Pathogenesis and Surgical Management of Dupuytren Contracture
Key Takeaway
Dupuytren contracture is a fibroproliferative disorder of the palmar fascia driven by myofibroblast activity and type III collagen deposition. Understanding the complex pathoanatomy—specifically the transformation of normal fascial bands into pathological cords—is paramount. This guide details the cellular pathogenesis, the critical anatomy of the spiral cord, neurovascular displacement, and step-by-step surgical techniques for regional fasciectomy to optimize functional outcomes and minimize neurovascular complications.
Pathogenesis and Cellular Biomechanics
Dupuytren contracture is a progressive, fibroproliferative disorder of the palmar fascia that results in disabling flexion contractures of the digits. The foundational understanding of its pathogenesis was revolutionized in 1972 when Gabbiani and Majno definitively implicated the myofibroblast as the dominant, orchestrating cell type in the disease process.
The myofibroblast is a highly specialized contractile cell characterized by the excessive production of type III collagen. It is theorized to originate from a transformed perivascular smooth muscle cell or a locally recruited tissue fibroblast. Compared to a normal fibroblast, the Dupuytren fibroblast exhibits significantly elevated levels of α-smooth muscle actin (α-SMA), granting it a profound contractile capacity that drives the macroscopic deformity seen in the hand.
Theories of Fascial Transformation
Historically, the exact mechanism by which normal fascial structures become diseased has been a subject of intense academic debate. Today, most investigators agree that the subcutaneous nodules and cords characteristic of Dupuytren contracture are formed through a combination of de novo fibroplasia and the hypertrophy of pre-existing fibers within the palmar fascia.
- Millesi’s Theory: Millesi theorized that pathological tissue arises exclusively through structural and biochemical changes in the existing fibers of the palmar fascia, rather than through the generation of entirely new tissue planes.
- Luck’s Concept of Intermittent Stress: Luck proposed a sequential developmental model. He suggested that highly cellular subcutaneous nodules develop first. Subsequently, cords form proximally in direct response to the intermittent mechanical stress placed upon the fascia, ultimately resulting in fixed contractures.
According to Luck’s widely accepted model, the disease progresses through three distinct histological stages:
1. Proliferative Stage: Characterized by the appearance of nodules with intense cellularity, predominantly consisting of active myofibroblasts.
2. Involutional Stage: Cellularity begins to decrease. The myofibroblasts align themselves along the lines of longitudinal stress, and dense collagen deposition (shifting from type III to type I) begins to form the characteristic cords.
3. Residual Stage: The tissue becomes largely acellular and avascular, leaving behind dense, contracted, tendon-like cords of collagen that tether the digits in flexion.
Clinical Presentation and Ectopic Deposits
Dupuytren disease is often part of a broader systemic fibroproliferative diathesis. Patients with a strong genetic predisposition may present with aggressive, early-onset disease and ectopic fibromatous deposits outside the palmar fascia.

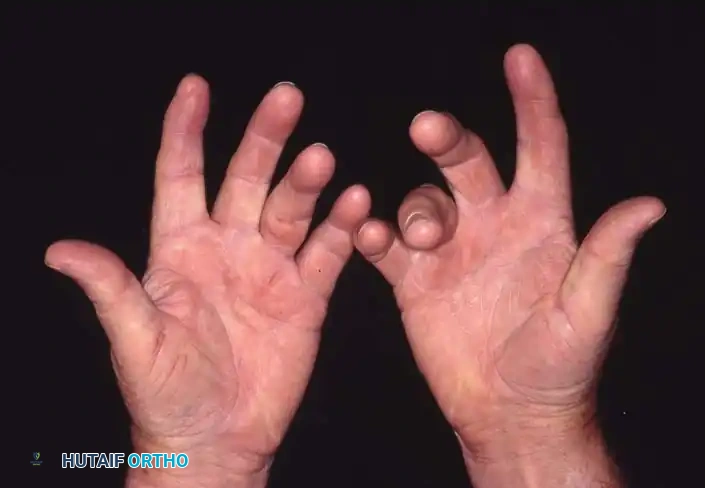
Fig. 72-1 Ectopic deposits of Dupuytren disease. A, Bilateral medial plantar and medial great toe involvement (Ledderhose disease). B, Right foot involvement. C, Dorsal proximal interphalangeal joint nodules (Garrod nodes).
