Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues, to this definitive masterclass on the percutaneous repair of the Achilles tendon. Over the past two decades, the paradigm of acute Achilles tendon rupture management has shifted dramatically. While open repair was once the undisputed gold standard, the advent and refinement of percutaneous and minimally invasive techniques have revolutionized our approach. This evolution is driven by the desire to mitigate the notoriously high wound complication rates associated with open posterior ankle surgery, while simultaneously achieving the robust mechanical strength required for early functional rehabilitation. The percutaneous approach, when applied judiciously and executed with technical precision, represents a powerful tool in the orthopedic surgeon's armamentarium, offering excellent functional outcomes, minimized morbidity, and an accelerated return to pre-injury activity levels.
To fully appreciate the utility of percutaneous repair, one must first understand the patho-epidemiology of Achilles tendon ruptures. These injuries are remarkably common, with an incidence rate that has been steadily rising, currently estimated at 18 to 32 per 100,000 person-years in Western populations. The classic demographic profile is the "weekend warrior"—typically a male between the ages of 30 and 50 who engages in episodic, high-intensity athletic activities. The male-to-female ratio is striking, ranging from 4:1 to 10:1. The left leg is statistically more frequently involved than the right, a phenomenon often attributed to right-handed individuals using their left leg as the primary push-off or dominant stabilizing limb during athletic maneuvers.
The pathogenesis of these ruptures is rarely a purely acute event on a perfectly healthy tendon. Instead, it is almost universally preceded by a period of asymptomatic, hypoxic degenerative tendinopathy. Histological analysis of ruptured tendons consistently reveals pre-existing degenerative changes, including decreased collagen fibril diameter, reduced tenocyte density, increased mucoid ground substance, and disorganized collagen cross-linking. This insidious degeneration weakens the structural integrity of the tendon matrix, priming it for catastrophic failure under sudden, eccentric loading. Systemic factors such as advancing age, obesity, hypercholesterolemia, and the use of certain medications—most notably fluoroquinolone antibiotics and systemic or locally injected corticosteroids—further exacerbate this matrix degradation, significantly elevating the risk of rupture.
The mechanism of injury typically involves a sudden, forceful eccentric contraction of the gastrocnemius-soleus complex. The three most common clinical scenarios include: pushing off with the weight-bearing forefoot while the knee is simultaneously extending (e.g., initiating a sprint or jumping); sudden, unexpected dorsiflexion of the ankle (e.g., stepping into a concealed hole or slipping on a stair); and violent dorsiflexion of a plantarflexed foot (e.g., a fall from a height). Understanding these mechanisms is crucial, as they dictate the pattern of the tear, which usually occurs as a "mop-end" shredding of the tendon fibers rather than a clean, transverse laceration. This shredded morphology is precisely what makes percutaneous approximation both challenging and highly reliant on specific suture-passing techniques to achieve adequate purchase.
Detailed Surgical Anatomy and Biomechanics
Before a scalpel is ever passed to the surgeon, a profound and intimate understanding of the surgical anatomy and biomechanics of the posterior lower leg is paramount. The Achilles tendon, or calcaneal tendon, is the thickest and strongest tendon in the human body. It is formed by the confluence of the gastrocnemius muscle (which originates from the posterior aspects of the medial and lateral femoral condyles and crosses two joints) and the soleus muscle (which originates from the posterior tibia and fibula and crosses only the ankle joint). The tendinous fibers of these two muscles coalesce approximately 15 centimeters proximal to the calcaneal insertion. As the tendon descends, the fibers undergo a complex 90-degree lateral rotation. This spiraling architecture is biomechanically significant; it allows for elongation and elastic energy storage during the stance phase of gait, releasing it during push-off, thereby maximizing the mechanical efficiency of plantarflexion.

The vascular supply to the Achilles tendon is a critical factor in both its vulnerability to rupture and its capacity for postoperative healing. The tendon is supplied by three distinct vascular territories: the musculotendinous junction proximally, the osseous insertion distally, and an extrinsic supply via the paratenon. Unlike tendons enclosed in true synovial sheaths, the Achilles is enveloped by a paratenon—a highly vascularized, multi-layered connective tissue sleeve consisting of a superficial epitenon and a deeper peritenon. The mid-substance of the tendon, specifically the region located 2 to 6 centimeters proximal to its calcaneal insertion, represents a relative hypovascular "watershed" zone. In this region, the intrinsic longitudinal blood supply is sparse, and the tendon relies heavily on the extrinsic vessels penetrating from the paratenon. It is no coincidence that the vast majority of acute ruptures occur exactly within this hypovascular zone. One of the primary biological advantages of the percutaneous repair technique is the meticulous preservation of the paratenon and its delicate vascular network, which optimizes the biological milieu for tendon healing.
