Percutaneous Scaphoid Fixation: An Intraoperative Masterclass
Key Takeaway
Join us in the OR for an in-depth masterclass on percutaneous scaphoid fracture fixation. We'll meticulously cover patient positioning, detailed anatomy, fluoroscopic techniques, and step-by-step execution for both dorsal and volar approaches. Learn to achieve optimal reduction and stable fixation, minimizing complications and ensuring superior patient outcomes. This guide provides invaluable pearls for every fellow.
Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues and fellows, to the operating theater. Today, we are undertaking an exhaustive examination of a ubiquitous yet notoriously unforgiving injury: the scaphoid fracture. Our definitive focus will be on percutaneous fixation, a modern surgical paradigm that, when executed with meticulous precision, offers exemplary clinical outcomes while minimizing the iatrogenic soft tissue disruption inherent to open approaches. This discourse transcends the mere mechanical insertion of a screw; it is a masterclass in comprehending the nuanced three-dimensional anatomy of the scaphoid, its precarious biomechanical environment, and the exacting surgical execution required to achieve anatomic union.
The scaphoid is the most frequently fractured carpal bone, representing approximately 60% to 70% of all carpal fractures. Epidemiologically, these injuries predominantly afflict young, active adults, typically males in their second or third decade of life. The classic mechanism of injury is a high-energy fall onto an outstretched hand (FOOSH). Biomechanically, when wrist extension exceeds 95 degrees coupled with ulnar or radial deviation, the scaphoid is forcibly compressed between the rigid palmar radiocarpal ligaments and the dorsal rim of the distal radius. This severe impingement focuses immense bending and sheer forces across the scaphoid waist, predictably culminating in fracture.
Historically, the management of nondisplaced or minimally displaced scaphoid fractures relied heavily on prolonged cast immobilization, often extending for eight to twelve weeks. However, the prolonged immobilization leads to significant morbidity, including wrist stiffness, muscle atrophy, and substantial socioeconomic costs due to delayed return to work and sport. The contemporary paradigm has shifted dramatically toward percutaneous fixation. By utilizing cannulated headless compression screws via minimally invasive dorsal or volar approaches, we can achieve rigid internal fixation, preserve the tenuous vascular supply, and facilitate immediate early mobilization, thereby mitigating the devastating sequelae of scaphoid nonunion and subsequent carpal collapse.
Detailed Surgical Anatomy and Biomechanics
Before a scalpel or guidewire ever breaches the skin, a profound understanding of scaphoid anatomy is paramount. The surgeon must visualize the bone not as a static two-dimensional image, but as a complex, dynamic structure intricately linked to the kinematics of the entire wrist.
Scaphoid Osseous Morphology and Articular Constraints
The scaphoid is frequently described as a "twisted peanut" or a boat-shaped structure (from the Greek skaphos), owing to its highly complex, multi-planar geometry. It serves as the critical mechanical bridge between the proximal and distal carpal rows. Anatomically, we divide the scaphoid into three distinct regions: the proximal pole, the waist, and the distal pole (which includes the scaphoid tubercle). Epidemiologically, 70% to 80% of fractures occur at the waist, 10% to 20% at the proximal pole, and 5% to 10% at the distal pole.
Crucially, the scaphoid articulates with five adjacent osseous structures: the radius, lunate, capitate, trapezium, and trapezoid. Because of this extensive articular requirement, approximately 80% of the scaphoid's surface area is covered by hyaline cartilage. This anatomical reality has profound implications for both healing and surgical technique. First, any hardware insertion must be meticulously calculated to ensure the implant is completely buried beneath the subchondral bone; prominent hardware will predictably cause rapid, devastating chondrolysis and secondary osteoarthritis. Second, the scaphoid possesses a distinct paucity of periosteum. Unlike diaphyseal long bones that heal via robust endochondral ossification and callus formation, the scaphoid must heal primarily through primary osteonal reconstruction. This requires absolute mechanical stability and intimate bone-to-bone contact, underscoring the necessity of rigid compression.