The presentation in the hands is frequently asymmetrical, often beginning with a palpable nodule in the distal palmar crease, most commonly aligned with the ring or small finger.
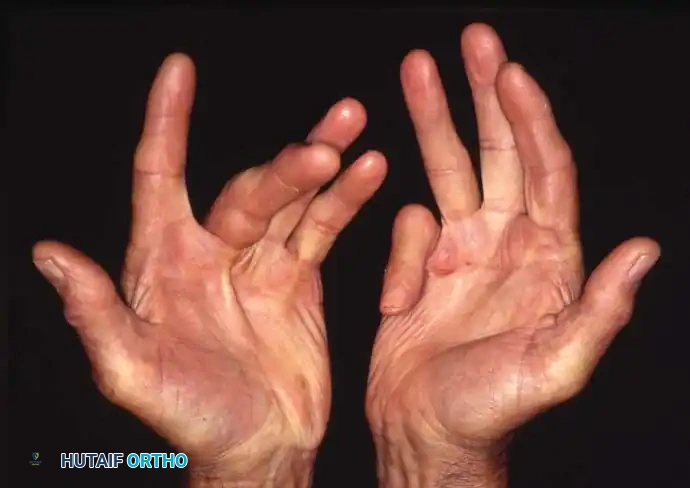
Fig. 72-2 Asymmetrical hand involvement. A, Mild bilateral ulnar hand disease. B, More diffuse bilateral disease with severe right small finger proximal interphalangeal joint contracture.
Clinical Pearl: The presence of ectopic deposits (Ledderhose disease, Peyronie disease, Garrod nodes), a strong family history, bilateral involvement, and early age of onset constitute the "Dupuytren Diathesis." These patients have a significantly higher rate of postoperative recurrence and may require more aggressive surgical management or dermofasciectomy.
Pathoanatomy: From Normal Bands to Diseased Cords
Surgical mastery of Dupuytren contracture requires a flawless understanding of the normal fascial anatomy and its pathological counterparts. McFarlane clearly outlined the predictable patterns of fascial cord involvement, establishing the principle that normal fascial bands hypertrophy and contract to become diseased cords.
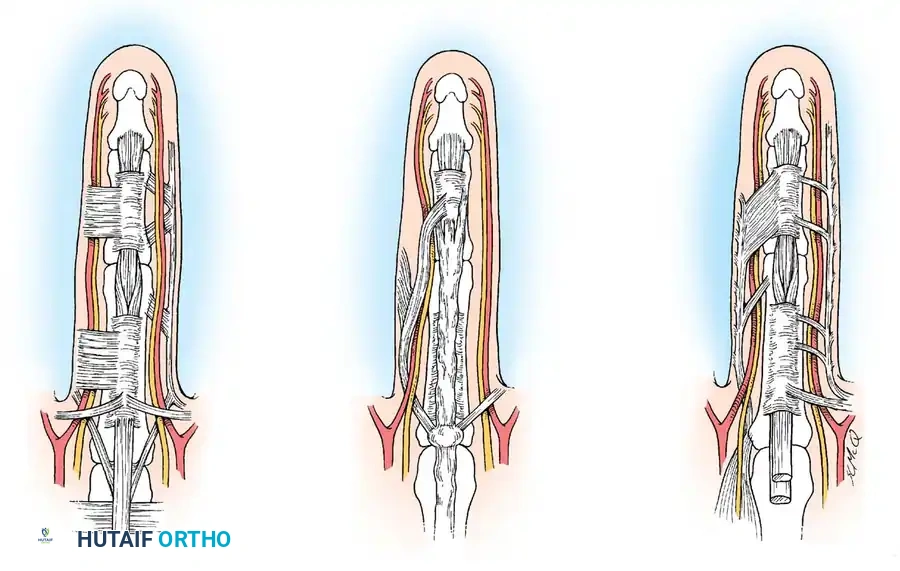
Fig. 72-3 A, Parts of palmar and digital fascia that become diseased in Dupuytren contracture. B, Diseased fascia associated with pretendinous cord. C, Diseased fascia not associated with pretendinous cord.