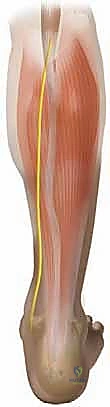
The neurovascular anatomy of the posterior leg dictates the safe zones for percutaneous instrumentation. The structure of most critical concern is the sural nerve. The sural nerve is formed by the union of the medial sural cutaneous nerve (a branch of the tibial nerve) and the sural communicating branch (from the common fibular nerve). It courses distally along the posterolateral aspect of the calf, initially deep to the crural fascia, before piercing the fascia to become superficial at a highly variable point—typically in the middle third of the leg. As it descends toward the lateral malleolus, it lies in close proximity to the lateral border of the Achilles tendon. Cadaveric studies have demonstrated that the sural nerve can be as close as 2 millimeters to the lateral margin of the tendon at the level of the standard rupture site. This anatomical intimacy makes the sural nerve highly susceptible to iatrogenic entrapment or laceration during blind percutaneous suture passage.
Biomechanically, the Achilles tendon is subjected to extraordinary forces during routine activities. During normal walking, the tendon experiences loads equivalent to 3 to 4 times body weight. During running and jumping, these loads can surge to 10 to 12 times body weight. The ultimate tensile strength of a healthy Achilles tendon is estimated to be between 5,000 and 10,000 Newtons. Therefore, any surgical repair—whether open or percutaneous—must possess sufficient initial biomechanical strength to withstand the resting tension of the triceps surae and the forces applied during early postoperative rehabilitation. While percutaneous repairs historically utilized weaker suture configurations, modern techniques employing high-strength, non-absorbable polyblend sutures and multiple core strands have demonstrated biomechanical properties that approach or equal those of traditional open repairs, allowing for safe, early functional weight-bearing.
Exhaustive Indications and Contraindications
The decision to proceed with a percutaneous Achilles tendon repair must be based on a rigorous assessment of patient-specific factors, the chronicity of the injury, and the morphological characteristics of the tear. Patient selection is the cornerstone of success in minimally invasive tendon surgery. The ideal candidate is an active individual with an acute rupture who possesses the cognitive capacity and discipline to adhere strictly to a demanding postoperative rehabilitation protocol.
Percutaneous repair is generally indicated for acute ruptures, strictly defined as those presenting within 2 to 3 weeks of the initial injury. During this acute window, the tendon ends have not yet undergone significant retraction, and the surrounding hematoma remains relatively fluid, allowing for the proximal stump to be mobilized and approximated to the distal stump with simple ankle plantarflexion. Furthermore, the percutaneous approach is highly advantageous for patients with a higher risk of wound healing complications, such as individuals with controlled diabetes mellitus, mild peripheral vascular disease, or those who use tobacco products, provided they meet all other criteria. The preservation of the posterior skin envelope and the paratenon significantly mitigates the risk of devastating soft tissue necrosis and deep infection.
Conversely, there are stringent contraindications to the percutaneous technique. Chronic ruptures (presenting after 3 to 4 weeks) are an absolute contraindication. In these cases, the proximal muscle belly retracts, the tendon stumps undergo fibrotic scarring, and the gap fills with disorganized scar tissue. Attempting to bridge a chronic gap percutaneously will inevitably result in a lengthened, functionally incompetent tendon and severe plantarflexion weakness. Similarly, acute ruptures with a massive tendon gap that cannot be closed with gravity equinus or gentle manipulation are not amenable to percutaneous repair. Open exploration with possible V-Y fascial advancement or turndown flaps is required in such scenarios.