The Precarious Vascular Network
The vascular anatomy of the scaphoid is perhaps its most defining and clinically relevant characteristic. The blood supply is notoriously precarious, dictating the high incidence of delayed union, nonunion, and avascular necrosis (AVN), particularly in fractures of the proximal pole.
The primary arterial supply is derived from the dorsal carpal branch of the radial artery. This dorsal branch provides 70% to 80% of the overall internal vascularity. It enters the scaphoid through a series of small foramina located along the non-articular dorsal ridge, situated at the level of the scaphoid waist. From this entry point, the intraosseous vessels travel in a retrograde fashion to supply the entire proximal pole.
Conversely, a smaller volar branch, also originating from the radial artery or its superficial palmar branch, enters the bone through the scaphoid tubercle. This volar supply accounts for only 20% to 30% of the internal vascularity and exclusively supplies the distal pole. Because the proximal pole relies entirely on the retrograde flow from the dorsal ridge, any fracture occurring at the waist or proximal to it mechanically severs this blood supply. Consequently, proximal pole fractures carry a nearly 100% rate of AVN if left unreduced and unfixed, demanding urgent surgical intervention to restore stability and facilitate revascularization.
Carpal Kinematics and Instability Patterns
The scaphoid is the vital mechanical link stabilizing the carpal architecture. During normal wrist kinematics, the scaphoid flexes during radial deviation and extends during ulnar deviation, forcing the entire proximal row (lunate and triquetrum) to move in concert via the intact scapholunate interosseous ligament.
When a scaphoid fracture occurs, this mechanical linkage is uncoupled. The distal fracture fragment, driven by the strong volar radiocarpal ligaments and the trapezium/trapezoid articulation, tends to flex into a palmar direction. Simultaneously, the proximal fragment remains attached to the lunate via the scapholunate ligament. Freed from the volar-flexing influence of the distal scaphoid, the lunate follows the natural extension moment of the triquetrum, rotating dorsally. This divergent rotation creates the classic Dorsal Intercalated Segment Instability (DISI) deformity. If left untreated, this altered kinematic state leads to abnormal contact stresses, predictably culminating in Scaphoid Nonunion Advanced Collapse (SNAC)—a progressive, debilitating pattern of pan-carpal arthritis that typically manifests within a decade of the initial injury.
Critical Neurovascular Topography
Whether approaching the scaphoid dorsally or volarly, the surgeon must navigate a minefield of critical neurovascular structures. A percutaneous approach does not excuse anatomical ignorance; in fact, it demands a higher level of topographical awareness to avoid iatrogenic injury during blind guidewire insertion or portal placement.
For the dorsal approach, the primary structures at risk are the terminal branches of the superficial radial nerve (SRN). These delicate sensory branches traverse directly over the anatomic snuffbox and the first and third dorsal compartments. Iatrogenic injury to the SRN can result in debilitating neuromas and complex regional pain syndrome (CRPS). Furthermore, the extensor pollicis longus (EPL) tendon (third compartment) and the extensor digitorum communis (EDC) tendons (fourth compartment) must be carefully retracted and protected during guidewire and drill insertion.
For the volar percutaneous approach, the radial artery and the palmar cutaneous branch of the median nerve (PCBMN) are the structures of highest concern. The radial artery lies immediately radial to the flexor carpi radialis (FCR) tendon, while the PCBMN travels in the interval between the FCR and the palmaris longus. Incisions and blunt dissection must be meticulously executed directly over the palpable scaphoid tubercle, staying strictly within the safe zones to prevent catastrophic vascular injury or painful sensory denervation.
