Guided Growth for Limb Deformity: An Intraoperative Masterclass in Physeal Modulation
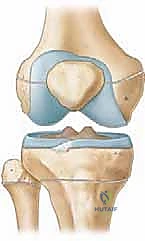
Key Takeaway
This masterclass details guided growth for limb deformity correction. We explore comprehensive surgical anatomy, meticulous preoperative planning, and precise intraoperative execution of eight-plate hemiepiphysiodesis. Fellows will learn real-time techniques, instrument handling, and critical decision-making, including identifying and mitigating neurovascular risks. Extensive pearls and pitfalls, alongside postoperative care, ensure a thorough understanding of this reversible growth modulation technique for optimal patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
The Hueter-Volkmann Principle and Physeal Modulation
Welcome to the operating theater. Today, we are delving into one of the most elegant, biomechanically sound, and rewarding procedures in pediatric orthopaedic surgery: guided growth for the correction of limb deformity. This intervention transcends the mere mechanical straightening of long bones; it relies upon the sophisticated harnessing of the patient’s intrinsic biological growth potential. The foundational concept underlying this technique is the Hueter-Volkmann principle, which dictates that increased mechanical compression across a physis inhibits longitudinal growth, whereas decreased compression or tension stimulates it. By strategically applying a tension-band construct across one side of a growing physis, we selectively tether that specific cortex, allowing the contralateral side to continue its natural expansion, thereby gradually correcting angular deformities in the coronal or sagittal planes.
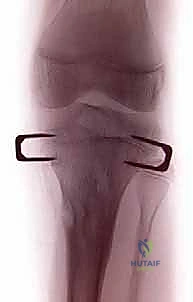
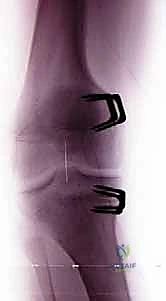
In contemporary pediatric orthopaedics, the paradigm has shifted significantly from invasive, high-morbidity corrective osteotomies to the elegantly simple, minimally invasive technique of temporary hemiepiphysiodesis. Historically, the modulation of the physis was achieved using rigid Blount staples, which, while effective, carried a notable risk of hardware migration, spontaneous extrusion, and permanent physeal arrest due to rigid compression of the perichondrial ring of LaCroix and the zone of Ranvier. The advent of the flexible tension-band plate (such as the eight-Plate) revolutionized this approach. These non-locking, extra-periosteal plates act as a flexible hinge, pivoting on a central fulcrum as the bone grows, thereby mitigating the risk of permanent physeal damage while achieving highly predictable, titratable skeletal realignment.
This masterclass will comprehensively cover the theoretical foundations, meticulous preoperative planning, precise surgical execution, and rigorous postoperative management required to master physeal modulation. It is imperative to recognize that while the surgical technique itself may appear straightforward, the cognitive load—encompassing patient selection, deformity analysis, and timing of intervention—is immense. The surgeon must possess a profound understanding of pediatric skeletal maturation, the natural history of various deformities, and the nuanced interplay between mechanical axis deviation and joint reactive forces.
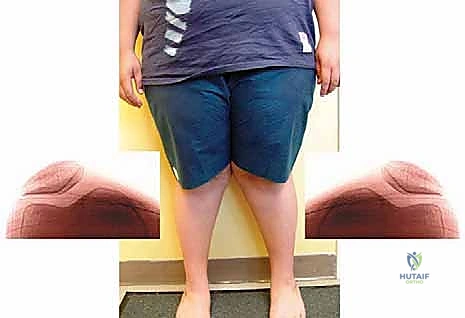
Natural History of Pathologic Deformities
Before contemplating surgical intervention, a rigorous understanding of the underlying pathology and its long-term biomechanical consequences is absolutely paramount. It is crucial to definitively differentiate between physiologic bowing or knock-knees—which follow predictable, self-correcting patterns—and inherently progressive pathologic deformities. Physiologic genu varum typically peaks at 18 to 24 months of age and resolves by age three, transitioning into physiologic genu valgum, which peaks around age four to six before settling into the normal adult alignment of slight valgus. In stark contrast, pathologic deformities, driven by intrinsic bone dysplasias, metabolic derangements, or asymmetric physeal injury, are relentlessly progressive.
