Thumb Carpometacarpal Joint Ligament Stabilization: An Operative Masterclass
Key Takeaway
Join us in the OR for a masterclass on thumb carpometacarpal (CMC) joint ligament stabilization. This guide provides an exhaustive, real-time breakdown of the flexor carpi radialis (FCR) reconstruction technique, detailing critical anatomical considerations, precise surgical execution, intraoperative pearls, and comprehensive postoperative management to restore thumb stability and function.
Comprehensive Introduction and Patho-Epidemiology
Welcome, fellows and colleagues, to the operating theater and to this definitive operative masterclass. Today, we are undertaking a rigorous examination of a common yet profoundly functionally debilitating condition: instability of the thumb carpometacarpal (CMC) joint. This masterclass is designed to guide you through the granular intricacies of its diagnosis, surgical planning, and the precise execution of a ligament reconstruction utilizing the flexor carpi radialis (FCR) tendon. Our overarching goal as orthopedic surgeons is not merely anatomical restoration, but the restitution of stability, the alleviation of pain, and the prevention of the inexorable progression to end-stage degenerative arthritis in these complex patients.
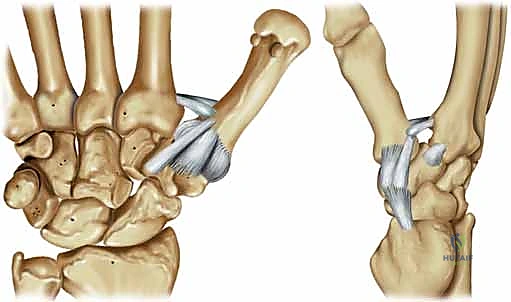
Thumb CMC joint instability typically arises from two primary pathophysiological mechanisms: inherent, often progressive, ligamentous laxity or an acute, high-energy traumatic event. Regardless of the inciting etiology, the fundamental biomechanical failure lies in the compromise of the critical stabilizing capsuloligamentous restraints surrounding this unique articulation. This structural compromise inevitably leads to abnormal joint kinematics, specifically manifesting as dorsoradial subluxation or frank dislocation of the first metacarpal base upon the articular surface of the trapezium. Left untreated, these abnormal shear forces precipitate rapid articular cartilage degradation.
The thumb CMC joint, by its very evolutionary design, is a marvel of biomechanics, offering the wide, multi-planar range of motion that is absolutely essential for human prehension, precise pinch, and forceful power grasp. It is morphologically a biconcave-convex, or saddle, joint. The distal articular surface of the trapezium is convex in the dorsopalmar direction and concave in the radioulnar direction, while the proximal articular surface of the first metacarpal base is reciprocally shaped. This saddle configuration, while enabling exquisite, multi-axial mobility, inherently sacrifices intrinsic osseous stability, rendering the joint profoundly reliant on its surrounding soft-tissue envelope.
Epidemiologically, idiopathic laxity is predominantly observed in middle-aged women, suggesting a multifactorial etiology involving hormonal influences, repetitive microtrauma, and genetic predispositions regarding collagen cross-linking. However, as astute clinicians, we must invariably maintain a high index of suspicion for systemic connective tissue disorders, such as Ehlers-Danlos or Marfan syndrome, which intrinsically predispose individuals to generalized ligamentous hypermobility. Conversely, in the setting of acute trauma, thumb CMC joint dislocations typically result from a massive axial load applied directly to a flexed thumb metacarpal. Historically and biomechanically, these dislocations are almost universally dorsoradial, accurately reflecting the catastrophic failure of the primary ligamentous restraints against this specific, high-stress vector of instability.
Detailed Surgical Anatomy and Biomechanics
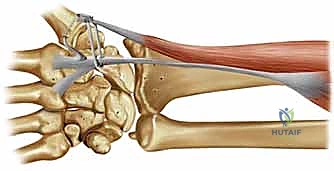
Let us review the critical regional anatomy, as a profound understanding of these structures dictates the safety and efficacy of every step in our surgical approach. The thumb CMC joint is stabilized by a highly complex, interwoven array of up to 16 distinct ligaments. However, for the specific purpose of preventing dorsoradial subluxation of the thumb metacarpal—our primary operative concern today—two specific ligamentous complexes are of paramount importance: the volar beak ligament and the dorsoradial ligament.
