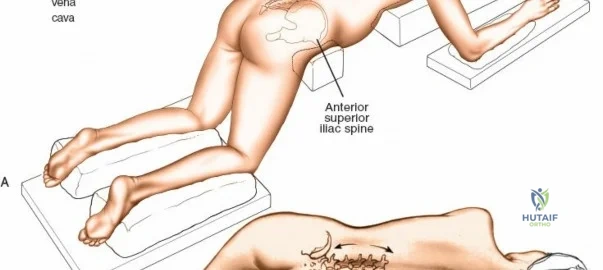
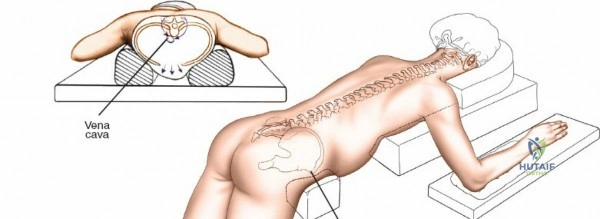
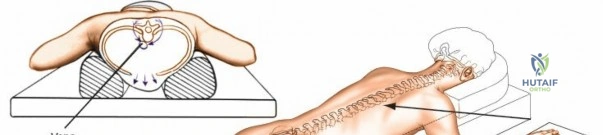
The posterior approach can be undertaken with the patient in either a prone position on a standard radiolucent table with chest/iliac bolsters, or on a specialized spine frame (e.g., Jackson table, Wilson frame). The primary goal of positioning is twofold: protecting the patient from pressure-related injuries and optimizing the surgical field.
Following intubation, logroll the patient into the prone position. It is absolutely critical that bolsters are placed longitudinally under the patient’s lateral thorax and iliac crests to allow the abdomen to hang entirely free. This prevents compression of the inferior vena cava, which would otherwise shunt venous return through Batson's epidural venous plexus, leading to catastrophic bleeding during decompression.
The shoulders should be placed at no more than 90 degrees of abduction and slightly flexed forward to relax the brachial plexus. Careful padding of the ulnar nerve at the cubital tunnel and the median nerve at the wrist must be assured. Position the head and neck in a relaxed, neutral position using a specialized foam face mask or Mayfield pins. Ensure absolutely no pressure is applied to the globes of the eyes. Avoid having the head lower than the rest of the body; maintaining a neutral or slightly elevated head position reduces venous congestion and mitigates the devastating risk of postoperative visual loss (POVL), which is associated with high hydrostatic pressure, prolonged operative times, and blood loss.
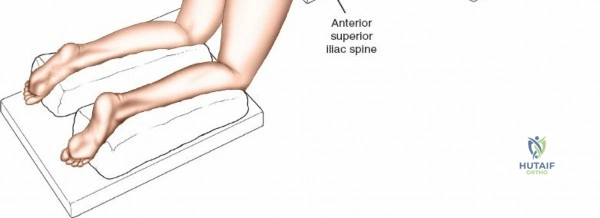
Pad the lower extremities carefully at the knees and feet. The positioning of the hips and knees dictates the sagittal profile of the lumbar spine. If the approach is to be used solely for decompression, flex the hips and knees (e.g., using a Wilson frame); this flattens the lumbar lordosis, opening the interlaminar and interspinous distances, thereby facilitating access to the spinal canal. Conversely, for lumbar fusions, place the hips and thighs in neutral or slight extension (e.g., on a Jackson flat frame) to restore and lock in physiologic lumbar lordosis. Flex the knees slightly and check that there is no pressure on the proximal fibula to protect the common peroneal nerve.
