Thumb-in-Palm Deformity Correction: An Intraoperative Masterclass
Key Takeaway
This masterclass guides fellows through the comprehensive surgical correction of thumb-in-palm deformity in cerebral palsy. We meticulously cover patient selection, detailed anatomy, precise intraoperative techniques for tendon releases and transfers, joint stabilization, and critical postoperative management. Emphasis is placed on neurovascular protection, tensioning, and avoiding common pitfalls to restore function and improve hygiene for these complex patients.
Comprehensive Introduction and Patho-Epidemiology
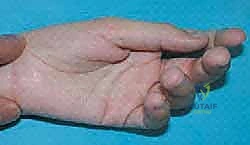
The thumb-in-palm deformity represents one of the most functionally debilitating upper extremity manifestations of spastic cerebral palsy (CP). As orthopedic surgeons, we recognize that the human hand's evolutionary advantage is predicated upon the opposable thumb, which accounts for approximately 40% to 50% of global hand function. When an upper motor neuron lesion disrupts the delicate balance of the pediatric upper extremity, the resultant spasticity disproportionately affects the flexor-pronator-adductor muscle groups. This predictable pattern of hypertonicity overwhelms the weaker, often paretic, extensor-supinator-abductor groups, driving the thumb into a tightly adducted and flexed posture within the palm. This is not merely a cosmetic anomaly; it constitutes a profound functional barrier, impeding grasp, precluding release, and severely compromising palmar hygiene.
The pathogenesis of this deformity is a dynamic continuum. In the infant and toddler, the posture is typically flexible, driven purely by dynamic spasticity and aberrant motor firing patterns. However, if left unchecked, the persistent hypertonia induces structural adaptations within the myotendinous units. Sarcomeres are lost, collagen is deposited, and the dynamic spasticity inexorably transitions into a fixed myostatic contracture. Secondary joint contractures, capsular tightening, and even osseous deformities of the first web space follow. Therefore, our surgical intervention must be precisely timed—ideally after the central nervous system has matured enough to permit postoperative rehabilitation compliance (typically between 5 and 7 years of age), but before irreversible arthrosis and severe fixed skeletal deformities manifest.
To systematically approach this complex deformity, we rely on established classification systems, most notably those described by House and Tonkin. The House classification categorizes the deformity into four types based on the predominant deforming forces: Type I (simple metacarpal adduction), Type II (metacarpal adduction with metacarpophalangeal [MCP] flexion), Type III (metacarpal adduction with MCP hyperextension or instability), and Type IV (metacarpal adduction with MCP and interphalangeal [IP] flexion). The Tonkin classification further refines this by explicitly delineating intrinsic versus extrinsic muscle dominance. Understanding these classifications is not an academic exercise; it is the absolute foundation of our surgical blueprint, dictating whether we must focus on the deep intrinsics, the extrinsic flexors, or a combination thereof.
Epidemiologically, the thumb-in-palm deformity is highly prevalent in spastic hemiplegic and quadriplegic CP subtypes. The functional impact extends beyond the hand itself; a child with a severely adducted thumb will often abandon the use of the affected limb entirely, leading to learned non-use and compounding the developmental deficit. As we approach this intraoperative masterclass, it is imperative to remember that our surgical objective is not to create a neurologically normal hand—an impossibility given the central etiology—but rather to rebalance the peripheral biomechanics to optimize the patient's existing motor control, thereby elevating their independence and quality of life.
Detailed Surgical Anatomy and Biomechanics
A profound mastery of the first web space and thenar cone anatomy is non-negotiable for the successful execution of this procedure. The deforming forces in a thumb-in-palm deformity are multifaceted, involving a complex interplay of intrinsic and extrinsic myotendinous units. The primary culprit in almost all cases is the Adductor Pollicis (AP) muscle. The AP is a massive, fan-shaped muscle comprising two distinct heads: the transverse head, originating from the palmar ridge of the third metacarpal, and the oblique head, originating from the capitate, trapezoid, and the bases of the second and third metacarpals. Both heads converge to insert onto the ulnar sesamoid and the ulnar base of the proximal phalanx. In the spastic hand, the AP exerts a relentless adduction force, obliterating the first web space and pulling the first metacarpal toward the third.
