Mastering Revision Total Knee Arthroplasty: Tibial Bone Loss & Advanced Grafting Techniques
Key Takeaway
Step into the OR for a detailed masterclass on revision total knee arthroplasty with tibial bone loss. This guide provides an exhaustive, real-time breakdown of surgical techniques, from meticulous dissection and defect assessment to advanced impaction and structural bone grafting. Learn to manage complex bone deficiencies and achieve stable, long-lasting implant fixation, ensuring optimal patient outcomes through expert execution and comprehensive postoperative care.
Comprehensive Introduction and Patho-Epidemiology
The management of a failed total knee arthroplasty (TKA) complicated by massive tibial bone loss represents one of the most formidable challenges in adult reconstructive orthopedic surgery. As the volume of primary TKAs performed globally continues to rise exponentially, the corresponding burden of revision procedures is increasing at an even steeper trajectory. This is not merely a procedure of component extraction and exchange; it is a complex reconstructive endeavor that demands meticulous preoperative planning, advanced surgical technique, and a profound understanding of joint biomechanics. The primary goals of revision TKA are the eradication of pain, the restoration of a stable and functional joint, the re-establishment of the anatomical joint line, and the achievement of durable, long-term implant fixation. When significant tibial bone stock is compromised, achieving these goals requires a sophisticated armamentarium of reconstruction strategies, ranging from morselized autograft to highly porous metaphyseal cones and structural allografts.
The pathogenesis of periprosthetic bone loss is multifactorial, with aseptic loosening and particle-induced osteolysis remaining the most prevalent culprits in late-term failures. Polyethylene wear debris, generated at the articular interface or from backside wear, infiltrates the effective joint space. This particulate debris is phagocytosed by macrophages, triggering a fierce biological cascade. The subsequent release of pro-inflammatory cytokines, including tumor necrosis factor-alpha (TNF-α), interleukin-1 (IL-1), and interleukin-6 (IL-6), stimulates osteoclastic bone resorption while simultaneously inhibiting osteoblastic bone formation. This results in progressive, often asymptomatic, cavitary and segmental bone defects that can severely compromise the structural integrity of the proximal tibia. Furthermore, stress shielding induced by rigid metallic implants can lead to localized osteopenia, exacerbating bone loss in the proximal metaphysis.

Beyond aseptic mechanisms, periprosthetic joint infection (PJI) is a critical and devastating cause of rapid bone destruction. The inflammatory response to bacterial pathogens, combined with the formation of a resilient biofilm on the implant surface, necessitates aggressive surgical debridement and often a two-stage exchange arthroplasty. Implant malalignment and instability can also contribute to accelerated wear and subsequent osteolysis due to eccentric loading of the polyethylene bearing. Finally, the iatrogenic bone loss that inevitably occurs during the extraction of well-fixed primary components, particularly when removing cemented stems or porous-coated metaphyseal sleeves, must be factored into the overall defect management strategy.
Clinical evaluation of these patients requires a high index of suspicion and a systematic approach. The patient's history is paramount; distinguishing between mechanical failure and infectious etiologies is the foundational step. "Start-up pain" that improves with ambulation often points toward mechanical loosening or mild synovitis, whereas relentless, progressive pain—especially pain at rest or night pain—is a glaring red flag for infection. Patients may also report a sensation of instability, describing the knee as "giving way," which may indicate progressive bone loss leading to ligamentous laxity or catastrophic component subsidence. A comprehensive physical examination must assess gait mechanics, range of motion, fixed deformities, and multiplanar stability. Crucially, the neurovascular status must be documented meticulously, as subsequent revision surgery carries an increased risk of ischemic or neuropathic complications.
Detailed Surgical Anatomy and Biomechanics
A profound mastery of the surgical anatomy of the knee is non-negotiable when undertaking revision TKA, particularly in the setting of distorted tissue planes and significant bone loss. The osteology of the proximal tibia dictates our reconstructive options. The proximal tibia is characterized by a dense metaphyseal flare that transitions into a narrower diaphyseal isthmus. In primary TKA, fixation relies heavily on the strong cancellous bone of the proximal metaphysis. However, in revision scenarios, this metaphyseal bone is often deficient, osteolytic, or structurally compromised. The most common areas of severe deficiency are the medial tibial plateau, due to chronic varus overloading, and the posterolateral corner. Understanding the geometry of the tibial medullary canal is critical for the utilization of diaphyseal stems, which are essential for bypassing metaphyseal defects and transferring loads to the intact cortical bone of the diaphysis.