Step-by-Step Surgical Approach and Fixation Technique
Landmarks and Incision
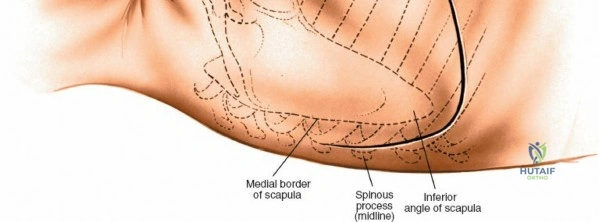
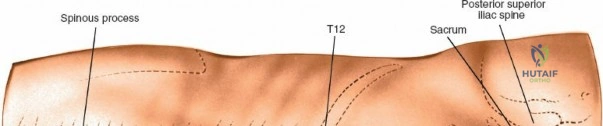
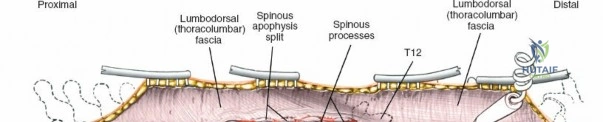
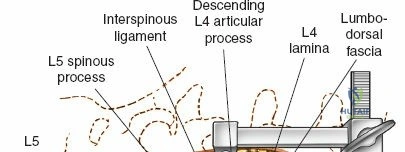
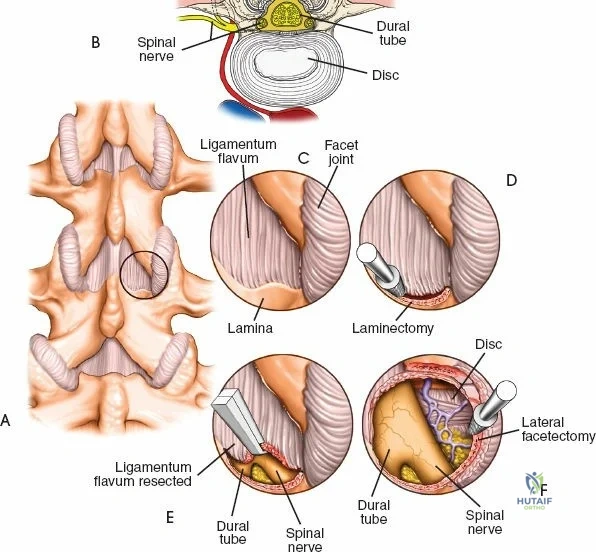
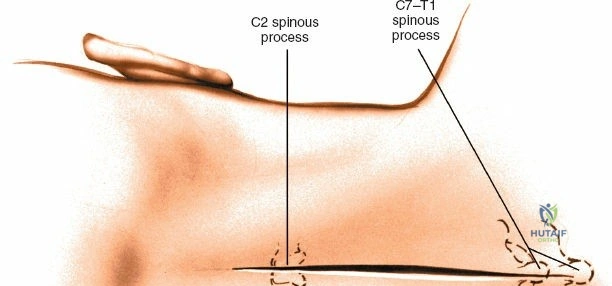
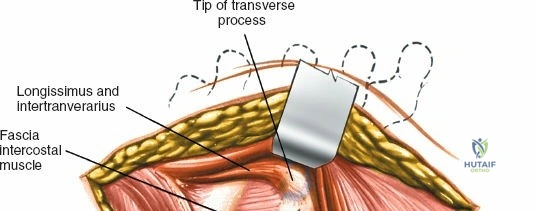
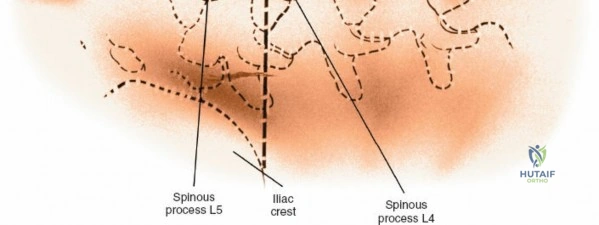
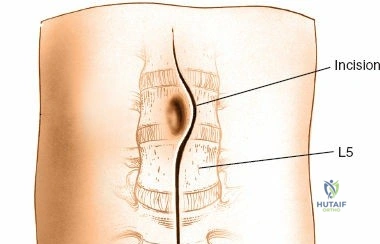
Accurate localization is the most critical initial step. Palpate the spinous processes in the midline. Note that Tuffier's line, an imaginary line drawn between the highest points of the iliac crests, classically intersects the L4-L5 interspace or the L4 spinous process. However, this is only a rough topographical guide and varies based on patient habitus and pelvic morphology. The only definitive means of determining the exact surgical level is via intraoperative fluoroscopy. Insert a spinal needle into the interspinous ligament or tap a clamp onto the spinous process and obtain a lateral radiograph prior to making the skin incision.