Working synergistically with the AP is the First Dorsal Interosseous (FDI) muscle, which originates from the adjacent shafts of the first and second metacarpals. While its primary role is index finger abduction, severe spasticity of the FDI severely limits the abduction of the first metacarpal, contributing significantly to the narrowing of the web space angle. Furthermore, the Flexor Pollicis Brevis (FPB), particularly its deep head, contributes to the flexion posture at the MCP joint. These intrinsic muscles are primarily innervated by the deep motor branch of the ulnar nerve (with the superficial head of the FPB innervated by the median nerve), making the deep palmar dissection a zone of high anatomical hazard.
Extrinsically, the Flexor Pollicis Longus (FPL) plays a critical role, particularly in House Type IV deformities. The FPL originates from the anterior radius and interosseous membrane, traversing the carpal tunnel to insert on the distal phalanx of the thumb. Spasticity of the FPL drives severe IP joint flexion, burying the distal phalanx into the palm. Conversely, the extrinsic extensors and abductors—the Extensor Pollicis Longus (EPL), Extensor Pollicis Brevis (EPB), and Abductor Pollicis Longus (APL)—are typically paretic, overstretched, and biomechanically disadvantaged. The EPL, originating from the posterior ulna and wrapping around Lister's tubercle, normally provides extension and adduction. In the spastic hand, its vector is often insufficient to overcome the flexor-adductor tone.
The biomechanics of the thumb joints themselves further complicate the clinical picture. The MCP joint of the thumb is inherently variable in its stability. In many CP patients, particularly House Type III, chronic spasticity of the intrinsics coupled with an attempt to actively extend the thumb using a paretic EPL results in a paradoxical hyperextension of the MCP joint. This occurs because the EPL force is transmitted entirely to the MCP joint when the IP joint is locked in flexion by the FPL or when the metacarpal is locked in adduction. Over time, the volar plate of the MCP joint attenuates, leading to severe instability that must be addressed surgically to provide a stable post for pinch mechanics.
Exhaustive Indications and Contraindications
Patient selection is arguably the most critical determinant of success in the surgical management of the spastic upper extremity. The decision to proceed with operative intervention for a thumb-in-palm deformity requires a meticulous synthesis of the patient's age, neurological status, functional deficits, and response to conservative measures.
Indications and Contraindications Table
| Category | Indications for Surgery | Contraindications for Surgery |
|---|---|---|
| Neurological Status | Stable, non-progressive upper motor neuron lesion (e.g., CP); adequate voluntary motor control. | Progressive neurological disorders; severe athetosis, chorea, or dystonia (unpredictable outcomes). |
| Patient Age | Typically > 5-7 years old (allows for CNS maturation, cooperation with therapy, and reliable clinical assessment). | Infancy/Toddlerhood (unless severe fixed contracture is rapidly progressing despite conservative care). |
| Physical Findings | Fixed myostatic contracture failing conservative management; dynamic spasticity severely limiting grasp/release. | Supple deformity fully manageable with orthoses and botulinum toxin; severe fixed joint arthrosis (requires salvage). |
| Sensory Function | Intact or functional stereognosis and two-point discrimination. | Profoundly insensate hand (motor correction will not yield functional use). |
| Cognitive/Social | Sufficient cognitive capacity to follow postoperative rehabilitation commands; supportive family environment. | Severe cognitive impairment precluding participation in structured postoperative therapy. |
The primary indication for surgery is a deformity that significantly impairs the functional use of the hand or severely compromises hygiene, and which has proven refractory to a comprehensive non-operative regimen. This regimen typically includes serial casting, custom thermoplastic orthoses, and targeted botulinum toxin type A injections into the adductor pollicis and FPL. When these modalities no longer provide functional benefit, or when clinical examination reveals the transition from dynamic spasticity to a fixed myostatic contracture, surgical intervention is warranted. The presence of voluntary grasp and release, even if weak or poorly coordinated, is a strong positive prognostic indicator.
