MCP & PIP Joint Surface Replacement Arthroplasty: An Intraoperative Masterclass
Key Takeaway
Master the complexities of MCP and PIP joint surface replacement arthroplasty. This intraoperative guide covers detailed anatomy, meticulous surgical execution, critical pearls and pitfalls, and comprehensive postoperative care for fellows. Gain insights into managing rheumatoid and degenerative arthritis deformities with advanced implant techniques, ensuring optimal outcomes for patients suffering from debilitating hand conditions.
Welcome, fellows and colleagues, to the operating theater and to this definitive masterclass. Today, we are tackling a challenging yet incredibly rewarding procedure: surface replacement arthroplasty (SRA) of the metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joints. This is a crucial, high-level skill for restoring function, preserving kinematics, and alleviating pain in patients with advanced arthritis, particularly those affected by rheumatoid disease or severe degenerative changes.
Comprehensive Introduction and Patho-Epidemiology
Before we make any incisions or even lift a scalpel, it is imperative that we thoroughly understand the underlying pathology dictating our surgical interventions. The paradigm of small joint arthroplasty has shifted significantly over the last few decades. While silicone interpositional arthroplasty remains a reliable workhorse for low-demand, severely deformed rheumatoid hands, surface replacement arthroplasty utilizing metal, ultra-high-molecular-weight polyethylene (UHMWPE), or pyrocarbon has emerged as the premier choice for preserving true joint kinematics and bone stock in appropriately selected patients.
Pathogenesis of Rheumatoid Arthritis
Rheumatoid arthritis (RA) is a systemic, autoimmune disorder that frequently targets the appendicular skeleton, with a profound predilection for the hands and wrists, leading to debilitating, progressive deformities. It is a multifactorial entity that, while still not entirely understood at the root cause level, involves a catastrophic autoimmune response. This response is potentially triggered by environmental factors, bacterial or viral infections, acting upon a host with a specific hereditary predisposition, notably the HLA-DR4 and HLA-DR1 genotypes.
At a cellular and molecular level, the pathophysiology is driven by an aggressive infiltration of the synovial lining by B and T lymphocytes, alongside activated macrophages. This inflammatory cascade leads to the proliferation and hypertrophy of synoviocytes—a process culminating in the formation of pannus. This hyperplastic synovial tissue is far from benign; it acts as a locally invasive tumor. The macrophages and fibroblasts within the pannus release a barrage of destructive enzymes, including matrix metalloproteinases (MMPs), alongside pro-inflammatory cytokines such as Tumor Necrosis Factor-alpha (TNF-α) and Interleukin-6 (IL-6).
The result of this enzymatic and cytokine storm is the systematic destruction of articular cartilage, the induction of osteoclastogenesis via the RANKL pathway leading to periarticular bony erosions, and the severe attenuation of critical stabilizing ligaments. Furthermore, the tenosynovium surrounding the extrinsic flexor and extensor tendons becomes similarly inflamed, leading to tendon attrition, rupture, and profound biomechanical imbalances. The primary target in the hand for RA is most frequently the MCP joints, where it manifests early as palpable tenosynovitis and boggy inflammation of the synovial recesses.
Pathogenesis of Degenerative Arthritis
In stark contrast to the systemic inflammatory storm of RA, degenerative arthritis, or osteoarthritis (OA), more commonly targets the PIP and distal interphalangeal (DIP) joints of the hand. This is a progressive process of irreversible articular cartilage wear, which can occur secondary to intra-articular trauma, crystalline arthropathy, previous infection, or as an idiopathic, genetically linked condition (such as primary generalized osteoarthritis).
The underlying mechanism of OA involves an initial fibrillation of the superficial chondrocyte layer, followed by a deeper fissuring of the cartilage matrix. As the cartilage fails, the underlying subchondral bone experiences increased mechanical stress, leading to sclerosis, stiffening, and the formation of subchondral cysts. Concurrently, the joint attempts to stabilize itself through periarticular new bone formation, resulting in the characteristic osteophytes (Bouchard's nodes at the PIP joints).
Ultimately, this cycle leads to severely restricted joint motion, angular deformities, and significant mechanical pain. While less common, primary OA can affect the MCP joints, though when encountered, the surgeon must maintain a high index of suspicion for secondary causes such as previous trauma, occult infection, hemochromatosis, or osteonecrosis (Dieterich's disease).