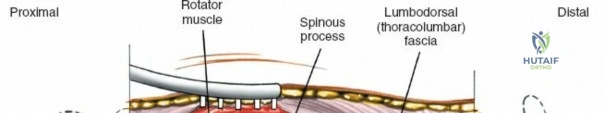
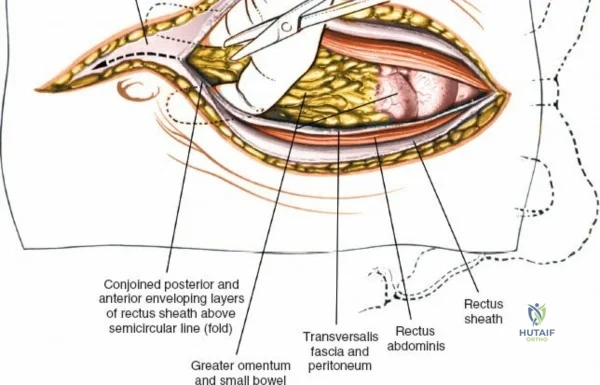
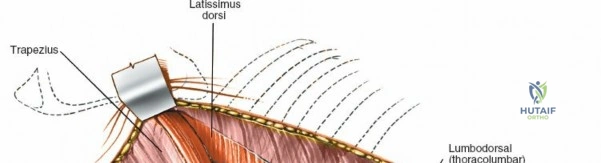
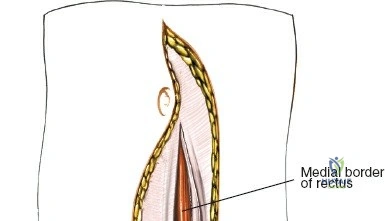
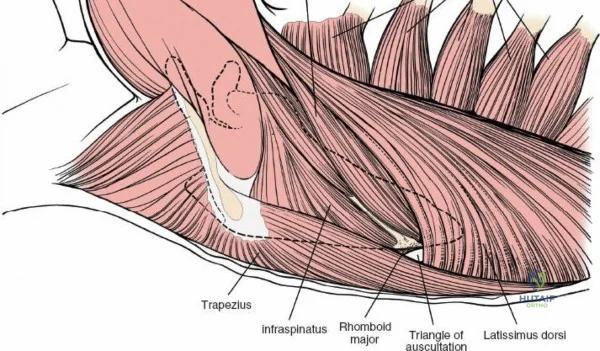
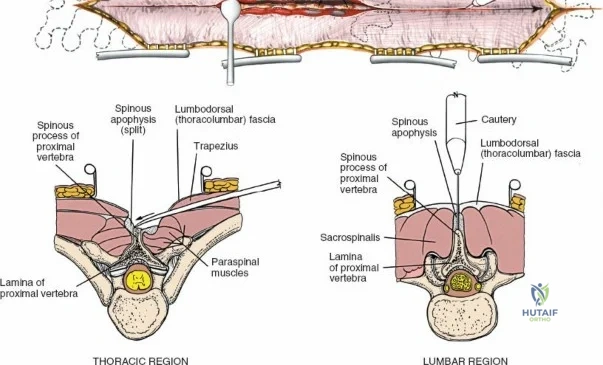
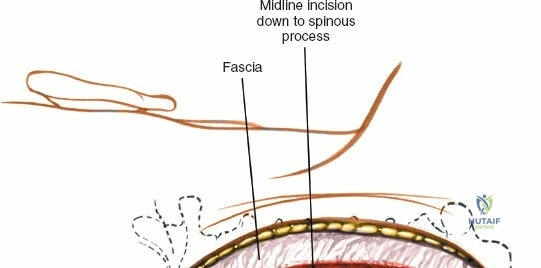
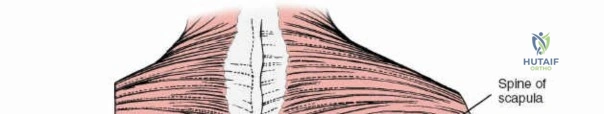
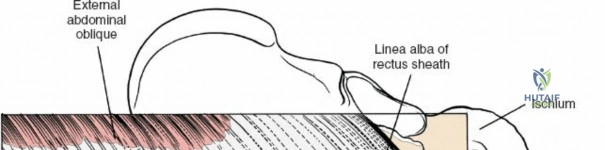
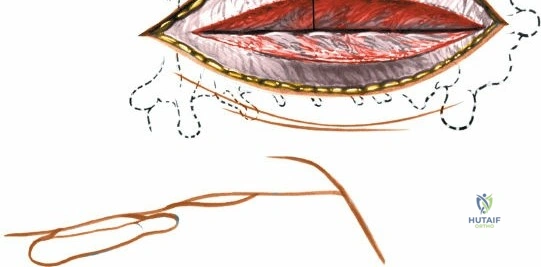
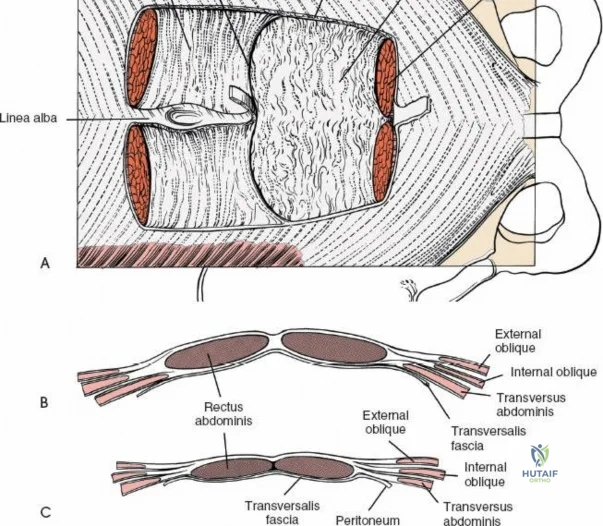
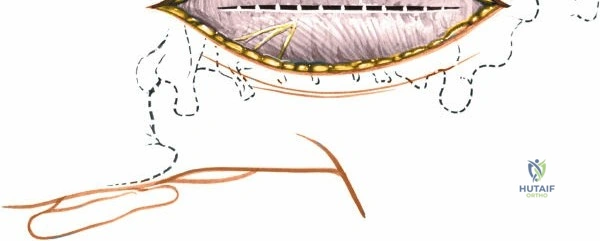
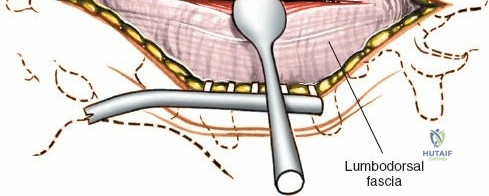
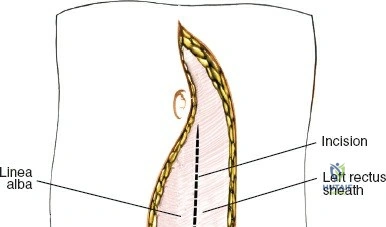
Make a longitudinal midline incision precisely over the spinous processes, extending from the spinous process above to the spinous process below the level of pathology. Deepen the incision through the subcutaneous fat using electrocautery until the thick, white lumbodorsal fascia is encountered. Maintain strict hemostasis in the superficial layers to prevent postoperative hematoma formation.