Below is a comprehensive table detailing the indications and contraindications for percutaneous Achilles tendon repair.
| Category | Indications for Percutaneous Repair | Contraindications for Percutaneous Repair |
|---|---|---|
| Chronicity | Acute ruptures (< 3 weeks from injury) | Chronic or neglected ruptures (> 4 weeks) |
| Gap Size | Palpable gap that closes in plantarflexion | Large gap (>3 cm) irreducible in equinus |
| Patient Profile | Active patients, athletes, compliant individuals | Non-compliant patients, severe psychiatric illness |
| Local Tissue | Intact, healthy overlying skin envelope | Active local infection, severe dermatological disease |
| Comorbidities | Controlled diabetes, mild vascular risk factors | Severe peripheral arterial disease, profound neuropathy |
| Re-rupture | Primary, first-time ruptures | Revision surgery for previous failed repair (relative) |
| Anatomy | Standard mid-substance tears (2-6 cm proximal) | Avulsion fractures of the calcaneus, purely MTJ tears |
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning begins with an exhaustive clinical examination. The diagnosis of an acute Achilles tendon rupture is primarily clinical, and reliance on advanced imaging should not supersede a meticulous physical exam. The hallmark triad includes a palpable gap in the tendon substance, a positive Thompson test, and decreased resting tension. The Thompson test (calf squeeze test) is performed with the patient prone; squeezing the fleshy bulk of the gastrocnemius should elicit passive plantarflexion. Absence of this motion is pathognomonic for a complete rupture. The Matles test evaluates resting tension: with the patient prone and knees actively flexed to 90 degrees, the affected ankle will fall into neutral or dorsiflexion compared to the uninjured side, which remains in slight plantarflexion.
While the diagnosis is clinical, imaging plays a vital role in surgical templating. High-resolution ultrasonography is an excellent, dynamic, and cost-effective modality. It allows the surgeon to visualize the exact location of the tear, measure the size of the gap in neutral and plantarflexion, and assess the degree of tendon retraction. Magnetic Resonance Imaging (MRI), while not strictly necessary for a straightforward acute rupture, provides unparalleled anatomical detail. T2-weighted sagittal and axial sequences will clearly delineate the "mop-end" morphology of the torn fibers, the extent of the surrounding hematoma, and the precise level of the rupture relative to the calcaneal insertion. MRI is particularly useful in equivocal cases or when evaluating subacute presentations to ensure the gap is amenable to percutaneous approximation.
Patient positioning in the operating theater is a critical step that dictates the ease and safety of the procedure. The patient is placed in the prone position on a standard radiolucent operating table. Meticulous attention must be paid to padding all bony prominences, particularly the face, anterior shoulders, iliac crests, and the contralateral knee and ankle. The feet should be positioned so they extend just beyond the edge of the operating table. This allows the surgeon to freely manipulate the ankle through its full range of motion, which is essential for assessing tendon tension and gap closure during the repair.
We strictly advocate against the use of a thigh tourniquet for percutaneous Achilles repairs. Operating without a tourniquet allows the surgeon to continuously monitor the perfusion of the delicate posterior skin flaps. Furthermore, avoiding a tourniquet eliminates the risk of tourniquet-induced ischemia to the already compromised watershed zone of the tendon. The limb is prepped and draped in a standard sterile fashion, ensuring exposure from the mid-calf down to the toes. Before making any incisions, the surgeon should use a sterile surgical marker to delineate the palpable medial and lateral borders of the Achilles tendon, the level of the palpable defect, and the anticipated course of the sural nerve along the posterolateral calf.
Step-by-Step Surgical Approach and Fixation Technique
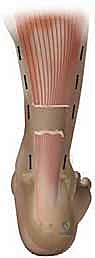
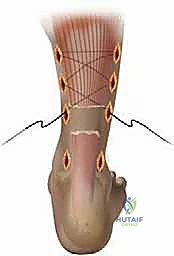
The execution of a percutaneous Achilles tendon repair demands a masterful blend of tactile feedback, spatial awareness, and meticulous suture management. The core philosophy is to achieve a robust, multi-strand core stitch that securely grasps both the proximal and distal tendon stumps while strictly avoiding entrapment of the sural nerve and preserving the paratenon. While various proprietary jig systems (e.g., PARS Arthrex, Achillon) exist to facilitate this process, the fundamental principles remain identical whether using a jig or a freehand technique.