Detailed Surgical Anatomy and Biomechanics
A profound mastery of hand biomechanics and three-dimensional surgical anatomy is the absolute prerequisite for successful surface replacement arthroplasty. The MCP and PIP joints are not simple hinges; they are complex, multi-axial articulations governed by a delicate balance of intrinsic and extrinsic forces.
The Metacarpophalangeal Joint Kinematics
The MCP joint is a condyloid joint, allowing for flexion, extension, abduction, adduction, and a small degree of circumduction. The metacarpal head is not perfectly spherical; it is cam-shaped, being broader volarly than dorsally. This cam effect is critical: in extension, the collateral ligaments are relatively lax, allowing for abduction and adduction. As the joint flexes, the cam shape and the eccentric origin of the collateral ligaments cause them to tighten, providing crucial lateral stability during grip.
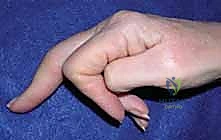
In the rheumatoid patient, the MCP joints are particularly susceptible to characteristic, devastating deformities. The classic presentation includes ulnar deviation of the digits coupled with volar subluxation or frank dislocation of the proximal phalanx on the metacarpal head.

These deformities are a direct consequence of synovial proliferation within the recesses of the collateral ligaments and the metacarpal head. This pannus physically distends and enzymatically weakens these critical stabilizing ligaments, particularly the radial collateral ligament, which is inherently thinner than its ulnar counterpart.
Furthermore, radial inclination of the metacarpals, often coupled with wrist joint destruction and ulnar translation of the entire carpus, exacerbates the problem. This proximal carpal shift alters the vector of the extrinsic tendons. The extensor tendons subluxate ulnarly and volarly into the valleys between the metacarpal heads, slipping off the apex of the joint. The combined effect of ulnar forces from the displaced extensor apparatus and volar forces from the flexors drives the fingers into an inescapable cycle of ulnar drift and fixed MCP flexion deformities.

The Proximal Interphalangeal Joint Kinematics
The PIP joint functions primarily as a hinge joint (ginglymus), boasting a normal arc of motion from 0 to 110 degrees. Its stability is derived from the congruent articular surfaces, the robust collateral ligaments (proper and accessory), and the thick, fibrocartilaginous volar plate. The extensor mechanism here is highly complex, splitting into a central slip that inserts on the middle phalanx base, and lateral bands that bypass the joint to form the terminal tendon.
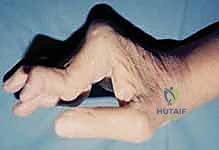
For the PIP joints, we frequently encounter two specific, complex deformities that dictate our surgical approach. The first is the swan-neck deformity, characterized by PIP joint hyperextension, dorsal subluxation of the lateral bands, and compensatory flexion of the DIP joint. This cascade is typically initiated by the failure of volar plate integrity or intrinsic muscle tightness, frequently seen in the rheumatoid population.
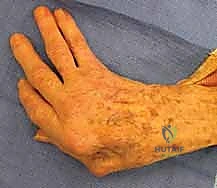
The second major deformity is the boutonnière deformity. This is caused by the attenuation or frank rupture of the central slip mechanism at its insertion on the middle phalanx. Consequently, the lateral bands subluxate volarly past the axis of rotation of the PIP joint, transforming from extensors to flexors of the PIP. This leads to fixed PIP joint flexion and secondary DIP joint hyperextension.
Neurovascular and Muscular Intervals
When we approach these joints surgically, we must be acutely aware of the delicate neurovascular structures that lie perilously close to our dissection planes. Dorsally, the skin is thin and mobile, housing the fragile dorsal venous network and the dorsal sensory branches of the radial and ulnar nerves. These must be meticulously identified and protected via gentle retraction; aggressive handling will lead to painful postoperative neuromas.
The muscular intervals are less of a primary concern directly at the MCP and PIP joints compared to the forearm, but we are constantly manipulating the extensor tendons, their intricate sagittal band attachments, and the intrinsic muscle insertions. The interossei and lumbricals contribute to the complex balance of the digits, and their meticulous release or repair is often the deciding factor between a beautifully balanced arthroplasty and a recurrent deformity.
