Scapulothoracic Bursitis: Unmasking a Mimic of Refractory Lateral Epicondylitis
Key Takeaway
Scapulothoracic bursitis diagnosis often involves a comprehensive physical exam, revealing tenderness, crepitus, and scapular dyskinesis, particularly after conservative treatments for conditions like lateral epicondylitis fail. MRI is crucial, demonstrating fluid and soft tissue thickening within the bursa, confirming the true origin of persistent pain in the scapulothoracic region.
Introduction and Epidemiology
Scapulothoracic bursitis, frequently presenting as part of the snapping scapula syndrome, is an insidious and often misdiagnosed pathology. While primary scapulothoracic symptoms are well-documented in orthopedic literature, the secondary manifestations of proximal kinetic chain dysfunction can obscure the true pain generator. The scapula serves as the foundational base for upper extremity kinematics. When periscapular mechanics are compromised—whether through structural bursitis, osseous abnormalities, or neuromuscular dyskinesis—the distal joints are forced to absorb supraphysiologic loads.
This phenomenon frequently manifests as refractory lateral epicondylitis. In overhead athletes and manual laborers, a stable scapular base is requisite for the efficient transfer of energy from the core to the distal extremity. Scapular dyskinesis, secondary to chronic scapulothoracic bursitis, leads to decreased shoulder elevation and increased glenohumeral internal rotation. To compensate during the acceleration and deceleration phases of upper extremity use, the wrist extensors (specifically the extensor carpi radialis brevis) undergo excessive eccentric loading, precipitating tendinosis.
The epidemiology of scapulothoracic bursitis demonstrates a bimodal distribution, frequently affecting young, active overhead athletes (baseball pitchers, tennis players, swimmers) and middle-aged manual laborers. The etiology is primarily categorized into osseous anomalies (osteochondromas, rib exostoses, scapular malunions) and soft-tissue derangements (primary bursitis, periscapular muscle fibrosis, interstitial tears).
Clinical Vignette and Kinetic Chain Dysfunction
The profound impact of scapulothoracic pathology on the distal kinetic chain is best illustrated through the following clinical presentation:
A 48-year-old right-hand dominant male presented with a 14-month history of progressive right lateral elbow pain. He described the pain as dull aching, exacerbated by repetitive forearm rotation, gripping, and overhead activities, and occasionally radiating distally into the forearm and proximally towards the shoulder. The pain had significantly impacted his occupational duties as a carpenter and his recreational tennis activities.
Initial diagnosis elsewhere was refractory lateral epicondylitis. He had undergone multiple conservative treatments, including activity modification, non-steroidal anti-inflammatory drugs (NSAIDs), corticosteroid injections into the lateral epicondyle, and a 6-month course of physical therapy focusing on eccentric strengthening of the wrist extensors. These interventions provided transient, partial relief at best, with symptoms consistently recurring and worsening. There was no history of acute trauma to the elbow.
Further inquiry revealed a remote history (approximately 5 years prior) of a fall directly onto his right shoulder, which resulted in significant bruising but no documented fracture. Since then, he reported intermittent, mild discomfort along the medial border of his right scapula, often described as a "grinding" sensation during specific movements, which he attributed to muscle stiffness and had largely ignored due to the more dominant elbow pain. He denied any overt neurological symptoms such as numbness, tingling, or weakness in the hand or arm. He was a non-smoker, with no significant medical comorbidities.
Clinical Examination Findings
On static examination, the patient demonstrated subtle right shoulder droop and mild scapular protraction and abduction. During dynamic evaluation, a discernible dyskinesis of the right scapula was observed. Specifically, a type II scapular dyskinesis (inferior medial border prominence) was noted during the eccentric phase of arm elevation and a subtle increase in internal rotation of the scapula. No overt muscle atrophy was visible. No skin changes, erythema, or gross swelling were apparent around the elbow or shoulder.