Superficial and Deep Surgical Dissection
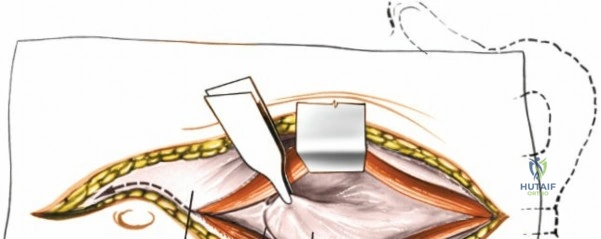
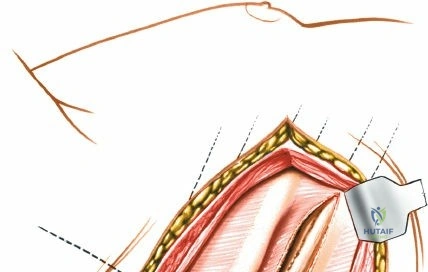
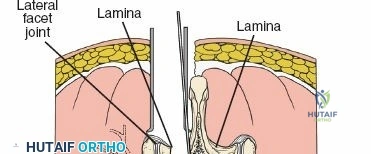
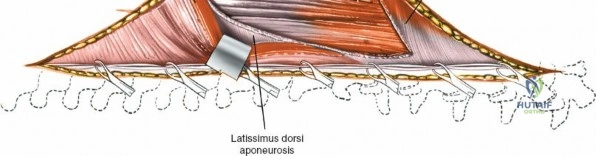
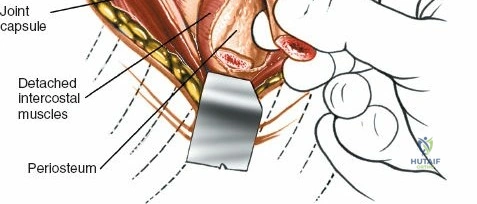
Incise the lumbodorsal fascia in the midline, directly over the tips of the spinous processes. Using a Cobb elevator and electrocautery, detach the paraspinal muscles subperiosteally from the spinous processes and laminae. In a young patient, the tip of the spinous process is a cartilaginous apophysis; it can be split sharply in the midline, leaving the muscular attachments to the apophysis intact, which makes subperiosteal muscle removal easier and improves postoperative healing.
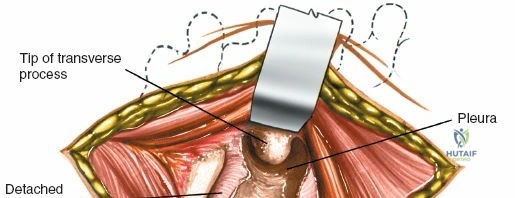
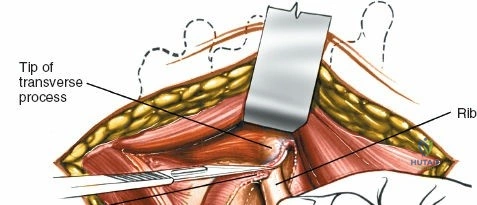
Dissect the paraspinal muscles laterally from the spinous process and lamina down to the facet joint. Pack the subperiosteal space tightly with surgical sponges to achieve hemostasis and maintain the plane. Continue dissecting laterally, stripping the joint capsule from the descending and ascending facets. The vessels supplying the paraspinal muscles on a segmental basis are located close to the facet joints, in the area between the transverse processes. These dorsal branches of the lumbar vessels frequently bleed vigorously as the dissection is carried out laterally. Vigorous bipolar or monopolar cauterization of these vessels is necessary. Note that the posterior primary rami of the lumbar nerves, which innervate the paraspinal muscles, run with these vessels and may be damaged by aggressive cautery. Fortunately, collateral segmental innervation prevents total denervation of the erector spinae.