Exhaustive Indications and Contraindications
Patient selection is the cornerstone of successful small joint surface replacement arthroplasty. Unlike large joint arthroplasty, where implants can often compensate for massive soft tissue defects, the unconstrained or semi-constrained nature of MCP and PIP surface replacements demands adequate bone stock and functional, reconstructable ligamentous support.
The primary, overriding indication for surface replacement arthroplasty of the MCP or PIP joints is severe, recalcitrant pain relief. Correction of deformity, restoration of a functional arc of motion, and improvement in aesthetic appearance are secondary, albeit highly desirable, considerations. We must recognize that a profoundly deformed hand might be entirely painless and functionally adapted for one patient, whereas a mild radiographic deformity might be exquisitely painful and disabling for another.
We must examine the entire upper extremity in a holistic manner. A fundamental rule of hand surgery is that proximal instability dictates distal failure. Deficits of the shoulder, elbow, and most importantly, the wrist, must ideally be addressed before or concurrently with MCP conditions. Specifically, uncorrected radiocarpal instability or ulnar translation of the carpus will invariably lead to the recurrence of ulnar drift after MCP arthroplasty, rendering our meticulous joint replacement futile.
| Category | Metacarpophalangeal (MCP) Joint | Proximal Interphalangeal (PIP) Joint |
|---|---|---|
| Primary Indications | Severe osteoarthritic or rheumatoid pain failing conservative management; Preserved bone stock; Intact or reconstructable collateral ligaments. | Painful OA or post-traumatic arthritis; Intact central slip; Functional flexor tendons; Adequate bone stock. |
| Absolute Contraindications | Active acute or chronic infection; Inadequate bone stock precluding implant seating; Grade 3 ligamentous instability (requires constrained/silicone option); Absent extensor mechanism. | Active infection; Disrupted central slip (absolute for standard SRA); Absent flexor tendon function; Severe coronal plane instability. |
| Relative Contraindications | Severe, active, poorly controlled rheumatoid systemic disease; Heavy manual labor requirements; Concomitant uncorrected wrist instability. | Fixed, rigid swan-neck or boutonnière deformities (high risk of stiffness); Active RA (silicone often preferred); Heavy manual labor. |
Careful examination of the flexor and extensor tendons is crucial during the preoperative assessment. The extensor digiti quinti minimi (EDQM), extensor pollicis longus (EPL), and flexor pollicis longus (FPL) are highly prone to attrition ruptures in active RA due to prominent ulnar head or scaphoid osteophytes. Any extensor or flexor tendon ruptures must be definitively addressed prior to, or in conjunction with, implant arthroplasty.
Pre-Operative Planning, Templating, and Patient Positioning
Preoperative planning is not merely a suggestion; it is the blueprint for surgical success. The margin for error in small joint arthroplasty is measured in millimeters. An oversized implant will cause joint stuffing, severe stiffness, and potential fracture, while an undersized implant will lead to subsidence, loosening, and instability.
Radiographic Evaluation and Templating
Standard preoperative imaging for MCP joints must include true posteroanterior (PA), true lateral, and oblique views of the hand. Brewerton views can offer additional, highly specific information, particularly for visualizing early collateral ligament avulsion fractures or occult erosions hidden in the metacarpal head recesses. For PIP joints, meticulously positioned PA and lateral views of the individual digits are mandatory.
We universally employ digital or physical sizing templates, typically accounting for a standard 3% to 5% parallax enlargement factor, for both MCP and PIP joint systems. This provides a crucial preoperative estimate of the required implant size and the necessary bone resection levels. We overlay these templates on our radiographs to assess the intramedullary canal diameter, the diaphyseal bow, and the quality of the subchondral bone.
In patients with severe RA, osteopenia is a major concern. If the medullary canals are excessively wide and the cortices paper-thin, a press-fit pyrocarbon or metal implant may subside, and the surgeon must be prepared to utilize a cemented component or pivot to a silicone interpositional arthroplasty.
Patient Positioning and Operating Room Setup
The patient is positioned supine on the operating table, brought close to the edge on the operative side. The arm is placed on a sturdy, radiolucent hand table, abducted to approximately 45 degrees, and externally rotated. The hand is pronated and secured to allow optimal, unencumbered access to the dorsal aspect of the digits.