Conversely, profound sensory deficits, particularly the absence of stereognosis, represent a significant relative contraindication. An insensate hand, regardless of its biomechanical alignment, will rarely be incorporated into spontaneous functional activities by the child. Furthermore, the presence of severe movement disorders such as athetosis or dystonia dramatically increases the unpredictability of tendon transfers and releases, often leading to recurrent or opposite deformities. In such cases, surgery is generally reserved for purely hygienic salvage rather than functional restoration.
Finally, the cognitive and psychosocial status of the patient and their support system cannot be overstated. The surgical procedure itself represents only a fraction of the therapeutic journey. The postoperative phase requires weeks of immobilization followed by months of rigorous, specialized occupational therapy to retrain the brain to utilize the newly rebalanced hand. If the child is cognitively unable to participate in this retraining, or if the family lacks the resources or commitment to adhere to the protocol, the surgical outcome will inevitably be compromised, and intervention should be reconsidered or delayed.
Pre-Operative Planning, Templating, and Patient Positioning
The foundation of a successful thumb-in-palm correction is laid long before the scalpel touches the skin. Preoperative planning demands a highly coordinated, multidisciplinary approach involving the orthopedic surgeon, neurologist, occupational therapist, and anesthesiologist. We begin with a rigorous clinical assessment, heavily relying on video analysis of the child performing standardized tasks (e.g., grasping blocks, releasing a ball, bringing food to the mouth). This dynamic assessment reveals the functional firing patterns and compensatory mechanisms that static examination often misses.
Diagnostic adjuncts play a pivotal role in our templating. Dynamic electromyography (EMG), while not universally available, provides invaluable data on muscle phase activity, helping us determine if a muscle (like the Flexor Carpi Ulnaris or Flexor Digitorum Superficialis) fires appropriately during the desired phase of grasp or release, making it a suitable candidate for tendon transfer. More commonly, we utilize selective diagnostic nerve blocks. By injecting a short-acting local anesthetic (e.g., 1% lidocaine) into the motor branch of the ulnar nerve or directly into the adductor pollicis muscle belly, we can temporarily abolish the spastic drive. The resulting improvement in thumb posture and active kinematics provides a highly accurate preview of the potential surgical outcome and confirms the primary deforming forces.
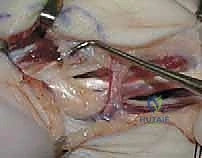
The most critical phase of preoperative planning occurs in the operating room, immediately following the induction of general anesthesia. The Examination Under Anesthesia (EUA) is mandatory. Once the patient is fully paralyzed and all dynamic spasticity is eliminated, we can definitively differentiate between reversible hypertonia and fixed myostatic contracture. We meticulously assess the passive range of motion of the CMC, MCP, and IP joints. If the thumb can be effortlessly abducted out of the palm under anesthesia, the deformity is primarily dynamic, and tendon lengthening or transfers may suffice. If the thumb remains rigidly adducted, extensive soft tissue and capsular releases are required. The EUA also allows for an unhindered assessment of MCP joint stability and volar plate integrity, dictating the need for concurrent capsulodesis or arthrodesis.
Patient positioning and setup must be optimized for meticulous microsurgical dissection and broad exposure. The patient is positioned supine with the affected upper extremity extended onto a radiolucent hand table. A well-padded pneumatic tourniquet is applied to the proximal arm to ensure a completely bloodless field, which is absolute necessity when dissecting around the delicate recurrent motor branch of the median nerve and the deep palmar arch. The arm is abducted to approximately 90 degrees, and the surgeon sits comfortably with loupe magnification (typically 3.5x to 4.5x) and a headlight. A C-arm fluoroscopy unit must be draped and readily available in the room, particularly if joint stabilization procedures (pinning or arthrodesis) are anticipated as part of the surgical plan.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution follows a strict, logical triad: Release, Augment, and Stabilize. We must first eliminate the deforming forces before we can effectively balance the thumb with tendon transfers or provide a stable skeletal platform.