These insidious deviations of the mechanical axis precipitate a cascade of deleterious biomechanical events. As the mechanical axis shifts medially (in varus) or laterally (in valgus), ground reaction forces are correspondingly displaced. This eccentric loading creates asymmetric compression across the distal femoral and proximal tibial physes. According to the Hueter-Volkmann law, this excessive, unphysiologic compression on the concave side of the deformity further suppresses chondrocyte proliferation and hypertrophy within the physis, thereby exacerbating the angular deformity. This creates a vicious, self-perpetuating cycle of progressive malalignment, secondary ligamentous laxity, patellofemoral tracking disorders, and ultimately, premature articular cartilage degeneration and early-onset osteoarthritis.
The clinical manifestations of these progressive deformities are multifaceted. They may present unilaterally or bilaterally, involving the femur, the tibia, or both segments simultaneously. Furthermore, coronal plane deformities are frequently accompanied by complex torsional abnormalities and clinically significant limb-length discrepancies. Without surgical intervention to break the cycle of asymmetric loading, the natural history culminates in severe gait disturbances, functional limitations, chronic pain, and the eventual necessity for complex joint reconstruction or arthroplasty in early adulthood.
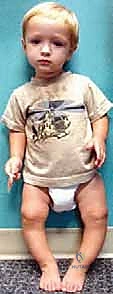
Clinical Presentation and Diagnostic Assessment
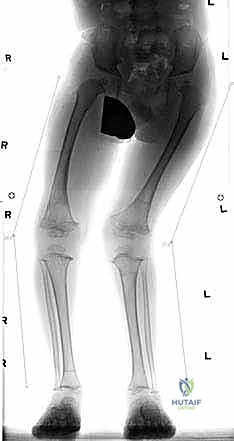
Patients typically present to the orthopaedic clinic with noticeable aesthetic deviations, specifically pronounced knock-knees (genu valgum) or bowlegs (genu varum), often accompanied by an altered gait pattern or parental concern regarding frequent tripping. The clinical assessment must be systematic and exhaustive. For suspected genu valgum, the intermalleolar distance is meticulously measured with the patient standing, knees gently touching, and the patellae oriented strictly forward (neutral rotation). Conversely, for genu varum, the intercondylar distance is measured with the medial malleoli apposed and the patellae neutral. These clinical metrics serve as a baseline for monitoring progression but must invariably be corroborated by rigorous radiographic analysis.
Beyond simple coronal plane measurements, a comprehensive physical examination must evaluate the entire kinetic chain. We meticulously assess the spine for compensatory scoliosis, the pelvis for obliquity, and the feet for associated conditions such as pes planovalgus, which frequently accompanies genu valgum. Stance, dynamic gait analysis, and a complete rotational profile (assessing hip internal/external rotation, thigh-foot angle, and transmalleolar axis) are mandatory. Patellar instability—often manifesting as lateral tracking or frank subluxation—is a highly prevalent concomitant finding. In valgus deformities, this is frequently exacerbated by excessive outward femoral or tibial torsion, whereas inward tibial torsion is more common in varus deformities.
While guided growth is primarily a tool for coronal and sagittal plane correction, realigning the mechanical axis profoundly influences these secondary pathologies. By restoring a neutral mechanical axis, we centralize the extensor mechanism vector, indirectly ameliorating patellar instability and optimizing joint kinematics. However, it is vital to counsel families that severe, rigid torsional malalignments may not fully resolve with coronal plane correction alone and may eventually necessitate derotational osteotomies near skeletal maturity if functional deficits persist.
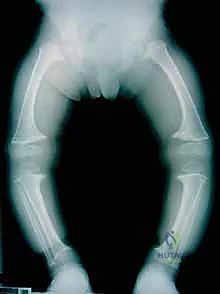
Detailed Surgical Anatomy and Biomechanics
Normal Lower Extremity Alignment and Mechanical Axis
A profound mastery of regional anatomy and normative biomechanical alignment parameters is the bedrock of safe and effective guided growth. The primary objective is to restore the mechanical axis of the lower extremity to a neutral state. During bipedal standing, the normal knee joint line and the adjacent distal femoral and proximal tibial physes remain essentially horizontal relative to the ground. The mechanical axis of the lower extremity—defined as a straight line connecting the center of the femoral head to the center of the tibial plafond—should ideally bisect the knee joint, passing slightly medial to the center of the tibial spines.