The Volar Beak and Dorsoradial Ligaments
The volar beak ligament, frequently referred to in historical literature as the deep anterior oblique ligament (dAOL), palmar ligament, or ulnar ligament, is a stout, intracapsular, and highly mechanoreceptive structure. It originates precisely from the volar central apex of the trapezium and inserts onto the prominent volar styloid process, or "beak," of the first metacarpal base. Biomechanically, it serves as the primary restraint against metacarpal dorsal translation and radial subluxation, particularly when the thumb is loaded in flexion and pronation during key pinch. It lies immediately deep to a broader, more superficial anterior oblique ligament (sAOL), which is situated directly beneath the protective bulk of the thenar musculature.
Conversely, the dorsoradial ligament (DRL) originates from the dorsoradial tubercle of the trapezium and inserts broadly onto the dorsal base of the thumb metacarpal. Anatomical studies consistently describe the DRL as the thickest, widest, shortest, and arguably the strongest of all the CMC ligaments. While historically the volar beak ligament was viewed as the sole critical stabilizer, contemporary biomechanical studies have elevated the DRL to equal or greater importance. It acts as a critical static stabilizer, vehemently resisting radial translation and pronation torque of the metacarpal. Its structural integrity is absolutely crucial for maintaining the intimate saddle congruity of the joint during the "screw-home" mechanism of opposition.
Neurovascular Risks and Muscular Intervals
Navigating the surgical approach requires meticulous respect for the regional neurovascular anatomy. The Superficial Radial Sensory Nerve (SRSN) and its terminal dorsal digital branches are highly vulnerable during the initial skin incision and subcutaneous dissection, particularly along the radial border of the thenar eminence. Iatrogenic injury to these branches can lead to devastating, recalcitrant neuromas or persistent dysesthesias that can eclipse the benefits of a successful ligament reconstruction. Furthermore, the radial artery lies in perilous proximity to the surgical field, traversing the anatomic snuffbox and diving between the two heads of the first dorsal interosseous muscle. Extreme caution is mandated during the deeper dissection around the FCR tendon, and specifically when drilling the metacarpal tunnel or driving K-wires.
The surgical approach involves careful, bloodless navigation around several critical muscle-tendon units. The thenar musculature—comprising the abductor pollicis brevis (APB), flexor pollicis brevis (FPB), and opponens pollicis (OP)—must be respected. We will incise the fascia and elevate the radial border of this muscle mass extraperiosteally to preserve the thenar motor branch of the median nerve. Dorsally, the abductor pollicis longus (APL), extensor pollicis brevis (EPB), and extensor pollicis longus (EPL) dictate our dorsal exposure. Finally, the Flexor Carpi Radialis (FCR) is our workhorse graft tendon. It runs through its own distinct fibro-osseous tunnel on the volar aspect of the wrist, inserting primarily into the volar base of the second metacarpal, with variable slips to the third metacarpal and trapezium.
Exhaustive Indications and Contraindications
Patient selection is the cornerstone of surgical success in thumb CMC ligament stabilization. The primary indication for this procedure is symptomatic, functionally limiting instability of the thumb CMC joint that has proven refractory to a comprehensive regimen of conservative management. This is most frequently observed in patients with Eaton-Littler Stage I or early Stage II disease. In Stage I, the articular contours are entirely normal, but there is radiographic evidence of joint space widening or subluxation on stress views. In early Stage II, there is minimal joint space narrowing and osteophyte formation is strictly limited to less than 2 millimeters. In these specific stages, the articular cartilage remains largely viable, and restoring the ligamentous tether can successfully halt the progression of degenerative joint disease.
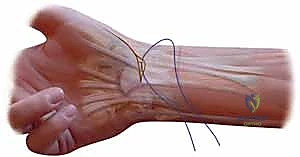
A secondary, yet equally critical, indication is the acute or subacute traumatic dislocation of the thumb CMC joint. While some purely ligamentous dislocations may be managed with closed reduction and percutaneous pinning, injuries that remain grossly unstable following reduction, or those presenting late (chronic dislocations), necessitate open ligamentous reconstruction. If the CMC joint remains unstable, the crucial biomechanical functions of key pinch and power grasp are severely compromised, leading to profound functional deficits. Open ligament reconstruction for unstable traumatic dislocations has consistently demonstrated superiority over closed reduction and pinning alone in preventing recurrent instability and subsequent post-traumatic arthrosis.
