Tibialis Anterior Tendon Reconstruction with EHL Transfer: An Intraoperative Masterclass
Key Takeaway
Join us in the OR for a detailed masterclass on tibialis anterior tendon reconstruction via extensor hallucis longus (EHL) transfer. This guide covers essential anatomy, preoperative planning, granular intraoperative steps, critical pearls and pitfalls for successful outcomes, and comprehensive postoperative management. Learn to meticulously address gait disturbances and restore ankle dorsiflexion, ensuring optimal patient recovery.
Comprehensive Introduction and Patho-Epidemiology
The Clinical Challenge of Anterior Compartment Failure
Fellows, welcome to the operating theater. Today, we are addressing a challenging yet profoundly impactful condition: the reconstruction of a ruptured tibialis anterior tendon. This procedure is crucial for restoring function, optimizing gait kinematics, and improving the quality of life in active patients suffering from the debilitating effects of foot drop. The tibialis anterior is the primary dorsiflexor of the ankle, responsible for over eighty percent of the dorsiflexion power required during the swing phase of the gait cycle. When this musculotendinous unit fails, the biomechanical cascade that follows is both predictable and disabling. Our patient today presents with a chronic tibialis anterior rupture, exhibiting the classic "slap-foot" gait. To restore this lost function, we will be performing an extensor hallucis longus (EHL) transfer to the medial cuneiform, a robust, highly reliable, and biomechanically sound technique that has become the gold standard for chronic, retracted ruptures.
Pathophysiology of Tibialis Anterior Rupture
Unlike Achilles tendon ruptures, which frequently occur in younger, highly active individuals during explosive athletic maneuvers, tibialis anterior tendon ruptures typically present in a slightly older demographic, most commonly men in their fifth to seventh decades of life. The pathophysiology is often rooted in chronic, insidious tendinosis rather than acute, high-energy trauma. Microvascular compromise at the critical zone of hypovascularity—located approximately two to three centimeters proximal to the tendon's insertion on the medial cuneiform—predisposes the tissue to mucoid degeneration and eventual mechanical failure. Patients may report a minor antecedent trauma, such as a misstep off a curb or a sudden forced plantarflexion event, but frequently, the rupture occurs spontaneously during normal ambulation. The underlying degenerative nature of the tendon dictates our surgical approach; direct end-to-end repair is rarely feasible in chronic cases due to poor tissue quality and significant proximal retraction of the muscle belly.
Diagnostic Nuances and Clinical Presentation
Before we even make an incision, we must comprehensively review the clinical picture, as misdiagnosis is alarmingly common. A tibialis anterior rupture primarily manifests as a profound gait disturbance. Upon observing the patient ambulating, you will immediately notice a slap-foot gait or a distinct foot drop. This acoustic and visual hallmark occurs because the ankle cannot adequately dorsiflex to clear the ground during the swing phase, nor can it eccentrically decelerate the foot after heel strike. To compensate, the patient will predictably adopt a steppage gait, hyperflexing the hip and knee to clear the toes and prevent tripping. On uneven surfaces or stairs, this biomechanical deficit is severely exacerbated, leading to a high risk of falls.
A critical diagnostic maneuver is the inability to heel-walk, which directly isolates and indicates tibialis anterior dysfunction. While acute ruptures may present with localized swelling, ecchymosis, and sharp pain over the anterior ankle, chronic ruptures—like the one we are treating today—often present with a painless foot drop. This lack of pain frequently leads to a significant delay in diagnosis, with patients often misdiagnosed as having an L5 radiculopathy or a compressive peroneal nerve palsy. Careful physical examination, noting the preservation of EHL and peroneal muscle function, is paramount in distinguishing a localized tendon rupture from a proximal neurologic lesion.
Detailed Surgical Anatomy and Biomechanics
The Tibialis Anterior Muscle-Tendon Unit
Let us orient ourselves with the essential surgical anatomy. The tibialis anterior muscle is a formidable structure, originating from the lateral tibial condyle, the proximal two-thirds of the lateral surface of the tibia, and the adjacent interosseous membrane. The musculotendinous junction typically occurs at the transition between the middle and distal thirds of the tibia. This is a critical landmark when assessing the degree of proximal retraction on preoperative imaging. The tendon then courses distally, enveloped within a distinct synovial sheath, passing deep to both the superior and inferior extensor retinacula of the ankle and foot. These retinacular structures function as crucial biomechanical pulleys, preventing bowstringing of the tendon during active dorsiflexion. The primary insertion of the tibialis anterior is dual-faceted: it anchors robustly onto the medial side of the medial cuneiform and the inferomedial base of the first metatarsal.