Phase 1: Release of Contractures
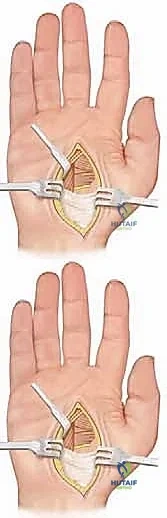
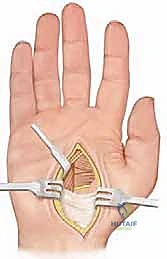
We initiate the procedure with the release of the static intrinsic contractures. A curvilinear incision is made just ulnar to, and paralleling, the thenar crease. This incision extends from the level of the distal carpal tunnel to the proximal flexion crease of the thumb.
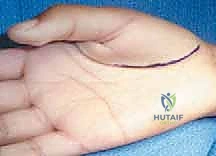
Subcutaneous dissection is performed meticulously. The paramount objective in this superficial plane is the identification and protection of the recurrent motor branch of the median nerve. This nerve, which supplies the opponens pollicis, abductor pollicis brevis, and superficial head of the FPB, is highly variable. It typically branches from the median nerve just distal to the transverse carpal ligament, but extraligamentous and transligamentous variants are common. Utilizing loupe magnification, we trace the nerve and protect it with a vessel loop. Accidental transection here is a catastrophic complication that will permanently abolish thumb opposition.

Once the neurovascular structures are secured, we retract the thenar musculature radially to expose the fascial covering of the Adductor Pollicis. We systematically release the transverse head of the AP from its origin along the third metacarpal. We then proceed deeper, identifying the deep palmar arch and the deep motor branch of the ulnar nerve, which must be protected. The oblique head of the AP is then sharply released from the capitate and trapezoid. If the first web space remains tight, we extend our dissection dorsally to release the First Dorsal Interosseous from its origin on the first metacarpal.

If the EUA revealed severe FPL spasticity contributing to IP joint flexion (House Type IV), we move to the distal forearm. Through a separate volar incision, we identify the FPL at its musculotendinous junction.
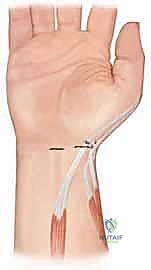
We perform a fractional lengthening by making transverse step-cuts in the tendinous aponeurosis while leaving the underlying muscle fibers intact. This allows the muscle to stretch and lengthen without completely severing the unit, preserving some active flexion power while eliminating the deforming contracture.
Phase 2: Augmentation of Paretic Muscles
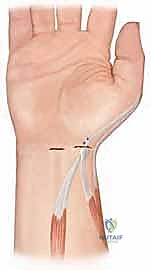
With the deforming flexor/adductor forces neutralized, we must augment the weak extensors and abductors. The most common strategy involves rerouting the Extensor Pollicis Longus (EPL) and augmenting it with a tendon transfer.
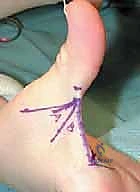
Through a small dorsal incision over the wrist, the EPL is released from the third dorsal compartment and transposed radially, out of Lister's tubercle. This subtly changes its biomechanical vector from an adductor/extensor to a pure abductor/extensor.
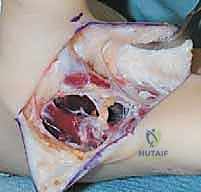
To provide power to this rerouted EPL, we frequently utilize the Brachioradialis (BR) or the Palmaris Longus (PL). The chosen donor tendon is harvested and mobilized extensively to ensure a straight line of pull.