Exhaustive Indications and Contraindications
The decision to proceed with percutaneous scaphoid fixation hinges on a careful analysis of fracture morphology, displacement, chronicity, and patient-specific functional demands. While the indications for operative intervention have broadened significantly over the past two decades, careful patient selection remains the cornerstone of successful outcomes.
| Parameter | Indications for Percutaneous Fixation | Relative / Absolute Contraindications |
|---|---|---|
| Fracture Displacement | > 1 mm step-off or gap at the fracture site | Truly non-displaced fractures in compliant, low-demand patients (Relative) |
| Angular Deformity | > 10 degrees of angular displacement (humpback deformity) | Severe comminution precluding stable screw purchase (Relative) |
| Carpal Alignment | Radiolunate angle > 15°; Scapholunate angle > 60°; Intrascaphoid angle > 35° | Concomitant severe perilunate dislocation requiring open repair (Relative) |
| Fracture Location | Proximal pole fractures (high risk of AVN/nonunion) | Very small proximal pole avulsions (unfixable with screws) (Absolute) |
| Chronicity | Delayed presentation (> 3-4 weeks post-injury) | Established nonunion with cystic changes requiring bone grafting (Absolute for solely percutaneous fixation without grafting) |
| Patient Factors | High-demand athletes, manual laborers, intolerance to prolonged casting | Active local infection, severe osteopenia, medical unfitness for anesthesia (Absolute) |
For acute, nondisplaced fractures of the scaphoid waist, percutaneous fixation is increasingly viewed as a highly favorable alternative to prolonged cast immobilization. While casting can achieve union in 85% to 90% of strictly nondisplaced waist fractures, it requires 8 to 12 weeks of immobilization, resulting in profound stiffness and functional impairment. Percutaneous fixation in these cohorts offers union rates approaching 98%, with a dramatically accelerated return to work and sport.
Conversely, absolute contraindications include established scaphoid nonunions with significant bone loss, cystic resorption, or structural collapse (humpback deformity). These scenarios necessitate an open approach to debride the nonunion site, correct the angular deformity, and interpose a structural autograft (e.g., iliac crest or vascularized pedicle graft) to restore carpal height and biology. Attempting to treat a collapsed nonunion with isolated percutaneous screw fixation will universally fail, as it does not address the biological deficit or the structural malalignment.
Furthermore, fractures with severe, multi-fragmentary comminution of the scaphoid waist may not be amenable to isolated central screw fixation. In such instances, the compressive forces of a headless screw may cause the fracture to telescope and shorten, exacerbating the deformity. These complex injuries often require open reduction, potential bone grafting, and neutralization with multiple K-wires or a specialized scaphoid plate.
Pre-Operative Planning, Templating, and Patient Positioning
Preoperative planning is the blueprint for surgical success. A poorly planned percutaneous fixation will invariably lead to eccentric guidewire placement, articular penetration, or inadequate fracture compression.
Clinical Evaluation and Advanced Imaging Modalities
The clinical evaluation of a suspected scaphoid fracture demands a high index of suspicion. Patients typically present with radial-sided wrist pain, localized swelling, and significantly restricted range of motion, particularly in dorsiflexion. Classic examination findings include exquisite tenderness within the anatomic snuffbox, pain upon deep volar palpation over the scaphoid tubercle, and a positive scaphoid compression test (pain elicited by axial loading of the first metacarpal).
Standard radiographic evaluation must include a minimum of four views: a posteroanterior (PA) view with the wrist in ulnar deviation (which extends the scaphoid to profile its true length), a true lateral view, and both semipronated and semisupinated 45-degree oblique views. The lateral view is critical for assessing the intrascaphoid angle and identifying DISI deformity.
However, plain radiographs are notoriously insensitive for occult scaphoid fractures and inadequate for precisely characterizing displacement. Therefore, a high-resolution Computed Tomography (CT) scan, acquired in the longitudinal axis of the scaphoid with 1-mm sagittal and coronal reformats, is considered the gold standard for preoperative planning. The CT scan allows the surgeon to definitively measure fracture gapping, assess comminution, calculate the intrascaphoid angle, and map the optimal trajectory for the central screw. Magnetic Resonance Imaging (MRI) is reserved for detecting radiographically occult fractures in the acute setting or assessing the vascular viability of the proximal pole in cases of delayed presentation.