Quantitatively, this mechanical axis should intersect the knee joint line at an angle of approximately 87 degrees. The distal femur normally exhibits an anatomic valgus alignment, characterized by a Lateral Distal Femoral Angle (LDFA) of 84 degrees (range 82-86 degrees). Conversely, the proximal tibia demonstrates a slight anatomic varus, with a Medial Proximal Tibial Angle (MPTA) of 87 degrees (range 85-89 degrees). The Joint Line Congruency Angle (JLCA) should be parallel, typically measuring 0 to 2 degrees. This meticulously balanced alignment ensures that weight-bearing forces are distributed evenly across the medial and lateral compartments of the knee, thereby preserving articular cartilage health and ensuring symmetric physeal loading.
To systematically evaluate the degree of deformity, we utilize the zone classification system popularized by Stevens. The knee joint is divided into quadrants (zones 1, 2, and 3) medial and lateral to the center. A mechanical axis passing through zone 1 (mild deviation) may represent a physiologic variation or mild pathology that can be observed. However, Mechanical Axis Deviation (MAD) extending into zone 2 or zone 3 represents a definitive, objective indication for surgical intervention via guided growth, as these degrees of deviation exponentially increase the risk of compartmental overload and progressive joint degeneration.

Distal Femoral Anatomy and Neurovascular Considerations
When addressing distal femoral deformities, precise anatomic dissection is required to access the medial or lateral physis while meticulously protecting surrounding neurovascular structures. For a medial distal femoral approach, the incision is centered over the medial epicondyle. We approach the bone through the investing fascia of the vastus medialis obliquus (VMO). The VMO muscle itself provides a safe, avascular interval. By elevating the anterior border of the VMO, we expose the medial femoral condyle and the undulating topography of the distal femoral physis.
The primary neurovascular concerns on the medial side are the saphenous nerve and the greater saphenous vein. While the main superficial femoral artery and vein reside deep within the adductor canal and are well protected by the vastus medialis, the saphenous nerve—a purely sensory terminal branch of the femoral nerve—exits the adductor canal and becomes superficial along the medial aspect of the knee and proximal leg. The surgeon must be hyper-vigilant during the initial skin incision and superficial dissection to identify and gently retract these structures. Iatrogenic transection or traction injury to the saphenous nerve will result in distressing numbness, dysesthesia, or painful neuroma formation along the medial aspect of the lower leg.
For the lateral distal femoral approach, the surgical corridor involves a longitudinal split in the iliotibial band (ITB). The ITB is a dense, fibrous fascial structure crucial for lateral knee stability. After incising the skin and subcutaneous tissue, the ITB is identified and split in line with its fibers. Deep to the ITB, the vastus lateralis is encountered and retracted anteriorly, exposing the lateral femoral condyle and the physis. In this specific approach, major neurovascular structures are generally not in the direct operative field. The common peroneal nerve courses significantly more distally and posteriorly, wrapping around the fibular head, and the lateral femoral cutaneous nerve remains proximal. However, meticulous hemostasis of the superior lateral genicular vessels is often required.

Proximal Tibial Anatomy and Neurovascular Considerations
Interventions at the proximal tibia require equally rigorous anatomic knowledge, particularly given the proximity of critical neurovascular bundles. For a medial proximal tibial approach, the incision is made longitudinally, centered over the medial aspect of the proximal tibial physis. The dissection proceeds through the subcutaneous tissues to expose the superficial medial collateral ligament (sMCL). The sMCL is a robust, critical static stabilizer of the medial knee. We perform a careful, limited longitudinal split in the fibers of the sMCL, followed by gentle blunt dissection to expose the underlying periosteum and the medial tibial physis. As with the medial femur, the saphenous nerve and its infrapatellar branches, along with the greater saphenous vein, course superficially in this region and must be meticulously protected throughout the procedure.
The lateral proximal tibial approach is arguably the most anatomically perilous of the four standard guided growth corridors. The primary objective is to access the lateral tibial physis while leaving the anterior compartment musculature (tibialis anterior, extensor digitorum longus) and the proximal tibiofibular joint entirely undisturbed. The approach is typically executed just anterior to the fibular head, developing the interval between the anterior tibial crest and the fibula.