Localized tenderness was present over the lateral epicondyle, consistent with prior diagnoses. However, a more critical finding on thorough examination was significant tenderness upon deep palpation along the superomedial aspect of the scapular border and over the scapulothoracic interval. Palpable crepitus was elicited with passive scapular manipulation, particularly during protraction and retraction. The muscles of the shoulder girdle, notably the rhomboids and trapezius, exhibited diffuse tenderness and spasm. No palpable masses or bony abnormalities were detected.
Elbow active and passive range of motion was full and pain-free. Resisted wrist extension and resisted supination elicited significant pain at the lateral epicondyle. Shoulder active and passive glenohumeral range of motion was full and pain-free. However, active elevation beyond 90 degrees was accompanied by compensatory scapular motion and a subtle "hitch" in the scapulohumeral rhythm, indicative of scapular dyskinesis.
Specific tests for scapular stability and control revealed deficits. The Scapular Assistance Test (SAT) provided partial relief of pain during active shoulder abduction, suggesting compromised scapular kinematics. A positive Wall Push-up Test demonstrated increased right scapular winging. Kibler's lateral slide test showed a significant difference ( >1.5 cm) in the resting position of the inferior angle of the scapula relative to the thoracic spine compared to the contralateral side. Distal neurovascular status of the right upper extremity was intact. Sensation to light touch was normal throughout the radial, ulnar, and median nerve distributions. Motor strength was 5/5 in all major muscle groups.
Surgical Anatomy and Biomechanics
The scapulothoracic articulation is not a true synovial joint but rather a complex fascial and muscular gliding plane. Understanding the intricate layering of this space is paramount for safe surgical intervention and for comprehending the biomechanical failures that lead to distal tendinopathies.
Osteology and Articular Configuration
The scapula is a flat, triangular bone that articulates with the posterior thoracic cage, typically spanning from the second to the seventh rib. The anterior (costal) surface is concave, forming the subscapular fossa, which mirrors the convex contour of the posterior thoracic wall. The superomedial angle of the scapula is a critical anatomic landmark; it is the most common site of osseous impingement and bursal inflammation. The anterior angulation of the superomedial border typically ranges from 124 to 162 degrees. An excessively anteriorly hooked superomedial angle (Luschka’s tubercle) significantly decreases the clearance between the scapula and the thoracic cage, predisposing the patient to mechanical crepitus and bursitis.
Musculature and Neurovascular Relations
The scapulothoracic space is delineated by distinct muscular layers and fascial planes.
1. Superficial Layer: Comprises the trapezius and latissimus dorsi.
2. Intermediate Layer: Contains the rhomboid major, rhomboid minor, and levator scapulae.
3. Deep Layer: Consists of the serratus anterior and subscapularis.
The primary anatomic spaces relevant to scapulothoracic bursitis are the supraserratus bursa (located between the subscapularis and serratus anterior) and the infraserratus bursa (located between the serratus anterior and the posterolateral chest wall). Two additional adventitial bursae may form at the superomedial angle and the inferior pole in response to chronic friction.
The neurovascular anatomy dictates the safe zones for surgical approach:
* Spinal Accessory Nerve (Cranial Nerve XI): Courses deep to the trapezius muscle.
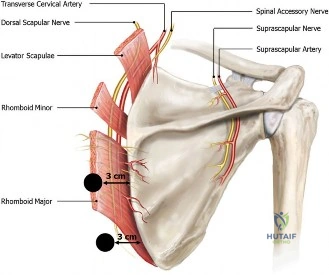
* Dorsal Scapular Nerve and Artery: Descend along the medial border of the scapula, deep to the rhomboids. The dorsal scapular nerve is typically located 1 to 2 cm medial to the medial border of the scapula. Surgical dissection or portal placement must remain lateral to this neurovascular bundle to prevent denervation of the rhomboids and levator scapulae.
* Long Thoracic Nerve: Courses vertically along the lateral chest wall, superficial to the serratus anterior.
Biomechanical Implications for the Distal Extremity
The scapula provides dynamic stability through a highly coordinated force couple system (trapezius and serratus anterior). Normal scapulothoracic kinematics require the scapula to upwardly rotate, posteriorly tilt, and externally rotate during arm elevation.