Extensile Measures and Decompression Techniques
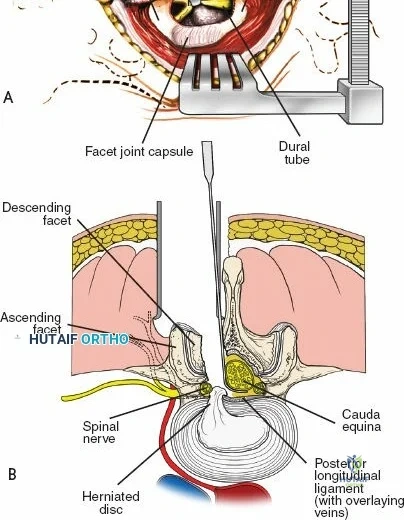
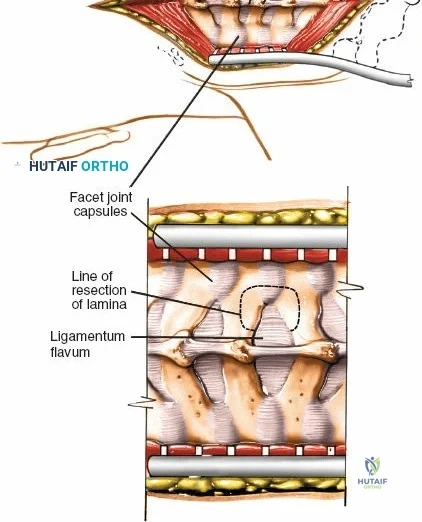
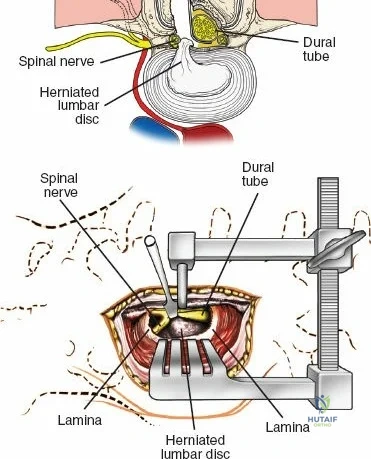
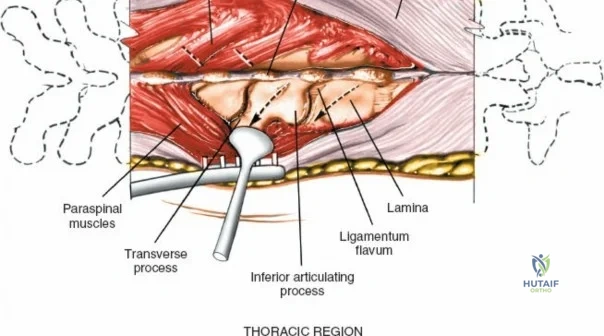
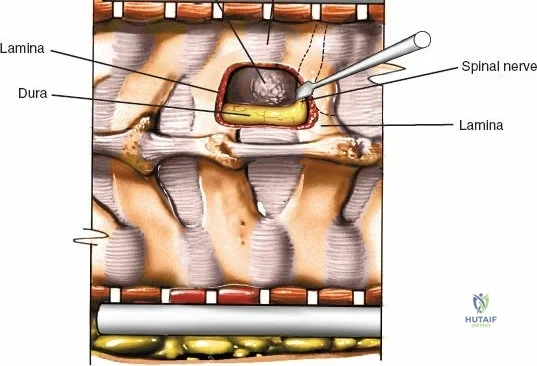
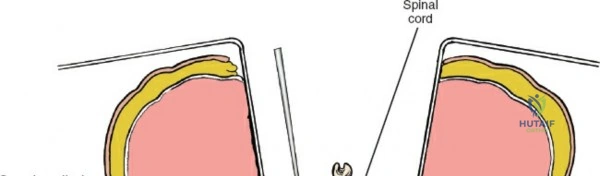
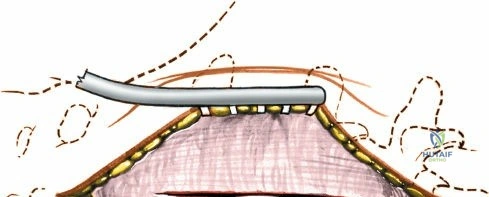
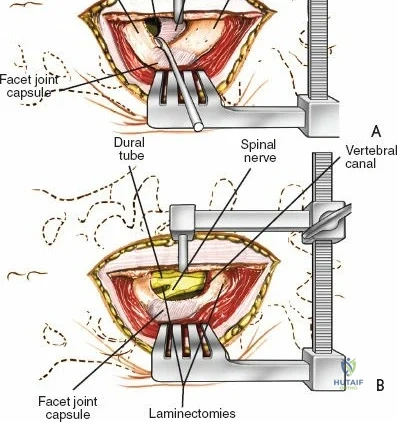
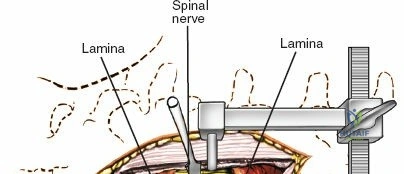
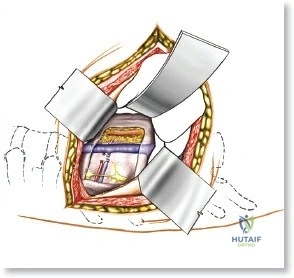
Once the bony anatomy is exposed, self-retaining retractors (e.g., Cerebellar, Gelpi, or Taylor retractors) are placed. To perform a laminectomy and decompression, begin by removing the spinous processes at the involved levels using a large Leksell rongeur. Next, address the ligamentum flavum. Insert a blunt dissector or a small curette under the cut edge of the ligamentum flavum to separate it from the underlying dura. Use a Kerrison rongeur to remove the distal end of the lamina and the ligamentum flavum at its attachment to the undersurface of the lamina.