The neurovascular anatomy of the posterior and lateral knee requires vigilant protection throughout the procedure. The popliteal artery and vein lie directly posterior to the posterior capsule, separated only by a thin layer of adipose tissue. In the setting of a contracted, scarred posterior capsule, aggressive soft tissue releases or the errant placement of retractors (such as a posterior Hohmann) can lead to catastrophic vascular injury. Similarly, the common peroneal nerve is highly vulnerable as it courses laterally around the fibular neck. Extensive posterolateral dissection, manipulation of the fibular head to manage massive lateral bone defects, or the correction of a severe fixed valgus deformity places this nerve at significant risk for traction neuropraxia or direct transection, resulting in a devastating foot drop.
The muscular intervals and soft tissue envelope present their own unique challenges. The extensor mechanism is the lifeline of the knee; its integrity must be preserved at all costs. The patellar tendon is particularly susceptible to avulsion from the tibial tubercle during exposure of a stiff, ankylosed joint. The medial collateral ligament (MCL) is often encased in scar tissue. In revision surgery, it is frequently necessary to elevate the MCL circumferentially from the proximal tibial metaphysis as a continuous subperiosteal sleeve. This "sleeve technique" allows for adequate exposure of the joint, facilitates component extraction, and permits the restoration of the joint line without compromising the ligament's origin or insertion, provided it is meticulously repaired or balanced later in the procedure.
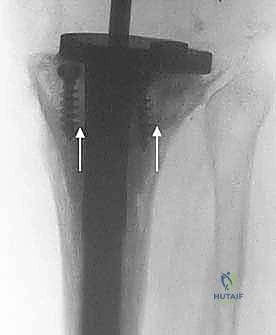
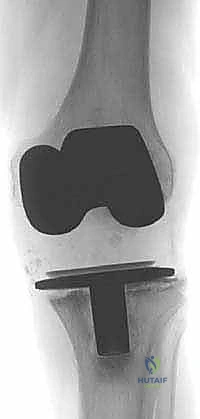
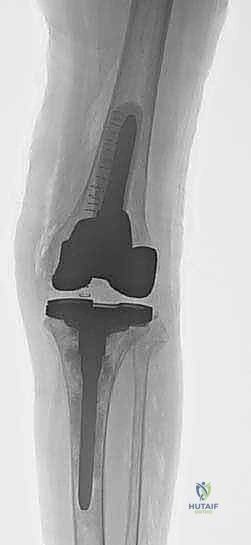
Biomechanically, the revision construct must adhere to the principles of "zonal fixation." Morgan-Jones et al. described three zones of the knee: Zone 1 (the epiphysis/joint surface), Zone 2 (the metaphysis), and Zone 3 (the diaphysis). To achieve a durable reconstruction, rigid fixation must be obtained in at least two of these three zones. When Zone 1 and Zone 2 are compromised by massive bone loss, the surgeon must rely heavily on Zone 3. This is achieved through the use of diaphyseal engaging stems. These stems bypass the deficient metaphyseal bone and offload stress from the joint line to the diaphyseal cortex. The choice between cemented and cementless (press-fit) stems, as well as straight versus offset stems, depends on the canal geometry and the need to restore the anatomical mechanical axis while accommodating the specific location of the bone defect.
Exhaustive Indications and Contraindications
The decision to proceed with a revision TKA involving complex bone grafting or augmentation is dictated by a careful analysis of indications and absolute contraindications. Absolute indications include aseptic loosening with progressive symptomatic subsidence, massive particle-induced osteolysis threatening the structural integrity of the joint, periprosthetic joint infection (typically managed via a two-stage exchange utilizing an antibiotic-impregnated cement spacer), and periprosthetic fractures around the tibial component that cannot be managed with internal fixation alone. Instability due to asymmetric bone loss that cannot be corrected with thicker polyethylene inserts also warrants revision of the metallic components to restore the joint line and balance the collateral ligaments.