A nonsterile pneumatic tourniquet is applied proximally on the brachium, over copious cast padding, to ensure a pristine, bloodless surgical field during the critical phases of bone resection and implant seating. While Wide Awake Local Anesthesia No Tourniquet (WALANT) is gaining traction in hand surgery, for multi-joint surface replacement, regional anesthesia (supraclavicular or axillary brachial plexus block) combined with a tourniquet remains the gold standard for optimal visualization.
The fluoroscopy C-arm is brought in from the head or foot of the table, positioned to allow immediate, effortless AP and lateral views of the operative digit without requiring the surgeon to reposition the patient or contort the hand. Real-time fluoroscopic feedback is non-negotiable for confirming guide pin placement, verifying bone cut angles, and ensuring the trial implants are perfectly seated flush against the resected cortical margins.
Step-by-Step Surgical Approach and Fixation Technique
Alright, fellows, let us scrub in. We have our patient perfectly positioned, prepped with chlorhexidine, and draped. The regional block is dense, and the tourniquet is inflated to 250 mmHg. We will proceed with a rigorous, systematic approach.
Metacarpophalangeal Joint Surgical Approach
For MCP surface replacement arthroplasty, the choice of incision is dictated by the number of joints involved. For a single joint, a dorsal longitudinal incision centered over the metacarpal head is preferred. When addressing all four fingers in a rheumatoid patient, a dorsal transverse incision situated just proximal to the metacarpal heads provides excellent, expansile exposure while minimizing the risk of longitudinal scar contracture.
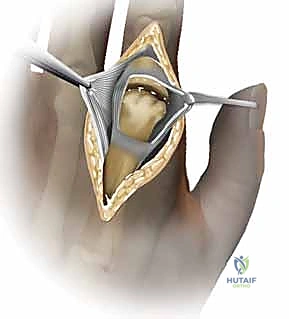
Upon incising the skin, meticulous hemostasis is achieved using bipolar electrocautery. We carefully elevate thick skin flaps, identifying and protecting the longitudinal dorsal sensory veins and nerves. The extensor apparatus is exposed. In the rheumatoid hand with ulnar drift, the extensor tendon is typically subluxated ulnarly. We perform a longitudinal incision through the radial sagittal band to release the extensor hood, allowing us to centralize the tendon later.
A dorsal longitudinal or transverse capsulotomy is performed, exposing the destroyed metacarpal head. A thorough synovectomy is mandatory; every remnant of invasive pannus must be excised using a rongeur and sharp dissection to prevent postoperative inflammatory flares. We must meticulously preserve the collateral ligaments, carefully releasing them from their metacarpal origins only as much as necessary to dislocate the joint and gain access to the articular surfaces.
Bone Preparation and Implant Trialing
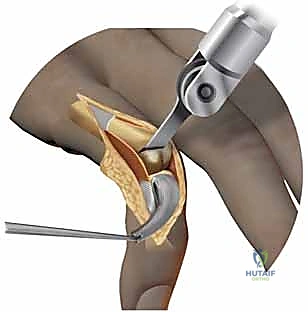
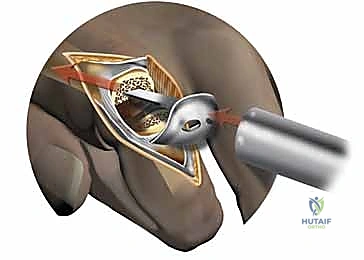
Using the system-specific intramedullary awl, we locate the anatomical axis of the metacarpal canal. A guide pin is inserted, and its position is verified on orthogonal fluoroscopic views. The metacarpal cutting block is slid over the pin, and an oscillating saw is used to resect the metacarpal head perpendicular to the long axis of the bone, removing minimal bone stock—typically just enough to accommodate the implant thickness.
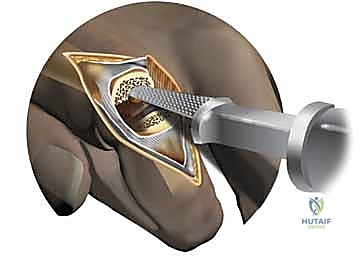
Attention is then turned to the proximal phalanx. The base is exposed, and peripheral osteophytes are meticulously removed to reveal the true anatomical footprint. The medullary canal is broached sequentially until cortical chatter is felt, indicating a secure, rotationally stable fit. The metacarpal canal is similarly prepared.