Immediately beneath the ligamentum flavum and epidural fat is the thecal sac, identifiable by its distinct blue-white appearance. The epidural venous plexus overlies the dura and nerve roots. If these veins are injured, control bleeding with bipolar electrocautery (Malis) set to a low wattage, or by applying hemostatic agents such as Gelfoam soaked in thrombin, FloSeal, or cottonoid patties. Never use monopolar cautery near the neural elements.
To gain better exposure of the dura, nerve root, and disc, remove additional portions of the lamina, both from the leading edge of the lamina below and from the caudal edge of the lamina above. A medial facetectomy may be required to decompress the lateral recess. Remember the surgical axiom: it is always safer to remove more bone than to retract nerve roots or the dura excessively. Retract the dural sac and nerve root medially using a Penfield 4 or a specialized nerve root retractor to reveal the posterior aspect of the intervertebral disc.
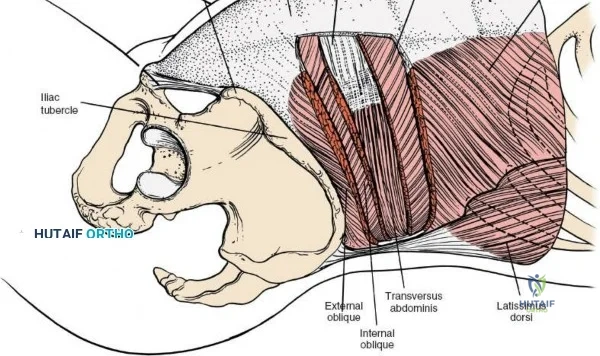
To extend the approach for a posterolateral fusion, carry the dissection as far laterally as possible, sweeping the muscle off the facet joints and onto the transverse processes. Complete lateral dissection exposes the transverse processes, providing the necessary vascularized bony bed for decortication and placement of bone graft.
Minimally Invasive Posterior Approach
Improved imaging techniques and the advent of tubular retractor systems have allowed surgeons to accurately localize pathology and perform decompressions with minimal tissue disruption. The main indications for the minimally invasive surgery (MIS) approach are excision of an intervertebral disc (microdiscectomy) and targeted decompression of a spinal nerve root.
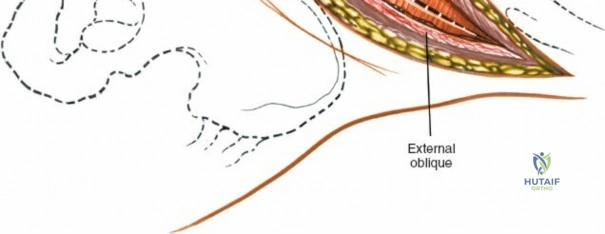
Place the patient in the prone position on a radiolucent table. Palpate the spinous processes to identify the midline. Use biplanar fluoroscopy to determine the exact disc level and the optimal trajectory. Make a 2 to 3-cm longitudinal incision approximately 1.5 to 2 cm lateral to the midline at the level of the targeted disc.
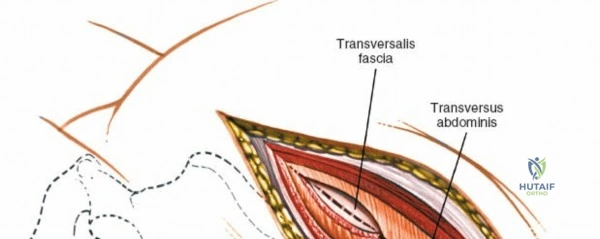
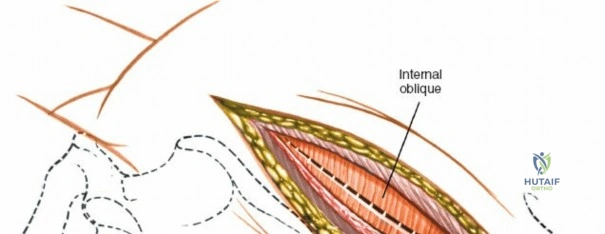
Deepen the approach through the subcutaneous adipose tissue and incise the fascia covering the erector spinae muscle. The MIS approach relies on an internervous plane; it bluntly splits the fibers of the erector spinae muscle group (which are innervated segmentally), ensuring no significant denervation or ischemic necrosis occurs.