Standard Terminology of Fascial Structures
Understanding the nomenclature is critical for navigating the surgical planes. The transition from normal anatomy to pathological anatomy is summarized below:
- Pretendinous Band $\rightarrow$ Pretendinous Cord: The pretendinous cord is nearly always responsible for the primary flexion contracture of the metacarpophalangeal (MCP) joint. It may attach to the skin of the distal palmar crease, the base of the proximal phalanx, or extend distally to attach to the flexor tendon sheath over the middle phalanx.
- Natatory Ligament $\rightarrow$ Natatory Cord: Involvement of the natatory ligaments (ligaments of Grapow) leads to web space contractures, preventing digital abduction.
- Lateral Digital Sheet $\rightarrow$ Lateral Cord: The lateral cord contributes heavily to flexion contractures of the proximal interphalangeal (PIP) and distal interphalangeal (DIP) joints. The surgical plane between this cord and the overlying skin is notoriously thin and must be developed sharply to prevent skin necrosis.
- Cleland Ligament $\rightarrow$ Spared:
Surgical Warning: The Cleland ligament, located dorsal to the neurovascular bundle, is generally believed to be spared in Dupuytren disease. This is a vital anatomical landmark. If a structure dorsal to the neurovascular bundle is contracted, it is likely the retrovascular cord, not Cleland's ligament.
The Retrovascular and Digital Cords
- Retrovascular Cord (Thomine): Thomine described a longitudinally oriented fascia located dorsal to the neurovascular bundle. When diseased, this retrovascular cord is frequently implicated as a primary cause for recurrent PIP joint contractures if not meticulously excised during the index procedure.
- Digital Cord (Strickland and Bassett): This unusual form of the disease most commonly affects the small finger. The cord originates from the periosteum of the proximal phalanx and the fascia overlying the intrinsic muscles. It courses distally, dorsal to the neurovascular bundles, and inserts into the middle phalanx or the flexor tendon sheath. Crucially, this cord frequently displaces the neurovascular bundle superficially and toward the midline, mimicking the danger of a spiral cord.
The Spiral Cord and Neurovascular Displacement
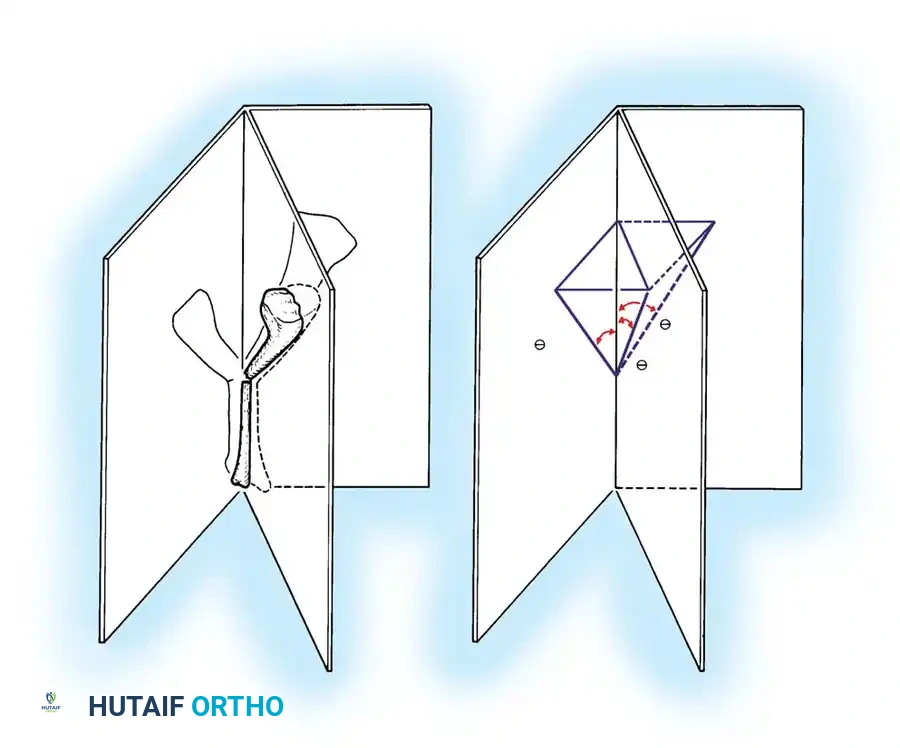
The most treacherous anatomical distortion in Dupuytren surgery is the formation of the spiral cord. A spiral cord occurs when four normally existing structures become sequentially diseased and contract together:
1. Pretendinous band
2. Spiral band
3. Lateral digital sheet
4. Grayson ligament
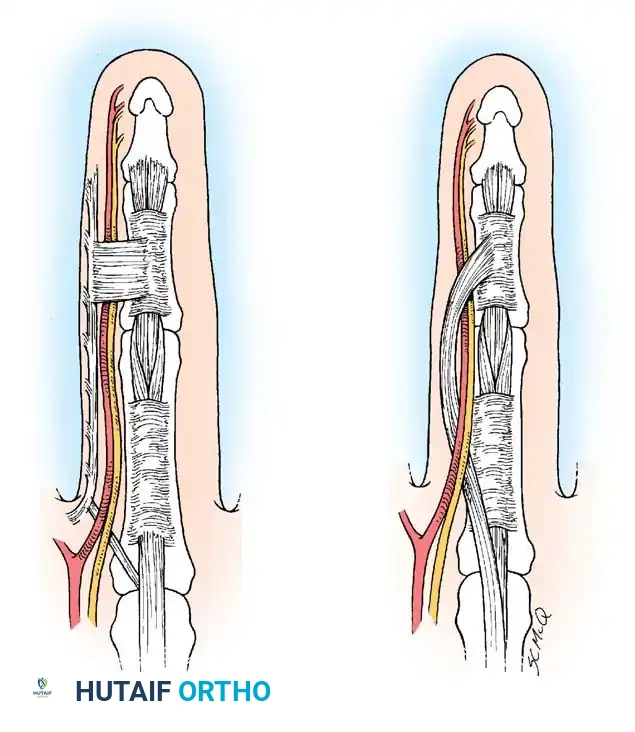