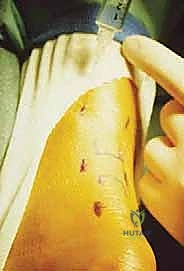
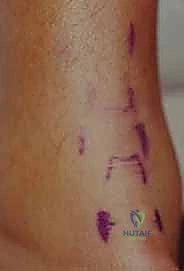
The procedure begins with the creation of a series of meticulously placed stab incisions. A transverse or longitudinal incision, approximately 1.5 to 2.0 centimeters in length, is made directly over the palpable tendon defect. Careful blunt dissection with a hemostat is performed through the subcutaneous tissue to incise the crural fascia and enter the paratenon longitudinally. The hematoma within the rupture site is gently evacuated using a combination of suction and blunt sweeping with a periosteal elevator. This clearing of the hematoma is crucial, as it allows for direct visualization of the mop-end tendon fibers and ensures that organized hematoma does not interpose between the tendon stumps, which would impede healing and functionally lengthen the repair.

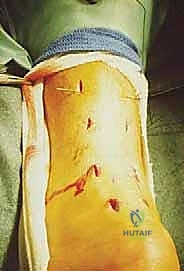
Next, attention is turned to capturing the proximal stump. Depending on the technique, 2 to 3 stab incisions are made along the lateral border of the proximal tendon, and corresponding incisions are made along the medial border. Crucial Step: When making the lateral stab incisions, the surgeon must be hyper-vigilant regarding the sural nerve. The scalpel should only incise the epidermis; a hemostat is then used to bluntly spread the subcutaneous tissues down to the tendon sheath, pushing any unseen neural structures safely away. A specialized passing instrument or a straight Keith needle is then used to pass high-strength, non-absorbable sutures (e.g., #2 or #5 FiberWire or Orthocord) transversely across the proximal tendon stump through these stab incisions. To maximize pull-out strength, a locking configuration (such as a modified Krackow or grasping box stitch) is created percutaneously by manipulating the suture ends between the stab incisions.

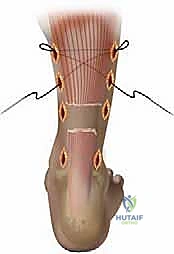
Once the proximal stump is securely controlled with 4 to 6 core strands, the process is repeated for the distal stump. Because the distal stump is shorter and closer to the calcaneal insertion, the stab incisions are placed closer together. The sutures are passed transversely through the distal stump, again utilizing a locking configuration to ensure a rigid grasp on the tendinous tissue. At this stage, all suture ends from both the proximal and distal stumps are shuttled subcutaneously, deep to the paratenon, exiting through the primary central incision over the rupture site. This subcutaneous shuttling is a delicate maneuver; the surgeon must ensure the sutures do not inadvertently loop around the sural nerve or bundle subcutaneous fat, which would lead to nerve entrapment or dimpling of the skin upon tensioning.
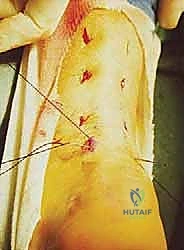
The final and most critical biomechanical step is tensioning and tying the knots. The ankle is placed into approximately 15 to 20 degrees of plantarflexion (equinus) to approximate the tendon ends and eliminate the gap. The surgeon applies axial traction to the proximal sutures to pull the proximal muscle belly distally. The corresponding proximal and distal suture strands are then tied to each other using multiple, secure square knots. It is imperative that the knots are tied deep within the tendon defect, burying them beneath the paratenon to prevent postoperative soft tissue irritation and knot prominence. After tying, the ankle is taken through a gentle range of motion to confirm that the resting tension matches the contralateral side and that the repair is mechanically solid. The paratenon is then meticulously closed over the knots with a fine absorbable suture (e.g., 3-0 Vicryl), followed by standard skin closure of the stab incisions.
Complications, Incidence Rates, and Salvage Management
While percutaneous Achilles tendon repair significantly reduces the incidence of wound complications compared to open surgery, it is not without its own unique set of risks. The operating surgeon must be acutely aware of these potential pitfalls, counsel the patient appropriately, and possess the surgical acumen to manage complications should they arise. The most feared complication specific to the percutaneous approach is iatrogenic injury to the sural nerve.