When scapulothoracic bursitis induces pain, reflex inhibition of the lower trapezius and serratus anterior occurs, leading to unopposed action of the pectoralis minor and upper trapezius. This results in an anteriorly tilted, internally rotated, and protracted scapula (Type I or Type II dyskinesis). During repetitive overhead activities or manual labor, this protracted scapular position alters the glenohumeral resting alignment, functionally decreasing internal rotation. To achieve the necessary terminal velocity and positioning of the hand, the distal kinetic chain compensates. The elbow experiences increased valgus stress, and the extensor carpi radialis brevis (ECRB) is subjected to massive eccentric loads to stabilize the wrist during the acceleration phase. Over time, this repetitive microtrauma to the ECRB origin manifests as refractory lateral epicondylitis.
Indications and Contraindications
Surgical intervention for scapulothoracic bursitis is strictly reserved for patients who have failed exhaustive non-operative management. A minimum of 6 to 12 months of conservative therapy—including periscapular strengthening, postural re-education, NSAIDs, and targeted fluoroscopic or ultrasound-guided corticosteroid injections into the scapulothoracic bursae—must be documented.
When conservative measures fail, and the primary pain generator is confirmed to be the scapulothoracic space (often validated by temporary complete relief following a diagnostic local anesthetic injection into the bursa), surgical management is indicated.
| Indication Category | Specific Operative Indications | Non-Operative Indications (Conservative Management) |
|---|---|---|
| Chronicity | Refractory symptoms > 6-12 months | Acute or subacute onset (< 6 months) |
| Structural Pathology | Osteochondroma, rib exostosis, malunion | Absence of overt osseous deformity |
| Bursal Pathology | Fibrotic, hypertrophic bursitis confirmed on MRI | Mild bursal fluid without architectural distortion |
| Diagnostic Response | Positive response to diagnostic bursal injection | Negative or equivocal response to diagnostic injection |
| Kinetic Chain | Distal tendinopathy driven by proximal structural impingement | Purely functional dyskinesis correctable with therapy |
Surgical contraindications include active local or systemic infection, cervical radiculopathy mimicking scapular pain (e.g., C5-C6 radiculopathy), and purely functional scapular dyskinesis resulting from generalized ligamentous laxity or isolated nerve palsy (e.g., long thoracic nerve palsy causing winging) without structural bursal pathology.
Pre Operative Planning and Patient Positioning
Thorough preoperative planning requires an exact understanding of the osseous and soft-tissue morphology.
Imaging Modalities
Standard radiographs should include a true anteroposterior (AP) view of the scapula, a lateral (Y) view, and an axillary view. These are primarily utilized to rule out gross osseous deformities or large osteochondromas.
Computed Tomography (CT) with 3D reconstruction is highly recommended when osseous impingement is suspected. It allows precise quantification of the anterior hook of the superomedial angle (Luschka's tubercle) and evaluates the congruity between the anterior scapula and the posterior thoracic wall.
Magnetic Resonance Imaging (MRI) is the gold standard for evaluating soft-tissue pathology. T2-weighted fluid-sensitive sequences (STIR or fat-suppressed T2) will demonstrate fluid distension within the supraserratus or infraserratus bursae. MRI is also critical for ruling out soft-tissue sarcomas or elastofibroma dorsi, which frequently present at the inferior pole of the scapula and can mimic bursitis.

Anesthesia and Positioning Setup
General anesthesia with endotracheal intubation is mandatory. Muscle relaxation is critical to allow adequate distraction of the scapula from the thoracic wall.
The patient is placed in the prone position on a radiolucent Jackson table with chest rolls to ensure free abdominal excursion. The operative extremity is placed in the "chicken wing" position. The arm is internally rotated, and the dorsum of the hand is rested securely against the lumbar spine. This specific positioning is paramount; it maximally retracts the scapula laterally and elevates the medial border away from the posterior chest wall, significantly increasing the working volume of the scapulothoracic space and minimizing the risk of iatrogenic pleural puncture.