Extensor Hallucis Longus Donor Anatomy
The extensor hallucis longus (EHL) serves as our ideal donor graft for this reconstruction. Originating from the middle half of the anterior surface of the fibula and the adjacent interosseous membrane, the EHL muscle belly lies deep and slightly lateral to the tibialis anterior. Its tendon emerges and runs distally, passing deep to the extensor retinaculum, typically housed within its own synovial sheath. It continues across the dorsum of the foot to insert on the base of the distal phalanx of the great toe, providing extension to the hallux. The EHL is chosen for transfer because its vector of pull is nearly identical to that of the tibialis anterior, and its muscle belly possesses sufficient excursion and cross-sectional area to provide adequate dorsiflexion power. Furthermore, the functional deficit incurred by harvesting the EHL is minimal, provided we perform a meticulous tenodesis of the distal stump to the extensor hallucis brevis (EHB) to prevent a subsequent hallux malleus deformity.
Neurovascular Danger Zones of the Anteromedial Ankle
The anteromedial ankle is a high-traffic area for critical neurovascular structures, demanding meticulous dissection. The deep peroneal nerve is of paramount concern. It provides motor innervation to the muscles of the anterior compartment—the tibialis anterior, EHL, extensor digitorum longus (EDL), and peroneus tertius—and supplies sensory innervation to the first dorsal web space. In the distal leg, it travels between the tibialis anterior and the EHL. As it crosses the ankle joint, it typically lies deep to the extensor retinaculum, often coursing directly beneath or slightly lateral to the EHL tendon. Extreme caution is required during the dissection and routing of our tendon graft to avoid iatrogenic injury, which could catastrophically exacerbate the foot drop or result in a debilitating neuroma. Accompanying the deep peroneal nerve is the anterior tibial artery, which transitions into the dorsalis pedis artery as it crosses the ankle joint. This vessel provides crucial vascularity to the dorsum of the foot and must be protected with careful blunt and sharp dissection.
Biomechanical Considerations and Equinus Contracture
A critical biomechanical point, especially in subacute and chronic injuries, is the inevitable development of heel cord contractures. When the tibialis anterior ruptures, its primary antagonist—the triceps surae complex—is left unopposed. The gastrocnemius and soleus muscles consequently undergo adaptive shortening, leading to a progressive equinus deformity. Before proceeding with any anterior reconstruction, we must ensure the ankle can achieve at least 10 degrees of passive dorsiflexion with the knee extended. Failure to recognize and correct an equinus contracture will place excessive, unyielding tension on our EHL transfer. This biomechanical mismatch will inevitably lead to graft stretch-out, catastrophic failure of the fixation, and a persistent, unresolvable functional deficit. Therefore, a concomitant gastrocnemius-soleus recession or Achilles tendon lengthening is often a mandatory adjunct procedure.
Exhaustive Indications and Contraindications
Patient Selection Criteria
The decision to proceed with an EHL transfer for tibialis anterior reconstruction must be carefully weighed against the patient's physiological age, functional demands, and medical comorbidities. The ideal candidate is an active, community-ambulating individual with a chronic, retracted tibialis anterior rupture who has failed or refused conservative management (such as an Ankle-Foot Orthosis) and desires a return to a high level of function. The patient must have a supple, passively correctable ankle joint and a functional EHL muscle belly. Conversely, sedentary patients with low functional demands or those with severe medical comorbidities that preclude safe anesthesia are better served with permanent bracing.
Table: Indications and Contraindications
| Category | Specific Clinical Scenarios | Rationale / Considerations |
|---|---|---|
| Absolute Indications | Chronic, retracted TA rupture (>4-6 weeks) with functional deficit. | Direct repair is impossible due to tendon gap and poor tissue quality. EHL transfer restores active dorsiflexion. |
| Relative Indications | Acute TA rupture with massive tendinosis and poor tissue integrity. | Even if ends can be approximated, severely degenerated tissue will likely fail. Augmentation or primary transfer is preferred. |
| Absolute Contraindications | Rigid, unyielding equinus deformity or severe ankle arthrosis. | A stiff joint will not benefit from a tendon transfer and will cause graft failure. Arthrodesis is indicated. |
| Absolute Contraindications | Global anterior compartment denervation (e.g., severe peroneal nerve palsy). | The EHL muscle will also be denervated and non-functional, rendering the transfer useless. |
| Relative Contraindications | Severe peripheral neuropathy or active Charcot neuroarthropathy. | High risk of surgical site complications, infection, and failure of fixation due to compromised bone density and lack of protective sensation. |
Pre-Operative Planning, Templating, and Patient Positioning
Advanced Imaging Modalities
Preoperative planning is the cornerstone of a successful reconstruction. While standard weight-bearing radiographs of the ankle and foot are obtained to rule out associated osseous pathology, degenerative joint disease, or subtle midfoot instability, Magnetic Resonance Imaging (MRI) is the definitive imaging modality. For chronic cases, MRI clearly delineates the lack of continuity of the tibialis anterior tendon, quantifies the extent of proximal retraction, and assesses the degree of fatty infiltration and muscle atrophy within the anterior compartment. Furthermore, it allows us to evaluate the caliber and integrity of the EHL tendon, ensuring it is a suitable donor. Pay close attention to the axial cuts to evaluate the muscle belly cross-sectional area, as severe fatty infiltration of the EHL would necessitate alternative strategies, such as an allograft interposition or a split anterior tibial tendon transfer.