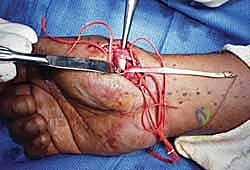
Contraindications must be strictly adhered to in order to prevent catastrophic clinical failures. The most absolute contraindication is advanced degenerative joint disease, specifically Eaton-Littler Stage III or IV. In these stages, the articular cartilage is irreparably destroyed, and significant osteophytosis or pantrapezial arthritis is present. Performing a soft-tissue ligament reconstruction in a profoundly arthritic joint will fail to relieve pain and will likely exacerbate symptoms due to increased joint contact pressures; these patients require a trapeziectomy with or without ligament reconstruction and tendon interposition (LRTI). Additional contraindications include active local or systemic infection, unmanageable systemic inflammatory arthropathies (such as advanced Rheumatoid Arthritis where the bone stock is severely osteopenic), and patient inability or unwillingness to comply with the rigorous postoperative rehabilitation protocol.
| Category | Indications | Contraindications |
|---|---|---|
| Disease Stage | Eaton-Littler Stage I (Normal cartilage, laxity) | Eaton-Littler Stage III (Significant joint space narrowing, cysts) |
| Disease Stage | Eaton-Littler Stage II (Osteophytes < 2mm) | Eaton-Littler Stage IV (Pantrapezial arthritis) |
| Trauma | Acute unstable dislocation failing closed reduction | Chronic dislocation with established severe secondary arthrosis |
| Trauma | Chronic symptomatic ligamentous laxity | Active local or systemic infection |
| Patient Factors | Refractory to splinting, NSAIDs, injections | Non-compliance, severe untreated systemic inflammatory disease |
Pre-Operative Planning, Templating, and Patient Positioning
Preoperative planning begins with an exhaustive clinical evaluation and meticulous radiographic analysis. A thorough history must document the exact nature of the symptoms, the impact on daily activities, and the failure of nonoperative treatments (including the duration of thumb spica splinting and the number of intra-articular corticosteroid injections). On physical examination, the Grind test (axial compression and circumduction) and the Ballottement test (dorsoradial translation of the metacarpal base) are paramount for confirming instability and assessing for crepitus. Crucially, the surgeon must evaluate the thumb metacarpophalangeal (MCP) joint for hyperextension laxity. If greater than 20 to 30 degrees of MCP hyperextension is present during lateral pinch, the deforming forces will doom the CMC reconstruction to failure; therefore, a concomitant MCP volar capsulodesis or arthrodesis must be planned.
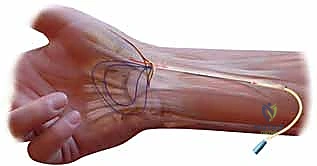
Radiographic evaluation requires standard AP, true lateral, and specialized oblique stress views. The True AP (Robert) view is obtained with the forearm in maximal pronation and the dorsum of the thumb resting flat on the imaging cassette, with the beam angled 15 degrees from distal to proximal. This projects the CMC joint clearly without overlap. The 30-degree oblique stress view is performed by having the patient press the radial sides of their thumb tips together forcefully. This maneuver accentuates the radial subluxation of the thumb metacarpal base, vividly demonstrating the degree of dynamic laxity. If any doubt exists regarding the integrity of the articular cartilage, preoperative MRI or even a diagnostic arthroscopy can be considered to definitively rule out advanced Eaton Stage III disease.
In the operating theater, precise patient positioning and equipment setup are critical for a seamless procedure. The patient is positioned supine with the operative arm extended onto a standard, rigid hand table. The main operating table is turned 90 degrees away from the anesthesia workstation. This specific configuration allows the primary surgeon and the first assistant to sit comfortably directly across from each other, providing optimal ergonomics and unhindered visualization of the surgical field. A pneumatic tourniquet is applied to the proximal brachium and inflated to 250 mmHg (or roughly 100 mmHg above the patient's systolic blood pressure) following exsanguination with an Esmarch bandage, ensuring the bloodless field required for safe dissection around the SRSN and radial artery.
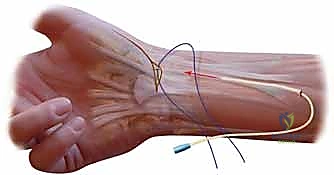
General anesthesia or a reliable regional block (such as an ultrasound-guided axillary or supraclavicular block) is utilized. The entire upper extremity is prepped with a standard chlorhexidine or povidone-iodine solution and draped in a sterile fashion. The surgical tray must be equipped with specialized hand instruments, including fine tenotomy scissors, Freer elevators, small Hohmann retractors, a wire driver, and an assortment of K-wires (typically 0.045-inch and 0.062-inch). A mini-C-arm fluoroscopy unit must be draped and available in the room prior to incision to confirm joint reduction, assess drill trajectories, and verify final hardware placement.