Two alternative techniques are available for deep dissection:
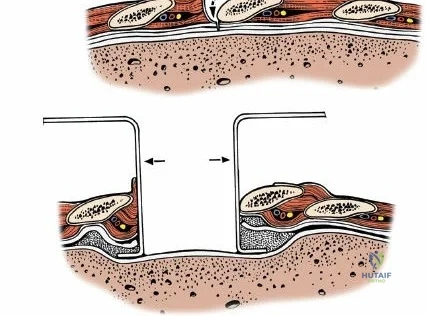
1. Transmuscular Technique: Sequential dilating tubes are passed through the muscle fascia down to the bony lamina. Remove all but the largest of the dilating tubes. Staying within this working channel, resect the distal lamina and ligamentum flavum on the affected side to expose the nerve root. This is performed using a high-speed burr, curettes, and Kerrison rongeurs. The use of an operating microscope or high-definition exoscope is mandatory for illumination and magnification.
2. Subperiosteal Technique: A small tubular retractor is docked directly onto the spinous process base, and the muscle is elevated subperiosteally in a limited fashion, mimicking a unilateral open approach but through a micro-incision.
Surgical Hazard (The "Dang" Factor): Meticulous positioning of the tubular retractors must be confirmed with fluoroscopy. Because the incision is incredibly small and visual landmarks are obscured, any deviation in trajectory may lead the surgeon to the wrong anatomical level or cause inadvertent injury to the facet joint or pars interarticularis. If the surgeon is disoriented, the tube must be removed, and the trajectory re-established with fluoroscopy.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, posterior lumbar surgery carries inherent risks. The surgeon must be adept at recognizing and managing these intraoperative and postoperative complications immediately.
Intraoperative Complications
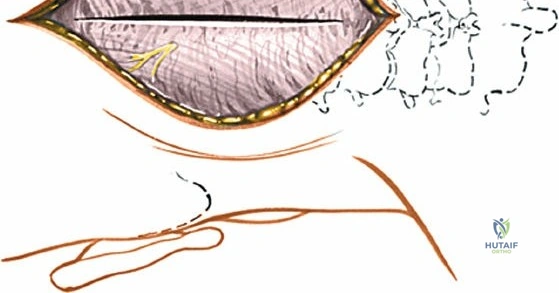
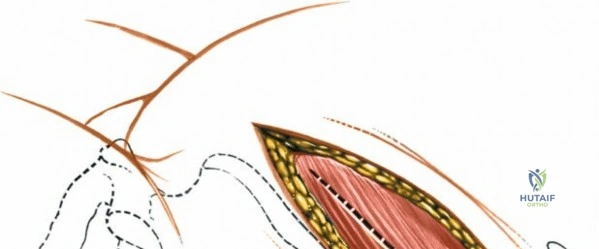
Incidental Durotomy (Dural Tear): This is the most common intraoperative complication, occurring in 3% to 14% of primary lumbar surgeries, and up to 20% in revision cases. It usually occurs during the resection of an adherent ligamentum flavum or massive disc herniation. When a tear occurs, clear fluid (CSF) will pool in the field. The defect must be primarily repaired using 4-0 or 5-0 non-absorbable suture (e.g., Prolene or Nurolon) in a running or interrupted fashion. A Valsalva maneuver should be performed by anesthesia to confirm a watertight seal. Fibrin glue and muscle/fat patches can be used to augment the repair. Postoperatively, the patient may need to remain flat in bed for 24-48 hours to prevent positional headaches and pseudomeningocele formation.
Nerve Root Injury: Direct mechanical injury from aggressive retraction, or thermal injury from electrocautery, can lead to permanent radiculopathy or motor deficits (e.g., foot drop). The nerve root must be handled with the utmost respect, utilizing bipolar cautery only, and limiting retraction time.
Vascular Injury: While epidural venous bleeding is common and manageable, catastrophic vascular injury can occur if instruments (such as pituitary rongeurs) are advanced too far anteriorly through the anterior annulus of the disc space, violating the aorta, inferior vena cava, or iliac vessels. This presents as sudden, profound hypotension and requires immediate packing, emergent laparotomy, and vascular surgery consultation.