Contraindications must be rigorously respected to prevent catastrophic outcomes. Absolute contraindications for a single-stage revision include active, uncontrolled periprosthetic joint infection, particularly with highly virulent or multidrug-resistant organisms. In such cases, a two-stage or even three-stage approach is mandatory. Severe medical comorbidities (ASA Class IV or V) that render the patient unfit for prolonged anesthesia and massive fluid shifts represent a strict contraindication. Furthermore, a severely compromised soft tissue envelope—characterized by adherent scarring, active sinus tracts, or inadequate skin coverage—must be addressed prior to or concurrently with the arthroplasty, often requiring the expertise of a plastic surgeon for local or free flap coverage (e.g., medial gastrocnemius flap).
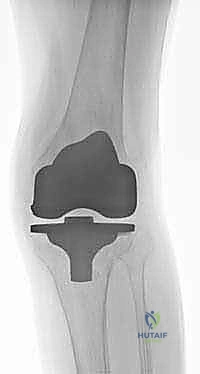
The selection of the specific reconstruction technique is highly dependent on the magnitude and morphology of the bone defect. The Anderson Orthopaedic Research Institute (AORI) classification is the gold standard for guiding these decisions. AORI Type I defects (intact metaphyseal bone) can often be managed with cement alone or morselized allograft. AORI Type II defects (damaged metaphyseal bone requiring augmentation) typically necessitate modular metallic augments (wedges or blocks) and diaphyseal stems. AORI Type III defects (deficient metaphyseal bone compromising the entire plateau or tubercle) represent the most severe challenge, mandating the use of structural allografts, highly porous metaphyseal cones, or custom-flanged endoprostheses to restore the structural foundation of the tibia.
| Indication / Contraindication Category | Specific Condition | Management Strategy / Rationale |
|---|---|---|
| Absolute Indication | Aseptic Loosening with Subsidence | Revision with diaphyseal stem fixation and metaphyseal augmentation. |
| Absolute Indication | Massive Osteolysis (AORI Type II/III) | Debridement of lytic membrane; reconstruction with porous cones or structural allograft. |
| Absolute Indication | Periprosthetic Joint Infection (PJI) | Two-stage exchange: Explant, antibiotic spacer, systemic antibiotics, followed by reimplantation. |
| Relative Indication | Global Instability / Component Malposition | Revision to a higher constraint implant (CCK or Hinge) with joint line restoration. |
| Absolute Contraindication | Active, Uncontrolled Systemic Sepsis | Life over limb; requires immediate medical stabilization and source control (I&D). |
| Absolute Contraindication | Inadequate Soft Tissue Envelope | Requires plastic surgery consultation for flap coverage prior to or during arthroplasty. |
| Relative Contraindication | Severe Neurologic Deficit (e.g., Charcot) | High risk of failure; may require arthrodesis or highly constrained hinged prosthesis. |
Pre-Operative Planning, Templating, and Patient Positioning
The success of a complex revision TKA is largely determined before the patient ever enters the operating room. A rigorous preoperative workup is mandatory, beginning with a definitive exclusion of infection. Every patient must undergo serological testing, including a Complete Blood Count (CBC), Erythrocyte Sedimentation Rate (ESR), and C-Reactive Protein (CRP). Regardless of serology, a preoperative knee aspiration is the gold standard. Synovial fluid must be analyzed for cell count with differential, aerobic and anaerobic cultures, and ideally, advanced biomarkers such as alpha-defensin or synovial CRP. If the aspiration is dry, a saline lavage may be attempted, or the surgeon must rely on intraoperative frozen sections to quantify polymorphonuclear leukocytes (PMNs) per high-power field before proceeding with reimplantation.
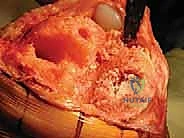
Imaging is the cornerstone of defect classification and templating. High-quality, weight-bearing anteroposterior (AP), lateral, and skyline patellar radiographs are the minimum requirement. The surgeon must meticulously scrutinize the implant-bone interface for radiolucent lines, assess the degree of component subsidence, and evaluate the overall mechanical alignment. For complex revisions involving suspected AORI Type II or III defects, a multi-detector Computed Tomography (CT) scan with metal artifact reduction sequence (MARS) is invaluable. The CT scan provides a three-dimensional map of the remaining bone stock, delineating contained cavitary defects from uncontained segmental bone loss, and allows for precise preoperative sizing of metaphyseal cones or sleeves.