Implant Selection and Pre-Surgical Templating
The contemporary gold standard for scaphoid fixation is the cannulated, headless, variable-pitch compression screw (e.g., Acutrak, Synthes Headless Compression Screw). These implants are engineered to be buried entirely within the intra-articular confines of the scaphoid. Their variable pitch design—where the threads at the leading edge have a wider pitch than those at the trailing edge—forces the distal and proximal fracture fragments to compress together as the screw is advanced.
Pre-surgical templating using the CT scan or calibrated radiographs is mandatory. The surgeon must determine the maximum length of the scaphoid along its central axis. The selected screw should ideally be 2 to 4 millimeters shorter than the total measured length of the scaphoid. This intentional undersizing ensures that the screw can be completely countersunk beneath the proximal and distal articular cartilage, even after fracture compression occurs. An oversized screw will inevitably protrude into the radiocarpal or scaphotrapezial joint, causing catastrophic chondral damage.
Operating Room Setup and Patient Positioning
Optimal operating room setup is critical for seamless fluoroscopic imaging, which acts as the surgeon's "eyes" during percutaneous procedures.
For the Dorsal Arthroscopy-Assisted Reduction and Fixation (AARF) approach, the patient is positioned supine with the operative arm extended on a radiolucent hand table. A pneumatic tourniquet is applied to the proximal arm. The shoulder is abducted to 90 degrees. If arthroscopy is planned, the hand is suspended vertically using sterile finger traps applied to the index and middle fingers, with 10 to 15 pounds of counter-traction applied across the upper arm. This vertical suspension distracts the radiocarpal and midcarpal joints, creating the necessary working space for the arthroscope. The mini C-arm is positioned parallel to the floor, entering from the head or foot of the table, allowing for unobstructed PA and lateral imaging simply by rotating the machine.
For the Volar Percutaneous Approach, the patient remains supine, but the arm positioning is modified. The forearm is fully supinated, and the wrist is extended and ulnarly deviated over a rolled towel or specialized wrist bolster. This specific maneuver serves to deliver the distal pole and the scaphoid tubercle volarly, making it highly palpable and accessible for guidewire insertion. The C-arm is positioned to allow rapid transition between PA and true lateral views without disturbing the sterile field.
Step-by-Step Surgical Approach and Fixation Technique
We will now detail the precise execution of both the dorsal and volar percutaneous techniques. The choice of approach is dictated by fracture location: proximal pole and proximal waist fractures are best addressed dorsally, while distal pole and distal waist fractures are typically approached volarly.
Dorsal Arthroscopy-Assisted Reduction and Fixation (AARF)
The dorsal approach leverages the anatomical reality that the proximal pole is situated superficially beneath the dorsal capsule. The key to this approach is achieving the fluoroscopic "ring sign."
-
Establishing the "Ring Sign" and Initial Guidewire Placement:
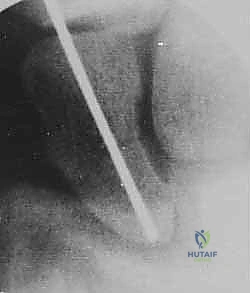
Under fluoroscopy, the wrist is placed in a true PA position. The surgeon then gently pronates the wrist until the scaphoid appears as a uniform, oblong cylinder, indicating sagittal alignment of the poles. Next, the wrist is flexed approximately 45 degrees. On the fluoroscopic monitor, the cylinder will foreshorten and rotate into a perfect cortical "ring."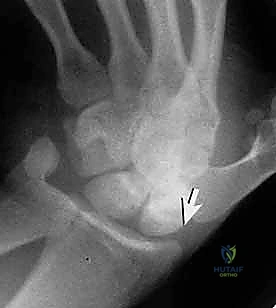
This ring represents the cortical outline of the scaphoid viewed perfectly down its central longitudinal axis. The absolute center of this ring is our precise target.
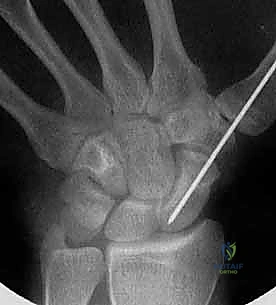
Using a 14-gauge angiocatheter or a specialized drill guide to protect the soft tissues, a 0.045-inch guidewire is advanced percutaneously directly into the center of the ring on the proximal pole.
