Mastering the Anterior Lumbar Approach: A Retroperitoneal Discectomy and Fusion Masterclass

Key Takeaway
This masterclass guides fellows through the anterior lumbar approach, focusing on retroperitoneal dissection for spinal access from L2-S1. We meticulously cover surgical anatomy, patient positioning, and step-by-step intraoperative execution, emphasizing neurovascular protection and critical decision-making. Learn to navigate complex vascular structures, manage potential pitfalls, and ensure optimal patient outcomes for discectomy, fusion, and deformity correction.
Comprehensive Introduction and Patho-Epidemiology
Welcome, esteemed colleagues and fellows, to the operating theater and this advanced masterclass. Today, we are undertaking a rigorous exploration of the intricacies defining the anterior lumbar approach. This surgical corridor is a foundational, indispensable skill for any comprehensive spine surgeon. The anterior approach offers unparalleled, direct, and expansive access to the anterior column of the lumbar spine, extending seamlessly from the L2-3 disc space down to the pivotal lumbosacral junction at the first segment of the sacrum, S1. Mastery of this approach equips the surgeon with a powerful tool to address a vast array of complex lumbopelvic pathologies. From severe degenerative disc disease necessitating robust interbody fusion or total disc replacement, to aggressive infectious processes such as pyogenic discitis and osteomyelitis requiring radical debridement, and even complex sagittal deformity correction or neoplastic tumor resection requiring multi-level corpectomy, the anterior approach remains the gold standard for anterior column reconstruction.
The patho-epidemiology of lumbar spine disease heavily favors interventions that directly address the anterior column. Biomechanically, the anterior column—comprising the vertebral bodies and intervertebral discs—bears approximately eighty percent of the axial load transmitted through the human spine. Consequently, it is the primary epicenter for degenerative cascade initiation, leading to disc space collapse, foraminal stenosis, and subsequent loss of regional lumbar lordosis. By accessing the spine anteriorly, we completely bypass the posterior paraspinal musculature, thereby eliminating iatrogenic crush injury to the multifidus and erector spinae muscles, which is a notorious cause of failed back surgery syndrome and chronic postoperative axial pain. Furthermore, the anterior approach allows for the insertion of interbody grafts with massive footprints. These large-footprint devices maximize the endplate contact area, dramatically enhancing the biomechanical environment for arthrodesis, load-sharing, and the restoration of physiological lumbar lordosis, which is critical for global sagittal balance.
Our primary objective in mastering this technique is to execute a safe, highly efficient, and comprehensive exposure while meticulously protecting the dense network of vital anterior visceral, neurologic, and vascular structures. We will focus predominantly on the retroperitoneal approach executed from the patient's left side. This specific trajectory is widely preferred in the academic community for its directness, anatomical predictability, and significantly reduced morbidity compared to transperitoneal or true lateral (transpsoas) approaches for most infra-renal lumbar levels. The left-sided approach capitalizes on the more favorable mobilization characteristics of the aorta compared to the thin-walled, easily torn inferior vena cava (IVC) located on the right.
Furthermore, accessing the L5-S1 disc space—the most frequently addressed level in anterior lumbar surgery—is highly optimized via the left retroperitoneal corridor. At this junction, the surgeon operates within the natural bifurcation of the great vessels. Approaching from the left allows the surgeon to gently retract the left common iliac artery and vein laterally, exposing the "bare area" of the L5-S1 promontory without requiring the extensive, high-risk vascular mobilization necessary for higher lumbar levels. This masterclass will systematically deconstruct the anatomical, preoperative, and technical nuances required to perform this procedure with the utmost precision and safety.
Detailed Surgical Anatomy and Biomechanics
Before a scalpel ever touches the skin, a profound, three-dimensional understanding of the regional retroperitoneal anatomy is absolutely paramount. We are operating in an unforgiving, densely packed anatomical landscape, rich with critical neurovascular, lymphatic, and visceral structures. A millimeter of miscalculation can lead to catastrophic, life-threatening complications.