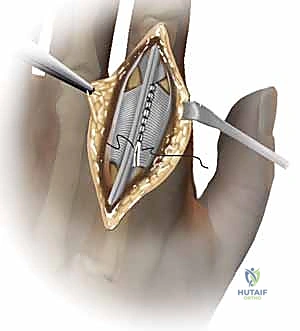
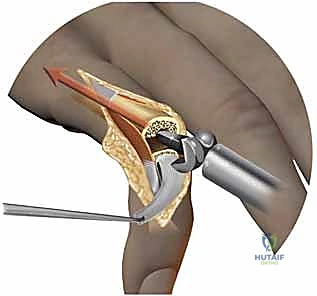
Trial components are inserted. This is the critical juncture where we assess tension, tracking, and stability. The joint should easily achieve 0 to 90 degrees of motion without the implants pistoning or impinging. If the joint is too tight, further minimal bone resection or soft tissue releases (such as an ulnar intrinsic release or fractional lengthening of the volar plate) may be required.
Final Implantation and Soft Tissue Balancing
Once the trialing is perfect, the definitive implants are opened. If using a press-fit pyrocarbon or uncemented metal system, the implants are gently impacted into place, ensuring they sit flush against the prepared bony surfaces. If the bone quality is poor, we will utilize highly viscous polymethylmethacrylate (PMMA) bone cement, injected via a syringe into the dried, pulsatile-lavaged canals, before seating the implants.
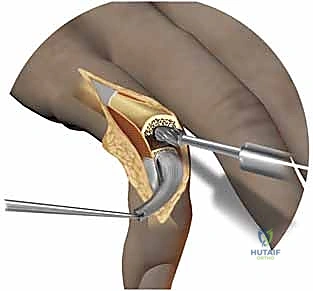
The most crucial step follows: soft tissue balancing. The joint has been replaced, but the deforming forces remain. We must reef the attenuated radial collateral ligament using non-absorbable sutures to restore lateral stability. The extensor tendon, previously subluxated ulnarly, is centralized over the new joint. This is achieved by imbricating the radial sagittal band and, if necessary, releasing the ulnar sagittal band.

The capsule is closed meticulously, followed by the skin. A bulky, non-adherent compressive dressing is applied, and the hand is placed in a volar resting splint in the intrinsic-plus position (MCPs flexed to 70 degrees, PIPs fully extended) before the tourniquet is deflated.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, surface replacement arthroplasty of the small joints carries a distinct complication profile. The unconstrained nature of these implants makes them unforgiving of soft tissue imbalance or poor bone preparation. Recognizing and rapidly managing these complications is the hallmark of a master surgeon.
Subsidence and aseptic loosening are the most common long-term radiographic complications, particularly with pyrocarbon implants in osteopenic bone. While many instances of radiographic subsidence are asymptomatic, progressive loosening accompanied by pain and loss of motion necessitates intervention. Infection, while relatively rare (1-3%), is devastating and typically requires immediate implant removal, aggressive debridement, and placement of an antibiotic cement spacer.
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage / Management Strategy |
|---|---|---|---|
| Aseptic Loosening / Subsidence | 10% - 25% (Long-term) | Poor bone stock, undersized implant, excessive early loading. | Observation if asymptomatic. Revision SRA with cement, or conversion to silicone arthroplasty. |
| Joint Stiffness / Contracture | 15% - 30% (Especially PIP) | Inadequate physical therapy, excessive scar formation, oversized implant. | Aggressive hand therapy, dynamic splinting. Surgical tenolysis/capsulotomy rarely indicated. |
| Recurrent Deformity (Ulnar Drift) | 5% - 15% | Failure to correct proximal wrist pathology, inadequate soft tissue balancing. | Soft tissue reconstruction, revision arthroplasty, or joint arthrodesis. |
| Deep Infection | 1% - 3% | Immunosuppression (RA meds), prolonged surgical time, hematoma. | Urgent I&D, implant removal, antibiotic spacer. Delayed arthrodesis or silicone spacer. |
| Periprosthetic Fracture | < 5% | Aggressive broaching, severe osteopenia, trauma. | Non-operative if stable. Internal fixation or revision with long-stemmed implants if unstable. |
Stiffness is the bane of PIP joint arthroplasty. The intricate extensor mechanism and the tendency for robust scar formation in the narrow digital confines frequently lead to a loss of extreme flexion or extension. Postoperative extensor lag is common if the central slip was attenuated or inadequately repaired during a dorsal approach.