The entire hemithorax, shoulder, and upper extremity are prepped and draped freely to allow intraoperative manipulation of the arm if dynamic assessment is required.
Detailed Surgical Approach and Technique
While open partial scapulectomy and bursectomy remain viable options, arthroscopic scapulothoracic decompression has emerged as the gold standard due to decreased morbidity, superior visualization of the bursal spaces, and accelerated rehabilitation.
Portal Placement and Trajectory
Precise portal placement is critical to avoid the dorsal scapular nerve and artery. The medial border of the scapula and the spine of the scapula are palpated and marked.
Two primary portals are utilized, established by Bell et al.:
1. Superior Portal: Located 3 to 4 cm medial to the medial border of the scapula, at the level of the scapular spine.
2. Inferior Portal: Located 3 to 4 cm medial to the medial border of the scapula, approximately 3 to 4 cm inferior to the superior portal.
A spinal needle is introduced at the superior portal site, angled laterally beneath the scapula. The trajectory must be strictly horizontal or slightly superior to avoid intercostal penetration. Once the needle touches the undersurface of the scapula, 20 to 30 cc of normal saline is injected to distend the space.
Arthroscopic Bursectomy and Resection
A standard 4.0-mm, 30-degree or 70-degree arthroscope is utilized. The 70-degree scope is highly advantageous for visualizing the extreme superomedial angle.
- Fluid Management: The scapulothoracic space lacks a true confining capsule. Extravasation of fluid into the thoracic cavity or fascial planes is a severe risk. Pump pressure must be maintained strictly below 40 mmHg, and the procedure should be completed as expeditiously as possible. Gravity flow is often preferred over automated pumps.
- Creation of Working Space: The arthroscope is introduced through the superior portal. A blunt trocar is used to sweep the areolar tissue. The inferior portal is established under direct intra-articular visualization.
- Bursectomy: An arthroscopic shaver and radiofrequency ablation wand are introduced through the inferior portal. The hypertrophic bursal tissue within the supraserratus and infraserratus spaces is meticulously resected. The fascial boundaries of the serratus anterior and subscapularis must be identified and preserved.
- Bony Resection (Partial Scapulectomy): If an osseous prominence or a hooked superomedial angle is identified as the mechanical trigger, a partial scapulectomy is performed. The periosteum on the undersurface of the superomedial angle is elevated using a radiofrequency wand. An arthroscopic burr or an osteotome (introduced via an accessory portal) is used to resect the superomedial angle. Typically, a triangular wedge measuring 2 cm horizontally and 2 cm vertically is removed. The resection must be smooth and beveled to prevent secondary impingement.
- Dynamic Assessment: The arm is taken out of the "chicken wing" position and moved through a full range of motion while visualizing the resected area arthroscopically to ensure complete clearance and absence of residual crepitus.

Following thorough irrigation, the portals are closed with non-absorbable sutures. No drains are typically required unless extensive open resection was performed.
Complications and Management
Arthroscopic scapulothoracic decompression is technically demanding. The proximity to the pleural cavity and major neurovascular structures necessitates meticulous technique.
| Complication | Incidence | Etiology / Risk Factors | Salvage Strategy / Management |
|---|---|---|---|
| Fluid Extravasation / Compartment Syndrome | Moderate | High pump pressure (>40 mmHg), prolonged surgical time | Immediate cessation of fluid, diuresis, rarely requires fasciotomy of the periscapular musculature. |
| Pneumothorax | Low (<1%) | Medial or deep trajectory of trocar/shaver, poor patient positioning | Intraoperative recognition, immediate chest tube thoracostomy, postoperative chest radiograph. |
| Dorsal Scapular Nerve Injury | Low | Portals placed < 3 cm from the medial border, aggressive medial resection | Observation; if complete transection is suspected on postoperative EMG, nerve grafting or tendon transfer may be required. |
| Postoperative Hematoma | Moderate | Inadequate hemostasis of the resected bone bed | Compressive dressings, observation. Rarely requires surgical evacuation unless causing skin necrosis or nerve compression. |
| Recurrence of Symptoms | 10-15% | Inadequate bony resection, failure to address the distal kinetic chain (elbow) | Revision arthroscopy vs. open resection. Comprehensive postoperative kinetic chain rehabilitation. |
Post Operative Rehabilitation Protocols
The postoperative rehabilitation protocol is vital not only for the recovery of the scapulothoracic articulation but also for the resolution of the secondary distal pathologies, such as the refractory lateral epicondylitis described in the clinical vignette.