Fig. 72-4 A, Normal parts of fascia that produce spiral cord: pretendinous band, spiral band, lateral digital sheet, and Grayson ligament. B, Spiral cord showing medial displacement of neurovascular bundle.
Biomechanics of Nerve Displacement
The spiral cord runs dorsal to the neurovascular bundle proximally, but volar to it distally. As this complex cord contracts, it creates a bowstringing effect. The neurovascular bundle is drawn proximally, superficially, and toward the midline of the finger.
Pitfall: Neurovascular displacement is found most commonly on the ulnar aspect of the little and ring fingers. The nerve may lie directly beneath the skin incision, completely out of its normal anatomical bed. Tedious, magnification-assisted dissection is absolutely required to prevent catastrophic digital nerve transection.
Surgical Indications and Preoperative Planning
Surgery is not indicated for painless nodules without contracture. The primary goal of intervention is the restoration of hand function and the prevention of irreversible joint capsular contractures.
Indications for Surgery
- The Hueston Tabletop Test: The patient is unable to place the palm flat on a hard surface. This is a simple, reliable indicator of functional impairment.
- MCP Joint Contracture: Flexion contracture exceeding 30 degrees. MCP joint contractures are generally correctable regardless of severity because the collateral ligaments are stretched in flexion and do not contract.
- PIP Joint Contracture: Flexion contracture exceeding 15 to 20 degrees. PIP joint contractures are notoriously difficult to correct fully if long-standing, as the collateral ligaments become contracted in the flexed position, and the volar plate becomes rigid.
Step-by-Step Surgical Approach: Regional Fasciectomy
Regional fasciectomy remains the gold standard for the surgical management of Dupuytren contracture, offering a balance between thorough disease eradication and acceptable morbidity.
1. Anesthesia and Positioning
- Anesthesia: Regional block (axillary or supraclavicular brachial plexus block) is preferred, providing excellent intraoperative anesthesia and postoperative pain control.
- Positioning: The patient is positioned supine with the operative arm extended on a radiolucent hand table.
- Tourniquet: A well-padded pneumatic upper arm tourniquet is essential. Exsanguination with an Esmarch bandage is performed prior to inflation (typically 250 mm Hg) to ensure a bloodless field, which is mandatory for safe neurovascular dissection.
- Magnification: Surgical loupes (minimum 2.5x to 3.5x) are mandatory.
2. Incision Design
The choice of incision is critical to prevent postoperative scar contracture and to allow adequate exposure of the displaced neurovascular bundles.