Electrodiagnostic Evaluation
In cases where the clinical history is ambiguous or there is a suspicion of a concomitant neurologic injury, electrodiagnostic studies (EMG/NCS) are invaluable. Because tibialis anterior weakness is a hallmark of both L5 radiculopathy and common peroneal nerve entrapment, distinguishing a localized tendon rupture from a proximal nerve lesion is critical. An EMG will confirm the denervation status of the anterior compartment. In a pure tibialis anterior tendon rupture, the EHL and peroneal muscles will demonstrate normal motor unit action potentials, confirming their viability for transfer. If the EHL shows signs of denervation, the planned transfer must be aborted.
Operating Room Setup and Patient Positioning
The patient is positioned supine on the operating table. A small bump may be placed under the ipsilateral hip to slightly internally rotate the limb, bringing the anteromedial aspect of the ankle and foot into a direct, orthogonal view. A well-padded pneumatic tourniquet is applied high on the thigh. Exsanguination and tourniquet inflation are critical for providing a bloodless surgical field, which is absolutely mandatory for identifying delicate neurovascular structures and achieving precise tendon tensioning. Ensure all pressure points, particularly the contralateral heel and fibular head, are meticulously padded. A sterile C-arm fluoroscopy unit should be available in the room to confirm drill hole trajectory and interference screw placement within the medial cuneiform, particularly in patients with atypical midfoot anatomy or osteopenia.
Step-by-Step Surgical Approach and Fixation Technique
Addressing Equinus: The Gastrocnemius Recession
We begin by addressing the heel cord contracture. Utilizing the Silfverskiöld test, we have determined that the equinus is primarily driven by gastrocnemius tightness. I am making a 3-4 cm longitudinal incision on the posteromedial aspect of the calf, approximately 2-3 fingerbreadths proximal to the musculotendinous junction of the Achilles. We dissect carefully through the subcutaneous tissue, identifying and protecting the sural nerve. Upon exposing the gastrocnemius fascia, a transverse incision is made through the aponeurosis, carefully avoiding the underlying soleus muscle fibers. By gently dorsiflexing the ankle, we rupture the remaining tight fascial bands, achieving the necessary 10 to 15 degrees of passive dorsiflexion. The wound is irrigated and closed in layers.
Anterior Approach and Tendon Sheath Preservation
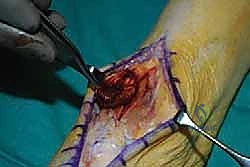
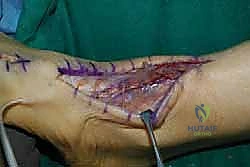
Now, we turn our attention to the anterior ankle. I am utilizing an anterior longitudinal incision, centered over the palpable, albeit empty, course of the tibialis anterior tendon. This incision extends from approximately 2 cm proximal to the ankle joint line, coursing distally to the medial cuneiform.
Meticulous subcutaneous dissection is performed to avoid creating ischemic skin flaps. We identify the superior and inferior extensor retinacula. Using a scalpel, I carefully incise these fascial bands longitudinally. It is imperative to open the tibialis anterior synovial sheath cleanly, as we will utilize this native sheath to house our transferred EHL tendon, minimizing postoperative adhesions and optimizing glide.
We isolate the retracted remnants of the tibialis anterior. The distal stump is often encased in scar tissue near the medial cuneiform, while the proximal stump is retracted and encased in a fibrous pseudotendon. We debride this non-viable tissue to healthy margins, confirming that direct repair is impossible.
Extensor Hallucis Longus Harvest and Preparation
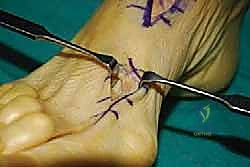
We now shift our focus to isolating the EHL tendon. Proximally, it is identified lateral to the tibialis anterior bed. To harvest the EHL distally, I make a separate 3-4 cm longitudinal incision directly over the first metatarsophalangeal (MTP) joint.