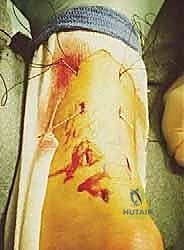
Sural nerve entrapment or laceration occurs in approximately 2% to 5% of percutaneous repairs, although some historical literature cites rates as high as 10% with older, blind techniques. Injury typically occurs during the passage of sutures through the lateral stab incisions or during the subcutaneous shuttling of the suture limbs. Clinically, a sural nerve injury presents postoperatively as paresthesia, numbness, or burning neuropathic pain along the lateral aspect of the foot and fifth toe. If a patient complains of severe, radiating lateral foot pain immediately upon awakening from anesthesia, the surgeon must maintain a high index of suspicion for acute nerve entrapment by a suture loop. In such cases, immediate re-exploration and release of the constricting suture is mandatory to prevent permanent axonal damage. If the neuropathy is delayed or mild, a period of observation and gabapentinoid therapy may be warranted, but persistent, debilitating neuromas may ultimately require surgical excision and burying of the nerve stump into adjacent muscle.
Re-rupture of the repaired tendon is another significant concern, with contemporary literature reporting an incidence of 2% to 4% following percutaneous repair. This rate is slightly higher than that of traditional open repair but significantly lower than the 10% to 12% re-rupture rate associated with non-operative management. Re-ruptures typically occur within the first 8 to 12 weeks postoperatively, often due to patient non-compliance, an accidental fall, or premature return to high-impact activities before adequate biological healing has occurred. Salvage management of a re-rupture is a complex undertaking that invariably requires an open surgical approach. Because the tendon tissue is now highly scarred and retracted, primary end-to-end anastomosis is rarely possible. Salvage techniques often involve V-Y fascial advancement of the proximal gastrocnemius aponeurosis, central turndown flaps, or, in cases of severe tissue loss, augmentation with a Flexor Hallucis Longus (FHL) tendon transfer to restore plantarflexion power.
Below is a summary table of common complications, their approximate incidence rates, and standard management strategies.
| Complication | Approximate Incidence | Mechanism / Risk Factor | Salvage / Management Strategy |
|---|---|---|---|
| Sural Nerve Injury | 2.0% - 5.0% | Lateral suture passage, blind shuttling | Immediate release if acute entrapment; Neuroma excision for chronic pain |
| Re-rupture | 2.0% - 4.0% | Non-compliance, early unprotected weight-bearing | Open revision, V-Y advancement, FHL transfer |
| Wound Infection | < 1.0% | Poor skin envelope, diabetes, smoking | Oral/IV antibiotics; rare need for debridement due to small incisions |
| DVT / PE | 1.0% - 3.0% | Immobilization, hypercoagulable state | Chemical prophylaxis (LMWH/Aspirin) per risk stratification; therapeutic anticoagulation if diagnosed |
| Tendon Elongation | Variable | Inadequate initial tensioning, aggressive early ROM | Conservative management with orthotics; late FHL transfer if severe weakness |
| Knot Prominence | 1.0% - 2.0% | Superficial knot placement, thin subcutaneous fat | Local padding; minor surgical excision of knot if persistent irritation |
Phased Post-Operative Rehabilitation Protocols
The success of a percutaneous Achilles tendon repair is inextricably linked to the postoperative rehabilitation protocol. The historical approach of prolonged, rigid cast immobilization in profound equinus has been largely abandoned due to its deleterious effects, including severe muscle atrophy, joint stiffness, and inferior long-term functional outcomes. Modern rehabilitation paradigms heavily favor early functional mobilization and controlled, protected weight-bearing. This approach leverages the concept of mechanotransduction, wherein controlled tensile loading stimulates tenocyte proliferation and promotes the longitudinal alignment of new collagen fibers, ultimately resulting in a stronger, more resilient tendon.
Phase 1: Protection and Healing (Weeks 0 to 2)
Immediately postoperatively, the patient is placed in a well-padded, rigid short-leg splint or a functional controlled ankle motion (CAM) boot locked in 20 to 30 degrees of plantarflexion. The primary goals during this phase are to protect the surgical repair, manage edema, and allow the initial inflammatory phase of healing to subside. The patient is strictly non-weight-bearing on the operative extremity, utilizing crutches or a knee scooter. Strict elevation of the limb above the level of the heart is emphasized to minimize swelling, which is crucial for the healing of the small stab incisions.