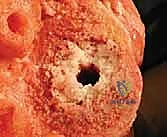
Digital templating is a critical exercise. The primary goal is the anatomic restoration of the joint line, which typically resides 20 to 25 millimeters distal to the lateral femoral epicondyle and approximately 10 to 14 millimeters proximal to the tip of the fibular head. The surgeon must template the anticipated size of the tibial component, the length and diameter of the diaphyseal stem required to bypass the defect by at least two cortical diameters, and the size of the metaphyseal augments or cones needed to fill the void. This templating process ensures that the hospital inventory is adequately stocked with the necessary revision systems, structural allografts, and backup options, minimizing intraoperative delays.
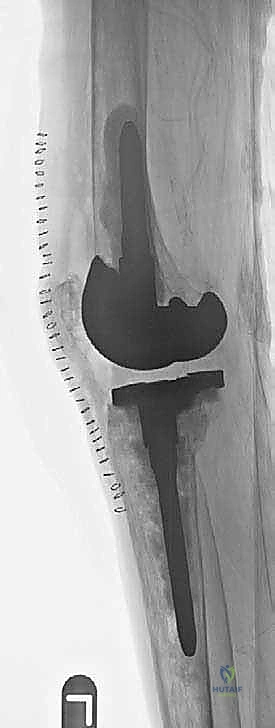
Patient positioning in the operating room must facilitate extensive exposure and intraoperative imaging. The patient is placed supine with a sandbag or specialized leg positioner taped to the table to allow for hyperflexion and stable positioning during component impaction. A sterile tourniquet is applied high on the proximal thigh but is used judiciously to minimize ischemic complications and allow for the assessment of soft tissue viability. Crucially, the operating room table must be radiolucent, and a C-arm fluoroscopy unit must be draped and readily available. Intraoperative fluoroscopy is essential for confirming the trajectory of intramedullary reamers, verifying the seating of metaphyseal cones, and ensuring that diaphyseal stems are not impinging on or perforating the tibial cortex.
Step-by-Step Surgical Approach and Fixation Technique
Exposure and Component Removal
The surgical approach for a revision TKA must balance the need for wide, unhindered exposure with the imperative to preserve the vascularity of the soft tissue envelope. The skin incision should ideally incorporate previous longitudinal incisions; however, if multiple scars exist, the most lateral usable incision is generally preferred to maintain a robust medial skin flap, as the blood supply to the anterior knee predominantly courses from medial to lateral. Subcutaneous dissection must be full-thickness, avoiding the creation of thin, devascularized planes. A standard medial parapatellar arthrotomy is the workhorse approach. The deep incision extends through the quadriceps tendon, leaving a sufficient medial cuff for later repair, and proceeds distally along the medial border of the patella and patellar tendon.
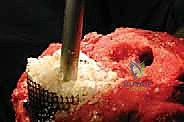
In the setting of severe arthrofibrosis or a patella baja, standard eversion of the patella places immense stress on the patellar tendon insertion at the tibial tubercle. To prevent catastrophic avulsion, the surgeon must be prepared to utilize extensile exposures. A "quadriceps snip" (an oblique incision extending superolaterally into the vastus lateralis) provides excellent relief of tension and does not alter postoperative rehabilitation. If further exposure is required, a tibial tubercle osteotomy (TTO) may be performed. The TTO involves elevating a 6-8 cm block of bone with the patellar tendon attached, hinging it laterally on the anterior tibial musculature. This provides unparalleled access to the diaphyseal canal but requires rigid internal fixation (typically with cerclage wires or screws) at the conclusion of the case.