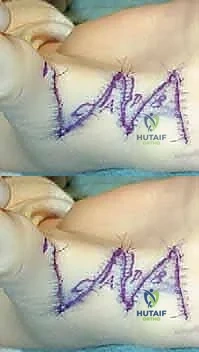
The transfer is executed using a classic Pulvertaft weave. The donor tendon is woven through the recipient EPL tendon at least three times, under appropriate tension (typically with the thumb held in full palmar abduction and extension, and the wrist in neutral). The weave is secured with multiple figure-of-eight non-absorbable sutures (e.g., 3-0 or 4-0 braided polyester), ensuring a biomechanically robust construct that can withstand early postoperative rehabilitation.
Phase 3: Joint Stabilization
The final, and often most critical, phase is addressing joint instability, particularly the paradoxical MCP hyperextension seen in House Type III deformities. If the MCP joint is supple but unstable, a volar capsulodesis is performed.
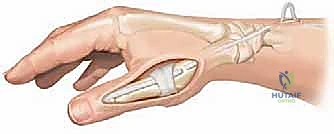
This can be achieved by tenodesing the EPB tendon to the volar base of the proximal phalanx, effectively creating a checkrein against hyperextension.
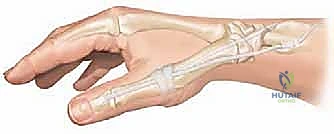
However, if the joint is severely unstable, arthritic, or if the patient is older, an MCP joint arthrodesis is the definitive solution.
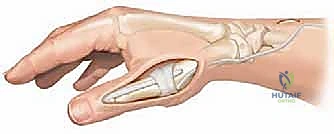
The articular cartilage of the metacarpal head and the base of the proximal phalanx is meticulously denuded down to bleeding subchondral bone using a rongeur or a small oscillating saw.
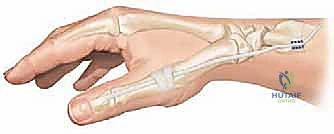
The joint is positioned in approximately 15 to 20 degrees of flexion, 10 to 15 degrees of pronation, and neutral abduction.
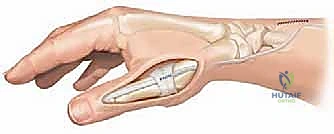
Fixation is typically achieved using crossed 0.045-inch Kirschner wires, or a dorsal tension band wiring technique if greater compression is required.
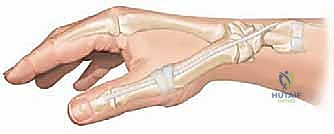
The K-wires are cut beneath the skin to prevent pin tract infections, and the incisions are closed meticulously over drains if necessary.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, the management of the spastic upper extremity is fraught with potential complications. The underlying upper motor neuron lesion remains entirely uncorrected by our peripheral interventions, meaning the central drive for spasticity persists postoperatively.
Complications and Management Table
| Complication | Estimated Incidence | Etiology / Risk Factors | Prevention and Salvage Management |
|---|---|---|---|
| Recurrence of Deformity | 10% - 20% | Ongoing spasticity; growth spurts; inadequate initial release; failure of postoperative splinting compliance. | Prevention: Aggressive initial release; nighttime splinting until skeletal maturity. Salvage: Revision release; addressing previously unrecognized extrinsic tightness (e.g., FPL). |
| Overcorrection (Swan Neck / Fixed Abduction) | 5% - 10% | Over-lengthening of FPL; excessive tension on tendon transfers; failure to stabilize the MCP joint. | Prevention: Precise tensioning of Pulvertaft weaves; mandatory MCP stabilization in House Type III. Salvage: FPL shortening; IP joint arthrodesis; release of overtightened transfers. |
| Nerve Injury (Median Motor / Ulnar Deep) | < 2% | Iatrogenic transection during thenar or deep palmar dissection; aggressive retraction. | Prevention: Mandatory use of loupe magnification; meticulous bloodless field via tourniquet. Salvage: Immediate microsurgical primary repair; secondary opposition tendon transfers (e.g., FDS ring to APB) if repair fails. |
| Nonunion of Arthrodesis | 3% - 5% | Inadequate |
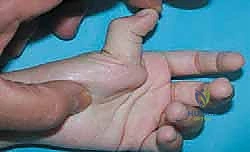
Clinical & Radiographic Imaging Archive