We dissect down to the EHL tendon and divide it, ensuring we leave a 1.5 cm distal stump attached to the base of the distal phalanx. This distal stump is critical for our subsequent EHB tenodesis.
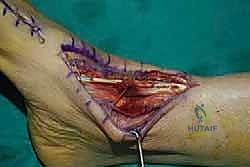
The proximal portion of the EHL is then carefully pulled through the proximal incision, liberating it from its retinacular constraints. I am now preparing the free end of the EHL graft by placing a robust, locking whipstitch using a #2 non-absorbable, high-tensile strength suture (e.g., FiberWire). This provides a secure grasp for tensioning and pulling the graft into the bone tunnel.
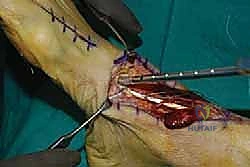
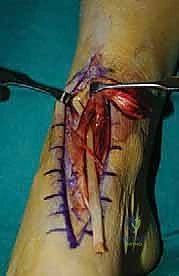
Graft Routing and Medial Cuneiform Preparation
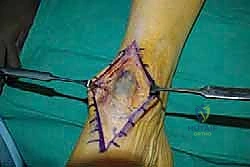
The prepared EHL tendon is now routed medially. It is critical to pass the EHL tendon deep to the extensor retinaculum and within the preserved tibialis anterior tendon sheath. This restores the anatomical pulley system and prevents anterior bowstringing.
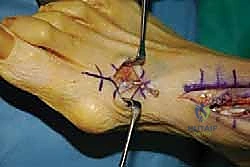
Next, we expose the dorsal and medial aspects of the medial cuneiform. A guide pin is placed from dorsal to plantar within the center of the medial cuneiform, verifying its position with fluoroscopy to avoid penetrating the intercuneiform or naviculocuneiform joints. Over the guide pin, a blind-ending bone tunnel is reamed, typically 6.0 mm or 7.0 mm in diameter, depending on the caliper-measured thickness of our EHL graft.

Tendon Fixation and Tensioning Kinematics
The whipstitched EHL tendon is passed into the medial cuneiform bone tunnel using a passing suture or a Beath pin. Tensioning is the most critical step of this procedure. The ankle is held strictly in 5 to 10 degrees of dorsiflexion, and the foot is slightly inverted to mimic the native pull of the tibialis anterior.
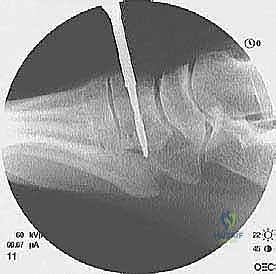
While maintaining this precise tension, a bio-tenodesis interference screw (matched to the tunnel diameter) is inserted to secure the tendon within the bone. The interference fit provides excellent initial biomechanical pull-out strength, allowing for early, safe rehabilitation. We confirm the tension by ranging the ankle; the foot should naturally rest in slight dorsiflexion, and there should be no excessive slack in the graft.
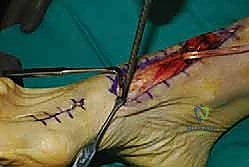
Distal EHL Stump Management and Closure
To prevent a devastating hallux malleus deformity—characterized by a plantarflexed great toe that can cause severe ulcerations and pain—we must address the distal EHL stump. Returning to our distal incision over the first MTP joint, the remaining EHL stump is identified. The adjacent extensor hallucis brevis (EHB) tendon is isolated. We perform a side-to-side tenodesis, suturing the distal EHL stump to the intact EHB tendon using 2-0 non-absorbable sutures while holding the hallux in neutral extension.
Finally, the extensor retinaculum is meticulously repaired over the transferred tendon using interrupted absorbable sutures. The subcutaneous tissues are closed, and the skin is approximated with a non-absorbable monofilament. The limb is placed in a well-padded, short-leg plaster splint with the ankle locked in neutral to 5 degrees of dorsiflexion.
Complications, Incidence Rates, and Salvage Management
Intraoperative and Early Postoperative Complications
Despite meticulous technique, complications can arise. Intraoperative injury to the deep peroneal nerve or dorsalis pedis artery is rare but catastrophic, emphasizing the need for blunt dissection in the deep anterior compartment. Early postoperative complications are primarily related to wound healing. The anterior ankle has a tenuous blood supply and limited soft tissue envelope. Wound dehiscence or superficial infection occurs in approximately 5-8% of cases, particularly in diabetic patients or smokers. Aggressive local wound care and oral antibiotics are usually sufficient, but deep infections threatening the graft or bone tunnel require immediate surgical debridement.