The paramount neurovascular concern in this region is the common peroneal nerve. This major nerve exits the popliteal fossa, courses laterally, and wraps directly around the fibular neck, making it highly susceptible to iatrogenic injury during lateral proximal tibial approaches. Excessive posterior retraction, deep errant dissection, or misplaced hardware can result in a catastrophic peroneal nerve palsy, manifesting as a devastating foot drop and sensory loss over the dorsum of the foot. Furthermore, the anterior tibial artery and vein penetrate the interosseous membrane and run deep within the anterior compartment; while deeper than the operative field for a superficial plate, they mandate respect during any drilling or screw insertion.
Exhaustive Indications and Contraindications
Defining the Ideal Candidate for Guided Growth
The ideal candidate for physeal modulation via guided growth is a skeletally immature patient presenting with a progressive coronal or sagittal plane angular deformity, who possesses sufficient remaining growth to achieve the desired correction. The fundamental prerequisite is an open, actively growing physis. We must accurately estimate skeletal maturity, typically utilizing standardized radiographic atlases such as Greulich and Pyle (hand/wrist) or the Sauvegrain method (elbow). As a general clinical rule, guided growth requires a minimum of 6 to 12 months of remaining active physeal growth to yield a clinically and radiographically demonstrable improvement.
The indications for this procedure are broad and encompass a wide spectrum of pediatric orthopaedic conditions. Idiopathic genu valgum and genu varum that fail to spontaneously resolve or progress beyond physiologic norms are the most common indications. However, the true utility of guided growth shines in the management of pathologic deformities. Patients with systemic conditions such as nutritional or X-linked hypophosphatemic rickets, renal osteodystrophy, and various skeletal dysplasias (e.g., achondroplasia, multiple epiphyseal dysplasia) are excellent candidates. Furthermore, guided growth is highly effective in treating asymmetric physeal growth secondary to trauma, infection, or prior tumor resection, provided a sufficient portion of the physis remains viable.
The advantages of guided growth over traditional corrective osteotomies are profound and dictate its position as the first-line surgical intervention. It is a minimally invasive, outpatient procedure with minimal blood loss, negating the need for postoperative casting or prolonged non-weight-bearing protocols. Crucially, the procedure is inherently reversible; once the mechanical axis is restored, the hardware is removed, and normal, symmetric physeal growth resumes. This reversibility provides a significant margin of safety and allows for early intervention, preventing the secondary joint changes that mandate more aggressive surgeries later in life.
| Indication Category | Specific Pathologies | Clinical Rationale for Guided Growth |
|---|---|---|
| Idiopathic Deformities | Persistent Genu Valgum, Adolescent Blount's Disease | Minimally invasive correction of progressive, non-physiologic axis deviation. |
| Metabolic Bone Disease | Rickets (Hypophosphatemic, Nutritional), Renal Osteodystrophy | Corrects alignment despite poor bone quality; avoids non-union risks of osteotomy. |
| Skeletal Dysplasias | Achondroplasia, Metaphyseal Chondrodysplasia | Addresses multi-level, progressive deformities with minimal morbidity. |
| Post-Traumatic/Infectious | Partial Physeal Arrest (with >50% open physis) | Modulates remaining healthy physis to correct angular deviation over time. |
Absolute and Relative Contraindications
While guided growth is highly versatile, stringent adherence to contraindications is essential to prevent surgical failure and patient morbidity. The most absolute contraindication is skeletal maturity. If the physes are closed or possess negligible remaining growth potential (typically girls >14 years, boys >16 years, though skeletal age supersedes chronologic age), tension-band plating will be entirely ineffective. In such mature patients, structural realignment must be achieved via definitive corrective osteotomies (e.g., distal femoral or proximal tibial osteotomies).
Another absolute contraindication is the presence of a complete or near-complete physeal bar (bridge). If a prior traumatic injury or infection has resulted in a dense bony bridge spanning the epiphysis and metaphysis, tethering the contralateral side with a plate will not induce correction; it will merely result in global physeal arrest and subsequent limb-length discrepancy. If a partial physeal bar is suspected, advanced imaging (MRI or CT mapping) is mandatory. If the bar involves less than 30-40% of the physeal cross-sectional area, a bar resection combined with guided growth may be attempted; otherwise, the condition is a contraindication.