Postoperative Complications
Epidural Hematoma: A rare but devastating complication (0.1% - 0.2%). Patients present with severe, progressive back pain, escalating neurologic deficits, and bowel/bladder dysfunction in the immediate postoperative period. This is a surgical emergency requiring immediate return to the operating room for evacuation to prevent permanent cauda equina syndrome.
Surgical Site Infection (SSI): Superficial and deep infections occur in 1% to 5% of cases. Risk factors include obesity, diabetes, smoking, and prolonged operative times. Deep infections require aggressive surgical debridement, copious irrigation, and prolonged culture-directed intravenous antibiotics.
| Complication | Estimated Incidence | Prevention / Salvage Strategy |
|---|---|---|
| Incidental Durotomy | 3% - 14% | Careful Kerrison use / Primary suture repair, fibrin glue, bedrest |
| Surgical Site Infection | 1% - 5% | Pre-op antibiotics, intrawound Vancomycin powder / I&D, IV antibiotics |
| Epidural Hematoma | 0.1% - 0.2% | Meticulous hemostasis, drain placement / Emergent surgical evacuation |
| Wrong-Level Surgery | < 0.1% | Mandatory intraoperative fluoroscopic localization / Revision surgery |
| Postoperative Visual Loss | < 0.01% | Avoid ocular pressure, keep head neutral/elevated / Emergent ophthalmology consult |
Phased Post-Operative Rehabilitation Protocols
The success of a posterior lumbar procedure is heavily reliant on a structured, phased rehabilitation protocol. The goals are to protect the surgical site, prevent neural tethering, and restore dynamic core stability.
Acute Inpatient Phase (Weeks 0-2)
Immediately postoperatively, the focus is on pain control, mobilization, and protecting the neural decompression or fusion construct. Patients are taught "log-rolling" techniques to get out of bed, ensuring the shoulders and pelvis move as a single unit to avoid twisting the lumbar spine. Sitting is generally limited to 30-45 minute intervals to prevent excessive axial loading.
For patients who underwent simple microdiscectomy or laminectomy, early ambulation is encouraged on postoperative day zero. Physical therapy focuses on basic activities of daily living (ADLs) and safe ambulation. For fusion patients, a rigid lumbar orthosis (LSO) may be prescribed depending on the surgeon's preference and the bone quality of the patient, though modern rigid pedicle screw constructs have made bracing less mandatory.
Outpatient Rehabilitation Phase (Weeks 2-12+)
At the two-week mark, surgical incisions are evaluated for healing, and sutures/staples are removed.
Weeks 2-6: Patients begin a formal outpatient physical therapy program. The primary focus is on neural mobilization (nerve glides) to prevent epidural fibrosis and tethering of the nerve roots. Gentle isometric core stabilization exercises (transversus abdominis bracing) are initiated. Heavy lifting (greater than 10 lbs), extreme flexion, and twisting are strictly prohibited.
Weeks 6-12: For decompression-only patients, range of motion exercises are advanced. For fusion patients, radiographic evaluation is performed at 6 weeks to assess hardware integrity and early graft incorporation. If stable, physical therapy is escalated to include dynamic core strengthening, closed-kinetic chain lower extremity exercises, and cardiovascular conditioning (stationary bike, aquatic therapy).
Months 3-6: Patients are gradually transitioned into work-specific hardening programs and sports-specific training. Return to heavy manual labor or contact sports is generally delayed until 6 months postoperatively for fusions, pending radiographic evidence of solid arthrodesis.
Summary of Landmark Literature and Clinical Guidelines
The surgical approach and indications for lumbar spine pathology are heavily supported by decades of rigorous academic literature. The most influential of these is the Spine Patient Outcomes Research Trial (SPORT).
The SPORT trials were massive, multicenter randomized and observational cohorts evaluating surgical versus non-operative treatment for three distinct pathologies: lumbar disc herniation, spinal stenosis, and degenerative spondylolisthesis.
1. For Lumbar Disc Herniation, SPORT demonstrated that patients treated with surgical microdiscectomy experienced significantly faster and greater relief of radicular pain compared to those treated conservatively, with benefits maintained at 8-year follow-up.
2. For Spinal Stenosis, surgical decompression (laminectomy) provided significantly greater improvement in pain and function compared to non-operative care.
3. For Degenerative Spondylolisthesis, the addition of a posterior instrumented fusion to the decompression yielded vastly superior clinical outcomes compared to decompression alone or conservative management, cementing the role of fusion in the setting of instability.
Furthermore, guidelines published by the North American Spine Society (NASS) strongly recommend the use of intraoperative fluoroscopy for precise localization to eliminate wrong-level surgery. Recent literature also strongly supports the use of intrawound Vancomycin powder during closure of posterior lumbar approaches, which has been shown in multiple meta-analyses to significantly reduce the incidence of deep surgical site infections without adverse systemic effects. Mastery of the posterior approach, combined with adherence to these evidence-based guidelines,
Clinical & Radiographic Imaging Archive