Step-by-Step Surgical Approach and Fixation Technique
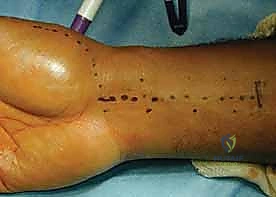
The surgical approach begins with a meticulously planned incision designed to maximize exposure while mitigating neurovascular risk. We utilize a modified Wagner incision, starting slightly dorsal to the abductor pollicis longus (APL) tendon at the base of the first metacarpal, curving volarly along the glabrous border of the thenar eminence, and extending proximally towards the distal wrist crease over the FCR tendon. Subcutaneous dissection is performed with extreme care using tenotomy scissors, spreading longitudinally to identify and protect the delicate, branching network of the superficial radial sensory nerve (SRSN). These branches are gently retracted dorsally with vessel loops.
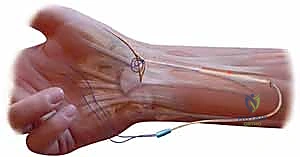
Once the superficial nerves are secured, we identify the fascia overlying the thenar musculature. The fascia is incised longitudinally, and the thenar muscles (APB and FPB) are sharply elevated extraperiosteally from the volar-radial aspect of the first metacarpal and the joint capsule. This extraperiosteal elevation is a critical maneuver to prevent inadvertent injury to the thenar motor branch of the median nerve. The dorsal flap is then developed by retracting the APL and EPB dorsally, exposing the underlying dorsal and radial joint capsule. A longitudinal or T-shaped capsulotomy is performed, allowing direct inspection of the articular surfaces of the trapezium and the metacarpal base. If advanced, full-thickness cartilage loss is unexpectedly encountered at this stage, the surgeon must be prepared to pivot the operative plan to a trapeziectomy.
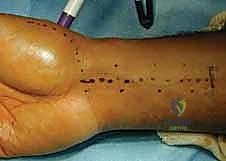
Assuming the cartilage is pristine or exhibits only minimal wear, we proceed with the FCR tendon harvest. The FCR is identified through the proximal extent of our primary incision, or via separate transverse step-ladder incisions in the volar forearm if a longer graft is required. We typically harvest the radial half of the FCR tendon, leaving the ulnar half intact to preserve native wrist flexion power, though a full-thickness harvest is acceptable in severe instability. The harvested slip is transected proximally at the musculotendinous junction and mobilized distally down to the crest of the trapezium, taking great care not to detach its ultimate insertion on the base of the second metacarpal.
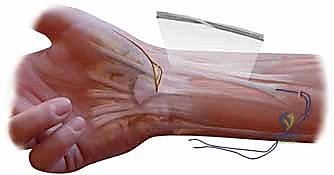
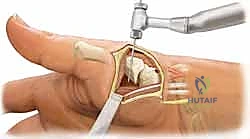
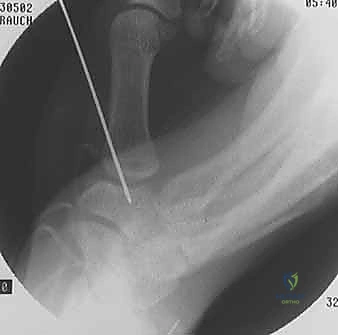
The next critical step is the creation of the osseous tunnel within the first metacarpal. Using fluoroscopic guidance, a drill hole (typically 3.2mm or 3.5mm, depending on graft thickness) is created. The trajectory is paramount: it must originate on the dorsal-radial aspect of the metacarpal base, at the anatomical insertion of the dorsoradial ligament, and exit precisely at the volar-ulnar beak of the metacarpal, replicating the insertion of the deep anterior oblique ligament. During drilling, the radial artery must be actively protected dorsally with a retractor or a Freer elevator.