Visceral and Genitourinary Anatomy
The visceral contents of the abdomen are contained within the peritoneal sac, which must be carefully mobilized medially to expose the retroperitoneal space. While the left kidney is rarely directly visualized during a routine infra-renal anterior lumbar approach, it is crucial to remember its anatomical presence. Typically residing at the L1-2 level and enveloped by thick perinephric fat and Gerota's fascia, the kidney can be inadvertently encroached upon during exposures of the upper lumbar spine. Aggressive superior dissection or reckless retraction high in the retroperitoneal space can, in rare instances, risk capsular injury or traction on the renal vasculature.
The left ureter represents a critical structure that must be definitively identified and meticulously protected in every single case. It typically lies anteriorly, adhering to the undersurface of the posterior peritoneum. As the peritoneum is swept medially, the ureter should be mobilized along with it. Its characteristic peristaltic waves, often stimulated by gentle mechanical palpation or a drop of sterile saline, can definitively confirm its identity and distinguish it from retroperitoneal blood vessels or the sympathetic chain. We must ensure the ureter is safely swept to the patient's right, well out of the operative field and protected beneath our medial retractors. Iatrogenic injury to the ureter is a devastating complication that can lead to significant morbidity, including urinoma, hydronephrosis, retroperitoneal fistula formation, and potentially the permanent loss of renal function.

Muscular and Fascial Anatomy
The muscular boundaries of our dissection define the safe corridors of approach. The psoas major muscle is the dominant landmark in the retroperitoneum. This large, fusiform paraspinal muscle originates from the transverse processes, intervertebral discs, and lateral aspects of the vertebral bodies of T12 through L5, eventually inserting onto the lesser trochanter of the femur. The medial border of the psoas forms the lateral boundary of our working window on the anterior spine. Retraction of the psoas must be performed with great care to avoid injury to the embedded lumbosacral plexus.
Navigating the anterior abdominal wall requires a precise understanding of its layered configuration, which varies significantly above and below the arcuate line of Douglas. Above the arcuate line (typically located midway between the umbilicus and the pubic symphysis), the rectus abdominis muscle is completely enclosed by the rectus sheath. The anterior layer is formed by the aponeurosis of the external oblique and the anterior leaf of the internal oblique, while the posterior layer is formed by the posterior leaf of the internal oblique and the aponeurosis of the transversus abdominis.
Below the arcuate line, a critical anatomical shift occurs. The posterior rectus sheath abruptly ends. Here, all three aponeurotic layers (external oblique, internal oblique, and transversus abdominis) pass anterior to the rectus abdominis muscle. Consequently, the rectus abdominis muscle lies directly upon the thin transversalis fascia, which is immediately superficial to the delicate extraperitoneal fat and the peritoneum. This structural difference fundamentally dictates our fascial incisions and deep retraction strategies. A careless deep incision below the arcuate line can easily violate the peritoneum, transforming a retroperitoneal approach into an unintended transperitoneal one, complicating the exposure and increasing the risk of postoperative ileus or bowel injury.
Neurologic Anatomy
The retroperitoneal space hosts several critical neural networks that are highly vulnerable during dissection and retraction. The sympathetic chain typically lies in a paraspinal position, running longitudinally anterior and slightly medial to the psoas muscle over the anterolateral aspect of the vertebral bodies. As it descends over the sacral promontory, it forms the superior hypogastric plexus (presacral plexus).
Injury to the superior hypogastric plexus, particularly at the L5-S1 level, carries a significant and devastating risk of retrograde ejaculation in male patients. This plexus innervates the internal vesical sphincter; its disruption prevents the sphincter from closing during emission, causing semen to be propelled retrogradely into the bladder. To mitigate this risk, dissection over the L5-S1 disc space must be strictly blunt. The use of monopolar electrocautery in this region is absolutely contraindicated. Instead, we utilize gentle blunt sweeping with Kittner dissectors or bipolar electrocautery strictly limited to targeted bleeding vessels.
The lumbosacral plexus is located posteromedial to and within the deep substance of the psoas muscle. While typically protected by the bulk of the psoas during a standard anterior approach, overly aggressive, prolonged, or excessively deep lateral retraction of the psoas muscle can risk stretch or crush injury to these vital nerve roots. Such injuries can manifest as profound motor weakness or sensory deficits in the distributions of the femoral, obturator, or lateral femoral cutaneous nerves. Additionally, the genitofemoral nerve rests directly on the anterior epimysium of the psoas muscle, coursing inferiorly and laterally. While typically sensory, iatrogenic injury can cause debilitating dysesthesia or numbness in the anterior thigh, scrotum, or labia majora.