Phase I: Protection and Tissue Healing (Weeks 0-2)
- The upper extremity is immobilized in a sling for comfort, typically for 7 to 10 days.
- Immediate initiation of active and active-assisted range of motion (ROM) of the elbow, wrist, and hand to prevent stiffness and begin mobilizing the extensor carpi radialis brevis.
- Passive glenohumeral ROM is permitted within a pain-free arc.
- Cryotherapy and strict adherence to lifting restrictions (no lifting > 1 lb).
Phase II: Scapular Mobilization and Activation (Weeks 2-6)
- Discontinue sling.
- Initiate active-assisted and active ROM of the shoulder.
- Focus on closed-kinetic chain exercises to promote scapular stabilization (e.g., wall slides, table washboards).
- Begin submaximal isometric strengthening of the periscapular musculature (serratus anterior, middle/lower trapezius, rhomboids).
- Concurrently, initiate gentle eccentric stretching of the wrist extensors. The resolution of scapular pain allows the patient to perform these exercises without proximal inhibition.
Phase III: Kinetic Chain Integration and Strengthening (Weeks 6-12)
- Progress to open-kinetic chain strengthening.
- Implement plyometric periscapular exercises.
- Focus heavily on the integration of the core, scapula, and elbow. Exercises such as PNF (Proprioceptive Neuromuscular Facilitation) D1/D2 patterns are utilized to re-establish the normal sequence of muscle activation from the proximal base to the distal effector.
- Aggressive eccentric strengthening of the lateral epicondyle musculature is now highly effective, as the proximal biomechanical fault has been corrected.
Phase IV: Return to Play / Heavy Labor (Weeks 12+)
- Work-specific or sport-specific functional training.
- Overhead athletes begin a structured interval throwing or serving program.
- Full return to activity is permitted when the patient demonstrates symmetrical scapular kinematics, 5/5 periscapular strength, and complete resolution of lateral epicondyle pain during provocative testing.
Summary of Key Literature and Guidelines
The paradigm shift towards recognizing proximal kinetic chain deficits as the primary etiology for distal tendinopathies is well-supported in contemporary orthopedic literature. Kibler et al. established the foundational framework for evaluating scapular dyskinesis, demonstrating that abnormal scapular kinematics significantly alter the loads transmitted to the elbow and wrist.
Regarding surgical management, Bell and colleagues pioneered the arthroscopic approach to the scapulothoracic space, establishing the safe portal placements and anatomic landmarks that are utilized today. Their studies demonstrated that arthroscopic bursectomy and partial scapulectomy yield excellent functional outcomes with significantly less morbidity compared to open procedures.
Manske et al. further defined the clinical entity of snapping scapula syndrome, emphasizing the necessity of a structured, non-operative rehabilitation program focusing on the serratus anterior and lower trapezius before considering surgical intervention. Recent systematic reviews confirm that when conservative measures fail in cases of structural impingement, arthroscopic superomedial angle resection provides >85% patient satisfaction and reliable return to pre-injury occupational and athletic activities.
Ultimately, the successful management of complex upper extremity pain syndromes, such as refractory lateral epicondylitis mimicking isolated elbow pathology, demands a holistic evaluation of the entire kinetic chain. Surgical correction of the scapulothoracic articulation, followed by comprehensive rehabilitation, restores the biomechanical foundation necessary for distal joint health.