Late Complications and Functional Deficits
The most significant late complication is graft stretch-out or failure of fixation, resulting in recurrent foot drop. This is almost exclusively due to unrecognized, uncorrected equinus contracture or premature weight-bearing. If the graft fails, salvage options are limited and often require an allograft reconstruction or, in severe cases, a solid ankle-foot orthosis. Another specific complication is the development of a hallux malleus or a functionally limiting loss of hallux extension. This occurs if the EHB tenodesis is inadequate or fails. While some loss of powerful hallux extension is expected, a painful plantarflexed toe may require a subsequent interphalangeal joint arthrodesis.
Table: Complication Management Profile
| Complication | Estimated Incidence | Prevention Strategy | Salvage / Management |
|---|---|---|---|
| Wound Dehiscence / Infection | 5% - 8% | Meticulous soft tissue handling; avoiding tight skin closures; smoking cessation. | Local wound care; oral/IV antibiotics; surgical debridement if deep. |
| Graft Failure / Recurrent Foot Drop | 2% - 5% | Mandatory correction of equinus; robust interference screw fixation; strict NWB protocol. | Revision with allograft; split anterior tibial tendon transfer; permanent AFO. |
| Hallux Malleus Deformity | 10% - 15% | Secure EHB to distal EHL stump tenodesis with hallux held in neutral. | Conservative shoe wear modification; IP joint arthrodesis of the hallux. |
| Deep Peroneal Nerve Injury | < 1% | Blunt dissection deep to retinaculum; careful retractor placement. | Nerve repair/grafting if transected; gabapentinoids for neuropraxia. |
Phased Post-Operative Rehabilitation Protocols
Phase I: Maximum Protection (Weeks 0-6)
The initial postoperative phase is dedicated exclusively to protecting the tendon transfer and allowing for biologic incorporation into the medial cuneiform bone tunnel. The patient is placed in a rigid, short-leg cast or a locked CAM boot with the ankle immobilized in neutral to 5 degrees of dorsiflexion. The patient is strictly non-weight-bearing (NWB) on the operative extremity, utilizing crutches or a knee scooter. Elevation is critical to manage edema and protect the tenuous anterior skin incision. At the two-week mark, sutures are removed, and the patient is transitioned back into a rigid cast or locked boot.
Phase II: Controlled Mobilization (Weeks 6-10)
At six weeks, radiographic and clinical healing of the bone tunnel is typically sufficient to begin controlled stress. The patient is transitioned to a removable CAM walker boot. Weight-bearing is progressively advanced from partial to full weight-bearing as tolerated, initially with the use of crutches. Physical therapy is initiated with a strict focus on active and active-assisted range of motion. Passive plantarflexion stretching is strictly avoided during this phase to prevent stretching the newly healing graft. The focus is on re-educating the EHL muscle to fire during the swing phase of gait, often utilizing biofeedback or electrical stimulation.
Phase III: Strengthening and Return to Activity (Weeks 10-16+)
By week ten, the patient is typically weaned out of the CAM boot and into a supportive athletic shoe, often utilizing a custom orthotic to support the medial longitudinal arch. Therapy shifts toward progressive resistance exercises, focusing on concentric and eccentric dorsiflexion strengthening. Proprioceptive training, balance exercises, and gait normalization are emphasized. Patients can typically begin low-impact activities, such as stationary cycling or swimming, around 12 weeks. Return to higher-impact activities or sports is generally restricted until 4 to 6 months postoperatively, contingent upon the restoration of at least 80% of the contralateral limb's dorsiflexion strength and a normal, symmetric gait pattern.
Summary of Landmark Literature and Clinical Guidelines
Evidence-Based Outcomes of EHL Transfer
The literature robustly supports the use of EHL transfer for chronic tibialis anterior ruptures. Biomechanical studies have consistently demonstrated that the EHL has an optimal line of pull and sufficient excursion to substitute for the tibialis anterior. Clinical series, such as those by various orthopedic foot and ankle thought leaders, report high rates of patient satisfaction, reliable restoration of a plantigrade foot, and the elimination of the steppage gait. While isokinetic testing often reveals a permanent 15% to 20% deficit in peak dorsiflexion torque compared to the uninjured side, this deficit is rarely clinically significant for activities of daily living or moderate athletic pursuits. The literature unanimously stresses the importance of recognizing and releasing concomitant gastrocnemius contractures, as failure to do so is the primary predictor of poor outcomes and graft failure. The evolution of fixation techniques from simple suture anchors to bio-tenodesis interference screws has further improved early construct stability, allowing for more reliable healing and functional recovery.