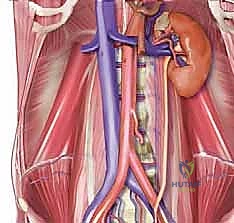
Lymphatic and Vascular Anatomy
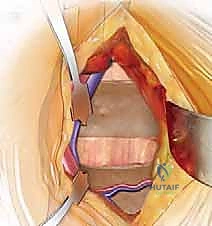
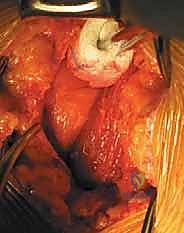
The vascular anatomy is undeniably the most critical, complex, and technically demanding aspect of the anterior lumbar approach. The abdominal aorta lies anterior to the spine and slightly to the left of the midline. It typically bifurcates into the left and right common iliac arteries at the lower margin of the L4 vertebral body or the L4-5 disc space. The inferior vena cava (IVC) lies posterior and to the right of the aorta, bifurcating into the common iliac veins slightly lower than the aortic bifurcation. Due to its thin wall and deep, right-sided position, the IVC should never be aggressively mobilized to the left, as this invites catastrophic avulsion of the short, fragile lumbar veins entering its posterior aspect.
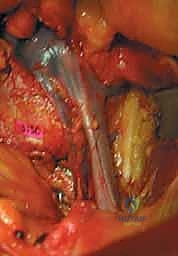
Accessing L5-S1 is generally straightforward because it lies within the bifurcation of the great vessels. The left common iliac artery and vein traverse the L5-S1 disc space diagonally. Gentle lateral retraction of these left-sided vessels typically exposes the entire L5-S1 disc. Conversely, accessing L4-5 requires significant mobilization of the great vessels from left to right. This necessitates the critical identification and ligation of the iliolumbar vein.
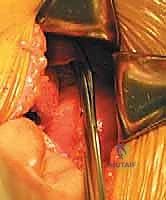
The iliolumbar vein is a robust tethering vessel that branches laterally from the left common iliac vein and courses posteriorly into the iliac fossa, deep to the psoas and obturator nerve. It must be meticulously dissected, double-ligated, and divided to permit safe medial mobilization of the left common iliac vein. Failure to identify and ligate this vein before retracting the common iliac vein will result in avulsion of the iliolumbar vein directly from the caval junction, resulting in torrential, rapidly exsanguinating hemorrhage that is incredibly difficult to control. Furthermore, the middle sacral artery and vein, which branch vertically from the bifurcation down over the L5-S1 disc, must be identified and securely ligated or cauterized with bipolar energy.
Exhaustive Indications and Contraindications
The decision to utilize the anterior lumbar approach must be rooted in a rigorous, evidence-based evaluation of the patient's specific pathology, anatomical constraints, and overall physiological reserve. Patient selection is the ultimate determinant of surgical success.
The primary indications for an Anterior Lumbar Interbody Fusion (ALIF) or Total Disc Replacement (TDR) revolve around the need for robust anterior column support and maximal surface area for arthrodesis. Severe degenerative disc disease (DDD) with disc space collapse, particularly at L4-5 and L5-S1, is a classic indication. The anterior approach allows for the insertion of a highly lordotic, large-footprint interbody cage that rests on the dense cortical apophyseal ring of the vertebral endplates, minimizing the risk of cage subsidence. This is particularly crucial in cases of iatrogenic flatback syndrome or degenerative sagittal imbalance, where restoring the natural lumbar lordosis is the primary biomechanical goal.
Furthermore, the anterior approach is highly indicated for the treatment of pseudarthrosis following a failed posterior fusion attempt. By approaching the spine through a virgin anterior surgical corridor, the surgeon avoids navigating through dense posterior epidural scar tissue, significantly reducing the risk of incidental durotomy or nerve root injury while providing a massive graft bed to secure fusion. Additionally, infectious processes such as pyogenic discitis or osteomyelitis often destroy the anterior column. The anterior approach allows for direct, radical debridement of infected necrotic bone and immediate structural reconstruction.