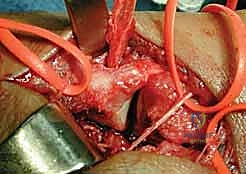
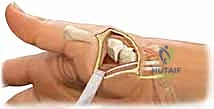
With the tunnel established, a wire loop or tendon passer is introduced from dorsal to volar. The mobilized FCR graft is passed deep to the APL tendon, threaded into the volar aperture of the metacarpal tunnel, and pulled through to exit dorsally. The thumb is then anatomically reduced into the saddle of the trapezium, placing it in palmar abduction and slight pronation to maximize congruity. This reduction is provisionally held by the assistant while a 0.045-inch K-wire is driven across the CMC joint from the metacarpal into the trapezium. Fluoroscopy confirms anatomic reduction and appropriate pin placement.
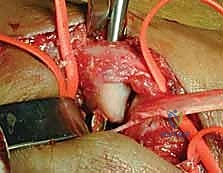
Finally, the graft is tensioned. With the joint pinned in anatomic reduction, the FCR graft is pulled taut dorsally. It is then sutured to the periosteum of the metacarpal base and the adjacent dorsal capsule using non-absorbable braided sutures (e.g., 2-0 Ethibond or FiberWire). To recreate the dorsal ligamentous complex, the remaining tail of the graft can be routed back volarly, passed deep to the APL, and sutured to the remaining capsule or the FCR insertion site, completing a robust, multi-planar tenodesis. The capsulotomy is closed, the skin is approximated with interrupted non-absorbable sutures, and a bulky, well-padded thumb spica splint is applied before tourniquet deflation.
Complications, Incidence Rates, and Salvage Management
While thumb CMC ligament reconstruction is highly successful in properly selected patients, it is a technically demanding procedure fraught with potential complications. Neurologic injury is arguably the most devastating soft-tissue complication. Injury to the Superficial Radial Sensory Nerve (SRSN) or the palmar cutaneous branch of the median nerve can result in a debilitating neuroma. The incidence of transient neurapraxia is reported to be between 5% and 10%, while permanent neuroma formation occurs in 1% to 2% of cases. Management of a confirmed neuroma requires surgical exploration, excision of the neuroma, and burying the proximal nerve stump into deep muscle belly or bone to mitigate mechanical irritation.
Hardware and structural complications are also notable risks. Iatrogenic fracture of the first metacarpal base during tunnel drilling or K-wire insertion can occur, particularly in osteopenic bone. If a fracture occurs, the reconstruction must be aborted or modified, and the fracture must be rigidly fixed, often requiring conversion to a temporary spanning external fixator or plate osteosynthesis. K-wire related complications, including pin tract infections, pin migration, or breakage, occur in approximately 5% to 8% of patients. Meticulous pin care and timely removal at 4 to 6 weeks minimize these risks. Deep surgical site infection is rare (less than 1%) but requires prompt operative debridement and targeted intravenous antibiotic therapy.
The most significant long-term complication is recurrent instability or the inexorable progression to symptomatic Eaton Stage III/IV osteoarthritis. Recurrent laxity can result from graft stretching, failure of tendon-to-bone healing, or technical errors in tunnel placement. Furthermore, if unrecognized diffuse articular wear was present at the time of index surgery, the altered joint kinematics post-reconstruction may accelerate cartilage degradation. When ligament reconstruction fails and the patient develops painful, end-stage arthrosis, salvage procedures are mandatory. The gold standard salvage operation is a trapeziectomy, typically combined with a Ligament Reconstruction and Tendon Interposition (LRTI) or suspensionplasty. Alternatively, in high-demand laborers, a CMC arthrodesis may be indicated to provide a stable, painless, albeit stiff, pillar for pinch.

| Complication | Estimated Incidence | Prevention Strategy | Salvage / Management |
|---|---|---|---|
| SRSN Neuroma | 1% - 2% | Meticulous blunt dissection, vessel loops | Excision and intra-muscular burying |
| Metacarpal Fracture | < 2% | Fluoroscopic guidance, sharp drill bits, avoid oversized tunnels | Internal fixation, conversion to trapeziectomy if unsalvageable |
| Pin Tract Infection | 5% - 8% | Sterile pin caps, regular cleaning, avoid tension on skin | Oral antibiotics, early pin removal if deep |
| Recurrent Instability | 5% - 10% | Robust graft tensioning, strict post-op immobilization | Revision stabilization or conversion to Trapeziectomy |
| Progression to OA | 10% - 15% (Long term) | Strict adherence to Eaton Stage I/II indications | Trapeziectomy with LRTI or CMC Arthrodesis |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation protocol is as critical to the ultimate success of the procedure