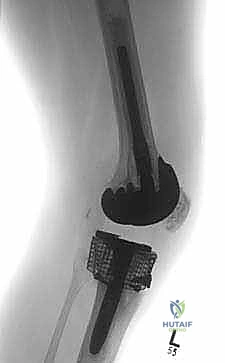
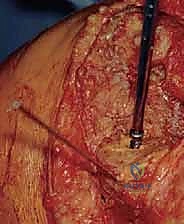
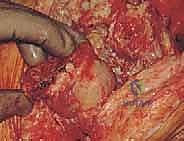
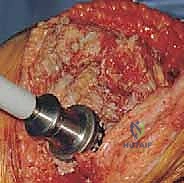
Component extraction is an exercise in patience and bone preservation. The goal is to remove the implants without sacrificing any viable host bone. For the tibial component, the polyethylene insert is removed first to create working space. The interface between the metal tray and the bone (or cement) is then systematically disrupted using thin, flexible osteotomes, oscillating micro-saws, and specialized extraction tools. If the component is cemented, a Gigli saw can be passed under the tibial tray to cut the cement mantle cleanly. Once the tray is removed, any remaining cement and the fibrous, osteolytic membrane must be meticulously curetted. A thorough synovectomy is performed to remove all particulate debris, which is essential to halt the biological cascade of osteolysis and provide a healthy bed for new implants or graft incorporation.
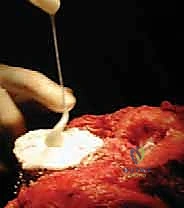
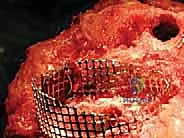
Joint Line Restoration and Defect Management
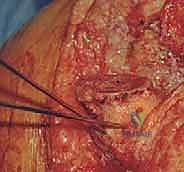
Following component extraction and meticulous debridement, the surgeon is presented with the true magnitude of the bone defect. The AORI classification templated preoperatively must now be confirmed intraoperatively. The first step in reconstruction is establishing the diaphyseal reference. The medullary canal is sequentially reamed using flexible or rigid reamers until cortical chatter is achieved in the diaphyseal isthmus. This establishes the longitudinal axis of the tibia. A trial stem is inserted, and a cutting block is attached to create a fresh, flat tibial platform. The goal is to resect the absolute minimum amount of bone necessary—typically 1-2 millimeters—to create a stable base, ensuring the cut is perpendicular to the mechanical axis of the tibia.
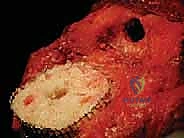
For AORI Type I defects, which are contained cavitary lesions with an intact cortical rim, management is relatively straightforward. Small defects (<5mm) can simply be filled with polymethylmethacrylate (PMMA) bone cement during final implantation. Larger contained defects are ideally managed with morselized cancellous bone graft. The host bone is decorticated with a high-speed burr to expose bleeding bone, and the morselized graft (autograft from bone cuts or fresh-frozen allograft) is vigorously impacted into the defect using specialized tamps. This "impaction grafting" technique restores bone stock and provides excellent biological support for the overlying tibial tray.
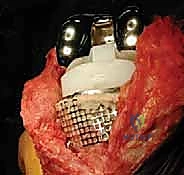
AORI Type II defects involve uncontained bone loss affecting one or both tibial plateaus, where the cortical rim is deficient. These are most commonly addressed using modular metallic augments attached to the tibial tray. Augments are available in various shapes (wedges, blocks, or half-blocks) and thicknesses. The deficient plateau is prepared with a specialized reamer or saw to match the geometry of the chosen augment perfectly. The use of metallic augments allows for immediate structural support, precise restoration of the joint line, and excellent load transfer to the underlying bone. When utilizing augments, a diaphyseal stem is mandatory to offload the metaphyseal bone and prevent catastrophic subsidence of the augmented construct.

Advanced Grafting and Augmentation Techniques
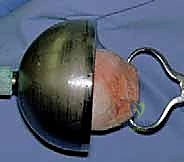
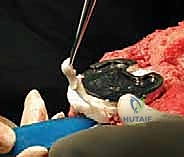
When faced with AORI Type III defects—massive, uncontained bone loss encompassing the entire metaphyseal region and often extending to the tibial tubercle—traditional augments and cement are insufficient. Historically, structural allografts (such as bulk femoral heads or proximal tibial allografts) were the standard of care. The host bone is machined to a flat surface, and the allograft is meticulously contoured using male/female reamers to achieve an intimate fit. The allograft is then rigidly fixed to the host tibia, often using step-cut osteotomies and provisional K-wires, before the diaphyseal stem is passed through both the graft and the host bone. While structural allografts restore bone stock, they carry risks of late resorption, non-union at the host-graft interface, and disease transmission.