| Category | Specific Conditions | Rationale / Considerations |
|---|---|---|
| Primary Indications | Degenerative Disc Disease (L4-S1), Spondylolisthesis (Grade 1-2), Sagittal Deformity/Flatback | Maximal lordosis restoration, large footprint for fusion, indirect foraminal decompression. |
| Secondary Indications | Pseudarthrosis (failed posterior fusion), Discitis/Osteomyelitis, Anterior Column Tumors | Virgin surgical plane, direct access for radical debridement and structural corpectomy. |
| Relative Contraindications | Prior extensive abdominal/pelvic surgery, Obesity (BMI > 35), Mild peripheral vascular disease | Increased risk of adhesions, difficult exposure depth, potential for vascular complications. Requires multidisciplinary planning. |
| Absolute Contraindications | Severe aortoiliac calcification/atherosclerosis, Active abdominal/pelvic infection, Prior retroperitoneal radiation | Extreme risk of catastrophic vascular rupture during mobilization. Hostile surgical field precluding safe access. |
Conversely, contraindications must be respected to avoid catastrophic outcomes. A history of multiple prior abdominal surgeries, particularly those involving the retroperitoneum or pelvis (e.g., radical prostatectomy, complex hysterectomy), presents a relative contraindication due to the presence of dense, obliterative adhesions that obscure normal anatomical planes. Severe obesity (BMI > 40) exponentially increases the technical difficulty of the procedure by deepening the surgical corridor and limiting the angle of approach, though it is not an absolute contraindication in experienced hands.
The most critical absolute contraindications relate to vascular pathology. Severe, circumferential calcification of the abdominal aorta or common iliac vessels (often visualized as "lead pipe" vessels on preoperative imaging) renders the vessels rigid and completely non-pliable. Attempting to retract such heavily diseased vessels will almost certainly result in intimal tearing, plaque rupture, or full-thickness avulsion, leading to uncontrollable hemorrhage or acute lower extremity ischemia. In such cases, an alternative approach (e.g., TLIF or PLIF) must be utilized.
Pre-Operative Planning, Templating, and Patient Positioning
Thorough, obsessive preoperative planning is the absolute cornerstone of a successful anterior lumbar approach. The surgeon must construct a complete mental model of the patient's unique retroperitoneal anatomy before entering the operating room.
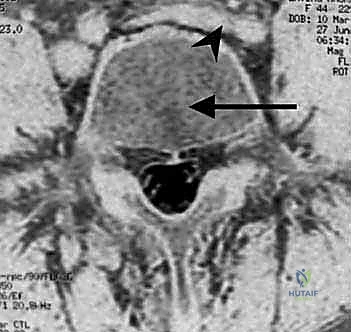
Standard plain standing radiographs (AP and Lateral) are initially reviewed to assess global lumbopelvic alignment, pelvic incidence, and the degree of disc space collapse. Crucially, the lateral radiograph must be scrutinized for the presence of aortic or iliac calcifications. However, the definitive planning tools are advanced cross-sectional imaging modalities. High-resolution, thin-cut MRI or CT scans are indispensable for mapping the vascular anatomy relative to the disc spaces.
The surgeon must precisely identify the axial level of the aortic bifurcation and the inferior vena cava confluence. A "high" bifurcation (e.g., at L3-L4) generally makes L4-5 access easier but can complicate L5-S1 access if the left common iliac vein descends at a steep, oblique angle across the disc space. Conversely, a "low" bifurcation (e.g., at the L5 vertebral body) can severely restrict the "vascular window" available at L4-5, requiring extensive, high-risk mobilization of the bifurcation itself. The surgeon must also evaluate the amount of retroperitoneal fat and the size and morphology of the psoas muscles.

For revision anterior lumbar approaches, the stakes are exponentially higher. The virgin retroperitoneal planes are replaced by dense, mature scar tissue that firmly tethers the great vessels to the anterior longitudinal ligament and the bowel to the peritoneum. In these highly complex scenarios, preoperative consultations with a vascular surgeon and a urologist are mandatory. Preoperative placement of illuminated ureteral stents is highly recommended; these stents allow for continuous visual and tactile identification of the ureters within the scarred retroperitoneum, drastically reducing the risk of iatrogenic transection. Furthermore, CT Angiography (CT-A) or MR Venography (MR-V) should be obtained to map out any altered vascular anatomy, collateral vessel formation, or occult deep vein thromboses.