-
Arthroscopic Assessment (Optional but Recommended):
If utilizing AARF, the hand is suspended, and standard radiocarpal and midcarpal portals (3-4, 4-5, and radial midcarpal) are established. The arthroscope allows direct visualization of the fracture hematoma, which is debrided. The surgeon can directly visualize the articular step-off and manipulate the wrist to achieve anatomic reduction under direct vision prior to advancing the guidewire across the fracture site.
Volar Percutaneous Approach Execution
The volar approach is preferred for waist fractures and relies on targeting the scaphoid tubercle.
-
Incision and Dissection:
With the wrist extended and ulnarly deviated, the scaphoid tubercle is palpated just proximal to the distal wrist crease, radial to the FCR tendon. A 1-2 cm longitudinal incision is made directly over the tubercle. Blunt dissection with a hemostat is mandatory to spread the soft tissues down to the capsule, meticulously protecting the radial artery radially and the palmar cutaneous branch of the median nerve ulnarly. -
Guidewire Placement:
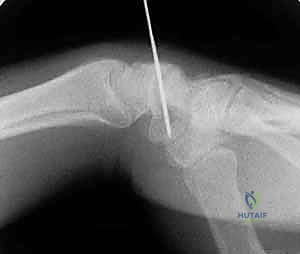
A 14-gauge needle or drill guide is placed directly onto the cartilaginous surface of the scaphoid tubercle. The trajectory is critical: the guidewire must be aimed dorsally, proximally, and ulnarly, targeting the proximal-ulnar corner of the scaphoid (near its articulation with the lunate).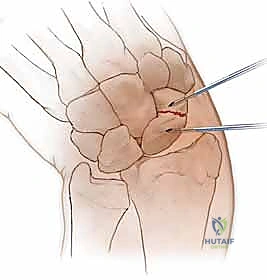
The wire is driven under continuous biplanar fluoroscopy until it engages the subchondral bone of the proximal pole.
Drilling, Measuring, and Headless Screw Insertion
Regardless of the approach (dorsal or volar), once the central guidewire is perfectly positioned, the fixation steps are identical.
-
Measuring Screw Length:
A specialized depth gauge is slid over the guidewire down to the level of the articular cartilage.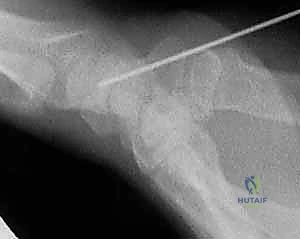
The surgeon reads the measurement and subtracts 2 to 4 millimeters. For example, if the gauge reads 26 mm, a 22 mm or 24 mm screw is selected to ensure it can be fully countersunk.
-
Overdrilling the Cortex:
A cannulated, stepped hand reamer or power drill is passed over the guidewire. It is imperative to use a soft tissue protector guide to prevent the drill flutes from wrapping and avulsing adjacent tendons or nerves. The drill is advanced to the predetermined depth.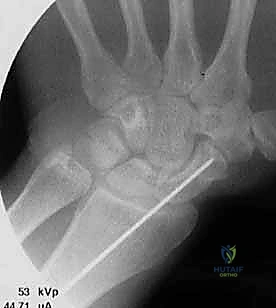
-
Screw Insertion and Compression:
The selected headless compression screw is loaded onto the cannulated screwdriver and advanced over the wire.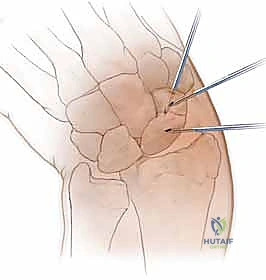
As the trailing threads engage the near cortex, the variable pitch mechanism will begin to actively compress the fracture site. The surgeon must carefully monitor this compression fluoroscopically and tactilely.