Phase 2: Early Mobilization and Progressive Weight-Bearing (Weeks 2 to 6)
At the two-week mark, the surgical incisions are inspected, and sutures are removed. The patient transitions to a CAM boot equipped with a heel lift system (typically three 1-centimeter wedges). The patient is allowed to begin progressive partial weight-bearing, advancing to full weight-bearing as tolerated while wearing the boot. Active, non-weight-bearing range of motion exercises are initiated. The patient is instructed to actively plantarflex the ankle and gently dorsiflex back to the neutral position defined by the current heel wedges. Stretching beyond neutral dorsiflexion is strictly prohibited to avoid elongating the healing tendon. Every 1 to 2 weeks, one heel wedge is removed, gradually bringing the ankle out of equinus and closer to a neutral plantigrade position.
Phase 3: Strengthening and Proprioception (Weeks 6 to 12)
By week 6, the patient typically reaches a neutral ankle position and can begin weaning out of the CAM boot into a supportive athletic shoe, often utilizing a small, temporary silicone heel cup. Formal physical therapy intensifies during this phase. The focus shifts toward regaining triceps surae strength and restoring proprioception. Exercises include double-leg heel raises, transitioning slowly to single-leg heel raises as strength permits. Stationary cycling with low resistance, swimming, and closed-kinetic-chain exercises on a leg press are incorporated. Eccentric strengthening protocols, which are highly effective for tendon remodeling, are introduced cautiously under the direct supervision of a physical therapist.
Phase 4: Return to Sport and Advanced Function (Months 3 to 6+)
The final phase of rehabilitation focuses on sport-specific training, plyometrics, and agility drills. Progression to running, jumping, and cutting maneuvers is permitted only when the patient demonstrates symmetrical ankle range of motion, normal gait mechanics without a limp, and the ability to perform repetitive, single-leg, unassisted heel raises without pain or fatigue. Return to high-impact, competitive sports is a highly individualized decision, typically occurring between 5 and 8 months postoperatively, depending on the patient's baseline athletic level and their physiological response to the rehabilitation program.
Summary of Landmark Literature and Clinical Guidelines
The transition toward minimally invasive Achilles tendon repair is firmly rooted in high-quality, evidence-based orthopedics. Over the past two decades, numerous randomized controlled trials (RCTs) and comprehensive meta-analyses have rigorously compared open repair, percutaneous repair, and non-operative management, shaping our current clinical guidelines.
One of the most pivotal studies in this domain was the randomized controlled trial published by Willits et al. in The Journal of Bone & Joint Surgery (2010). This landmark study challenged the traditional dogma by demonstrating that when an accelerated functional rehabilitation protocol is employed, the re-rupture rates between operatively and non-operatively treated patients are statistically similar. However, subsequent meta-analyses, such as the highly cited work by Soroceanu et al., have nuanced this finding. Soroceanu's analysis confirmed that while functional rehab narrows the gap, surgical repair still offers a statistically significant reduction in the absolute risk of re-rupture compared to non-operative treatment, particularly in younger, high-demand athletic populations.
When comparing open versus percutaneous surgical techniques, the literature overwhelmingly supports the minimally invasive approach for mitigating soft tissue complications. A comprehensive meta-analysis comparing open and percutaneous repairs revealed that the percutaneous cohort experienced a drastically lower rate of superficial and deep wound infections (often less than 1% compared to 5-10% in open cohorts). While early iterations of percutaneous repair showed a slightly higher incidence of sural nerve injury, the advent of modern instrumentation and the mandatory use of blunt dissection down to the paratenon have reduced this risk to levels comparable to open surgery.
The American Academy of Orthopaedic Surgeons (AAOS) Clinical Practice Guidelines for the treatment of acute Achilles tendon ruptures reflect this robust body of evidence. The guidelines provide a "Moderate" recommendation for surgical repair in active patients to minimize the risk of re-rupture and optimize functional outcomes. Furthermore, the guidelines explicitly acknowledge percutaneous and minimally invasive techniques as valid, effective alternatives to traditional open repair, specifically highlighting their benefit in reducing postoperative wound morbidity. As surgical technology continues to advance, and as our understanding of tendon mechanobiology deepens, percutaneous Achilles tendon repair will undoubtedly remain a cornerstone procedure, representing the optimal intersection of mechanical stability, biological preservation, and accelerated patient recovery.
Detailed Chapters & Topics
Dive deeper into specialized chapters regarding percutaneous-achilles-tendon-repair-perspective-1