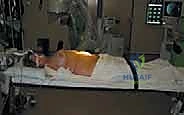
Patient positioning is critical for optimizing exposure and facilitating intraoperative fluoroscopy. The patient is positioned strictly supine on a radiolucent operating table. To maximize access to the disc spaces, we routinely utilize a motorized table break or position an inflatable radiolucent bolster transversely beneath the patient's lumbar lordosis. This maneuver essentially "cracks open" the anterior disc spaces, placing the anterior longitudinal ligament under tension and facilitating the insertion of larger interbody grafts. The patient's arms are extended and secured on arm boards at 90 degrees to permit unimpeded access for the C-arm fluoroscopy unit. Wide sterile draping is essential, exposing the abdomen from the xiphoid process down to the pubic symphysis, and laterally to the mid-axillary lines, allowing for extension of the incision if catastrophic vascular injury necessitates emergent laparotomy.

Step-by-Step Surgical Approach and Fixation Technique
The surgical execution demands meticulous tissue handling, precise anatomical recognition, and unyielding patience. We typically employ a left-sided, retroperitoneal approach.
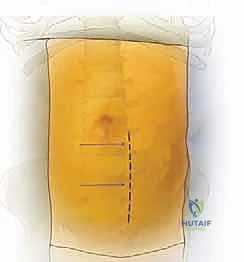
The skin incision is planned based on the target levels. For an isolated L5-S1 exposure, a transverse Pfannenstiel incision or a low oblique incision parallel to the inguinal ligament is highly cosmetic and provides excellent access. For L4-5 or multi-level exposures (e.g., L3-S1), a longitudinal paramedian incision, located approximately 3 to 5 centimeters to the left of the midline, offers superior extensibility.
Following the skin incision, dissection proceeds through the subcutaneous fat (Camper's and Scarpa's fasciae) to expose the anterior rectus sheath. The anterior rectus sheath is incised vertically. The medial border of the rectus abdominis muscle is identified, and the muscle belly is gently retracted laterally. This exposes the posterior rectus sheath (if above the arcuate line) or the transversalis fascia (if below the arcuate line). The transversalis fascia is carefully incised, revealing the delicate, underlying extraperitoneal fat and the thin, translucent peritoneal sac.
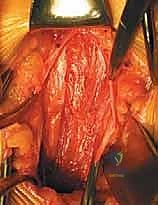
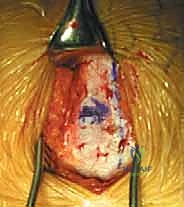
The critical phase of retroperitoneal mobilization now begins. Using blunt dissection with a combination of sweeping finger movements and moist laparotomy sponges, the peritoneal sac is gently mobilized off the undersurface of the transversalis fascia and the lateral abdominal wall. The dissection proceeds laterally and posteriorly until the psoas major muscle is encountered. The ureter must be identified adhering to the deep surface of the mobilized peritoneum and swept safely medially across the midline.
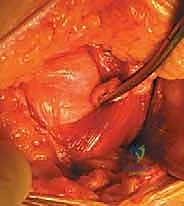
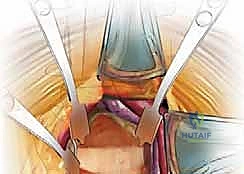
Once the psoas muscle is identified, the dissection is directed medially over the anterior aspect of the spine. The great vessels are sequentially identified. For L5-S1, the bifurcation of the aorta and IVC is located. The middle sacral artery and vein are isolated, ligated with heavy silk ties, and divided. The left common iliac vein is gently retracted laterally, exposing the L5-S1 disc space.
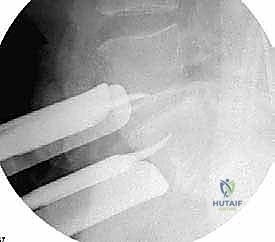
For L4-5 exposure, the mobilization is significantly more complex. The left common iliac artery and vein must be mobilized from left to right across the midline. This absolutely requires the identification and ligation of the iliolumbar vein. The left common iliac vein is gently rolled medially; the iliolumbar vein is identified under tension, branching laterally. It is meticulously skeletonized, double-ligated with non-absorbable suture, and transected. Only then can the great vessels be safely retracted to the right using specialized radiolucent retractor blades (e.g., SynFrame or Omni-Tract) secured to the table.