* Brunner Zig-Zag Incisions: Volar zig-zag incisions are highly effective. The apices of the flaps must end at the mid-axial lines, and the transverse limbs must cross the flexion creases at acute angles.
* Longitudinal Incisions with Z-Plasty: A straight longitudinal incision over the cord can be utilized, provided it is subsequently broken up with multiple Z-plasties during closure to lengthen the skin and prevent a secondary scar contracture.
3. Exposure and Neurovascular Dissection
- Skin Flap Elevation: The skin flaps are elevated sharply. The plane is immediately subdermal. In severe disease, the dermis may be infiltrated by the cord, leaving the skin extremely thin and prone to full-thickness necrosis.
- Identification of the Neurovascular Bundle:
> Surgical Pearl: Never search for the nerve in the area of maximal disease. Always identify the normal neurovascular bundle proximally in the palm, proximal to the diseased pretendinous cord. - Once identified proximally, the nerve and artery are meticulously traced distally into the digit. A blunt probe or tenotomy scissors are used to gently separate the bundle from the surrounding spiral or lateral cords.
4. Excision of the Diseased Fascia (Fasciectomy)
- Pretendinous Cord Excision: The pretendinous cord is transected proximally at the level of the normal palmar fascia. It is elevated and dissected distally off the flexor tendon sheath.
- Spiral and Lateral Cord Excision: With the neurovascular bundle safely retracted and protected, the spiral cord and lateral cords are excised. The retrovascular cord (Thomine) must be specifically sought out dorsal to the neurovascular bundle and excised to maximize PIP joint extension.
- Natatory Cord Excision: If web space contracture is present, the natatory cords are excised, taking care not to injure the digital nerves as they bifurcate near the web spaces.
- Joint Release (If Necessary): If PIP joint contracture persists after complete fasciectomy, a sequential joint release may be required (check-rein ligament release, volar plate release). However, aggressive joint release increases the risk of postoperative stiffness.
5. Hemostasis and Closure
- The tourniquet is deflated prior to closure. Meticulous pinpoint bipolar electrocautery is used to achieve absolute hemostasis. Hematoma formation is the most common cause of postoperative morbidity and flap necrosis.
- The skin is closed loosely with interrupted non-absorbable sutures (e.g., 4-0 or 5-0 nylon). If skin tension is excessive due to chronic contracture, the wound may be left partially open to heal by secondary intention (McCash open palm technique) or covered with a full-thickness skin graft (dermofasciectomy).
Postoperative Protocol and Rehabilitation
The surgical procedure is only half of the treatment; rigorous postoperative hand therapy is paramount to a successful outcome.
- Immediate Postoperative Phase (Days 1-5): The hand is placed in a bulky, non-compressive soft dressing with a volar plaster splint holding the MCP and PIP joints in maximal comfortable extension. Elevation is strictly enforced to minimize edema.
- Early Mobilization (Days 5-14): The bulky dressing is removed. A custom thermoplastic volar extension splint is fabricated by a certified hand therapist. The patient begins active and active-assisted range of motion (ROM) exercises out of the splint multiple times a day. The splint is worn at night and between exercise sessions.
- Late Phase (Weeks 2-12): Sutures are removed at 10 to 14 days. Night splinting in extension is continued for 3 to 6 months to counteract the biological tendency for scar contracture. Strengthening exercises are gradually introduced at 4 to 6 weeks.
Complications and Pitfalls
- Digital Nerve Injury: The most devastating intraoperative complication. Risk is highest with spiral cords and recurrent disease. If transected, immediate epineural microsurgical repair is mandatory.
- Hematoma: Leads to increased pain, delayed wound healing, and skin flap necrosis. Prevented by meticulous hemostasis after tourniquet deflation and the use of drains if necessary.
- Complex Regional Pain Syndrome (CRPS) / Flare Reaction: Characterized by disproportionate pain, severe edema, stiffness, and vasomotor changes. Treated with aggressive therapy, sympathetic blocks, and short courses of oral corticosteroids.
- Recurrence: Recurrence of the disease (in the same operative field) or extension (in a previously unoperated area) is common, particularly in patients with the Dupuytren diathesis. Patients must be counseled preoperatively that surgery is a treatment, not a cure.
You Might Also Like