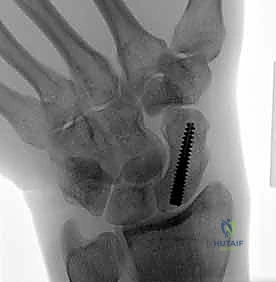
Final Fluoroscopic Verification and Closure
The final, and arguably most critical, step is verifying that the hardware is entirely intraosseous.
The C-arm must be swept through a full 180-degree arc of motion, capturing multiple oblique profiles of the scaphoid.
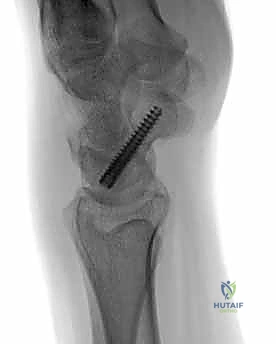
Even a single millimeter of screw protrusion into the radiocarpal or midcarpal joint is unacceptable and mandates immediate screw removal and replacement with a shorter implant.

Once perfect placement and compression are confirmed, the guidewire is removed.
The small percutaneous incisions are irrigated and closed with simple interrupted non-absorbable sutures. A sterile compressive dressing and a volar thumb spica splint are applied.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, percutaneous scaphoid fixation is not devoid of complications. The surgeon must be prepared to identify and manage these adverse events promptly.
| Complication | Estimated Incidence | Prevention and Salvage Management Strategies |
|---|---|---|
| Hardware Prominence | 2% - 5% | Prevention: Accurate templating; subtracting 2-4mm from measured length; 360-degree fluoroscopic sweep. Management: Immediate reoperation for screw exchange or removal to prevent rapid chondrolysis. |
| Nonunion | 2% - 10% (Higher in proximal pole) | Prevention: Anatomic reduction; central screw placement; robust compression. Management: Open reduction, rigid fixation, and vascularized bone grafting (e.g., 1,2 ICSRA or medial femoral condyle graft). |
| Avascular Necrosis (AVN) | 5% - 15% | Prevention: Expedited surgical intervention for proximal pole fractures to restore blood flow. Management: Vascularized bone grafting; if fragmented, salvage procedures (e.g., proximal row carpectomy or four-corner fusion). |
| Superficial Radial Nerve Injury | 1% - 3% | Prevention: Meticulous blunt dissection during dorsal portal/incision placement; use of soft tissue drill guides. Management: Observation for neuropraxia; surgical exploration and neuroma excision/burying if permanent and debilitating. |
| Infection | < 1% | Prevention: Strict adherence to sterile technique; prophylactic antibiotics. Management: Oral or IV antibiotics; hardware removal if infection tracks deep to the bone (rare). |
Hardware prominence remains the most common technical error. Because the scaphoid is almost entirely covered in cartilage, a screw that is too long will inevitably act as a mechanical rasp against the adjacent articular surfaces of the radius or trapezium. If a patient presents post-operatively with new-onset, mechanical clicking or severe pain with wrist motion, hardware prominence must be suspected and evaluated with a CT scan.
In cases where percutaneous fixation fails and results in a nonunion, salvage management becomes highly complex. If the scaphoid architecture is maintained without collapse, an open approach with non-vascularized cancellous bone grafting (e.g., from the distal radius) and revision screw fixation may suffice. However, if there is AVN of the proximal pole or structural collapse (humpback deformity), the surgeon must employ vascularized bone grafts. The 1,2 Intercompartmental Supraretinacular Artery (ICSRA) pedicled graft from the dorsal distal radius is a workhorse for restoring vascularity. For recalcitrant cases with significant bone loss, free vascularized grafts from the medial femoral condyle offer robust structural and biological support.
Phased Post-Operative Rehabilitation Protocols
The primary advantage of percutaneous fixation is the ability to bypass prolonged cast immobilization, thereby preserving wrist kinematics and accelerating functional recovery. However, rehabilitation must be carefully phased to protect the healing bone.
Phase I: Acute Protection and Edema Control (Weeks