When an SRA fails catastrophically due to infection, massive bone loss, or irreparable soft tissue destruction, salvage options are limited but critical. Conversion to a silicone interpositional arthroplasty is often feasible if sufficient diaphyseal bone remains. If the bone loss is massive or the joint is flail, a definitive arthrodesis (fusion) using a tension band construct or intramedullary compression screw provides a stable, painless, albeit immobile, digit.
Phased Post-Operative Rehabilitation Protocols
Surgery is only fifty percent of the battle; the remaining fifty percent relies entirely on an exquisite, rigidly controlled postoperative rehabilitation protocol administered by a certified hand therapist. The protocol must delicately balance the need for early motion to prevent scar adhesion with the necessity of protecting the repaired soft tissues and allowing implant osteointegration.
Phase I: Tissue Protection and Early Motion (Days 3 to 14)
The bulky surgical dressing is removed within 3 to 5 days postoperatively. The hand is assessed for viability, edema, and early signs of infection. A custom thermoplastic splint is fabricated immediately. For MCP arthroplasty, a dynamic extension splint with outriggers is often utilized to support the centralized extensor tendons while allowing active flexion and passive extension.
Edema control is paramount, utilizing compressive wrapping and elevation. Active and active-assisted range of motion exercises are initiated within the safe zones determined intraoperatively. The goal in this phase is to achieve 30 to 40 degrees of motion while preventing any lateral or rotational stress on the healing collateral ligaments.
Phase II: Progressive Mobilization (Weeks 2 to 6)
Sutures are removed at approximately 14 days. The splinting regimen is gradually modified. For PIP joints, buddy taping may be initiated to encourage motion while providing lateral support from the adjacent digit. If an extensor lag develops, static progressive extension splinting is instituted at night.
Patients are encouraged to perform tendon gliding exercises to prevent adhesions between the flexor and extensor mechanisms. Gentle passive overpressure may be applied by the therapist to overcome early capsular tightness, but aggressive manipulation is strictly avoided to prevent implant loosening or soft tissue rupture.
Phase III: Strengthening and Functional Return (Weeks 6 to 12+)
By six weeks, the soft tissues have achieved significant tensile strength, and early osteointegration (for uncemented implants) is underway. Splints are typically discontinued during the day, though nighttime resting splints may be continued if deformities threaten to recur.
Progressive strengthening using putty, hand grippers, and functional task simulation is initiated. Patients are counseled that maximal medical improvement following small joint arthroplasty often takes 6 to 12 months. They must understand that while pain relief is usually profound and immediate, the final arc of motion will likely be functional but not entirely normal, and heavy impact loading or repetitive sheer stresses must be avoided for the lifetime of the implant.
Summary of Landmark Literature and Clinical Guidelines
The evolution of small joint arthroplasty is deeply rooted in decades of rigorous clinical research. To truly master this field, one must be familiar with the foundational literature that dictates our current evidence-based practices.
Alfred Swanson's pioneering work in the 1960s and 70s introduced the concept of the flexible silicone hinge, revolutionizing the treatment of the rheumatoid hand. His landmark publications established the baseline against which all modern arthroplasties are measured, proving that resection arthroplasty combined with an interpositional spacer could reliably relieve pain and correct deformity, albeit with an expected loss of motion over time and the risk of silicone synovitis.
The transition to surface replacement was championed by Linscheid and Dobyns, who recognized the biomechanical limitations of silicone and sought to restore true joint kinematics. Modern systematic reviews, such as those by Chung et al., have critically compared silicone and surface replacement arthroplasties. These studies generally conclude that while SRA provides superior early postoperative grip strength and a more anatomical arc of motion, silicone remains non-inferior for long-term pain relief in severe RA, highlighting the critical importance of patient selection.
Recent literature focusing on pyrocarbon implants (e.g., the Ascension system) has demonstrated excellent biocompatibility and wear characteristics. However, long-term registries indicate a non-negligible rate of radiographic subsidence, emphasizing the absolute necessity of precise surgical technique, perfect sizing, and the avoidance of press-fit implants