Relative contraindications require careful surgical judgment. Severe, rigid intra-articular deformities or extreme ligamentous instability may not be adequately addressed by extra-articular physeal modulation alone. Furthermore, while guided growth can indirectly improve mild rotational abnormalities by centralizing the mechanical axis, it is not a primary treatment for severe, isolated torsional deformities (e.g., severe femoral anteversion or tibial torsion). In cases of "miserable malalignment syndrome" with profound rotational components, guided growth may correct the coronal plane but leave the patient with persistent functional deficits, eventually requiring derotational osteotomies. Lastly, active local infection at the proposed surgical site precludes hardware implantation.
Pre-Operative Planning, Templating, and Patient Positioning
Radiographic Analysis and Deformity Templating
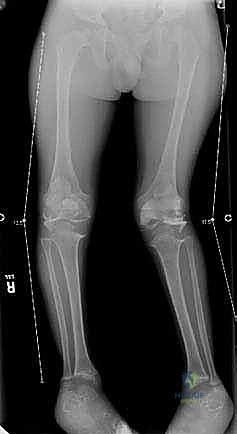
The foundation of a successful guided growth procedure is laid in the preoperative templating phase. The standard radiographic protocol mandates high-quality, full-length, weight-bearing anteroposterior (AP) and lateral radiographs of the bilateral lower extremities. It is absolutely critical that the patient is positioned with the patellae facing strictly forward (neutral rotation) to accurately assess coronal plane alignment; rotational malpositioning will artificially distort the apparent varus or valgus. If a concurrent limb-length discrepancy is clinically apparent, the shorter limb must be supported with calibrated wooden blocks until the pelvis is perfectly leveled before the radiograph is acquired.
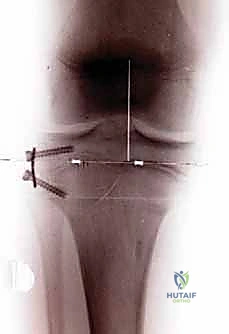
Upon obtaining these optimal images, meticulous digital or manual templating commences. The mechanical axis is drawn from the center of the femoral head to the center of the ankle joint. The Mechanical Axis Deviation (MAD) is quantified in millimeters from the center of the knee joint. We then systematically measure the joint orientation angles: the Lateral Distal Femoral Angle (LDFA) and the Medial Proximal Tibial Angle (MPTA). These specific angles allow the surgeon to precisely localize the apex of the deformity. Often, a deformity is uniapical (e.g., isolated distal femoral valgus), but it may also be multiapical, involving both the femur and the tibia.
Identifying the Center of Rotation of Angulation (CORA) dictates the anatomical site for intervention. If the LDFA is abnormal (e.g., <82 degrees indicating femoral valgus), the distal femoral physis is targeted. If the MPTA is abnormal (e.g., >90 degrees indicating tibial valgus), the proximal tibial physis is targeted. If both are abnormal, double-level guided growth is indicated. Unlike the rigid trigonometric calculations required for opening or closing wedge osteotomies, templating for guided growth is more qualitative regarding the magnitude of correction, as the growth plate will dynamically correct the limb over time. However, precise localization of the deforming segment is non-negotiable.
Advanced Imaging and Torsional Assessment
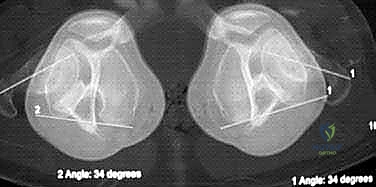
While standard orthogonal radiographs are sufficient for the majority of coronal and sagittal plane deformities, complex clinical presentations necessitate advanced imaging modalities. When physical examination reveals significant rotational asymmetry—such as severe in-toeing or out-toeing that does not correlate with the coronal plane deformity—a rotational "gunsight" CT scan is highly recommended. This specialized CT protocol captures axial slices through the femoral necks, femoral condyles, proximal tibias, and distal tibias.
The "gunsight" CT allows for the highly accurate, objective quantification of femoral anteversion and tibial torsion. Normal femoral anteversion in an older child or adolescent is approximately 10 to 15 degrees, while normal tibial torsion is 15 to 20 degrees external. Documenting these precise rotational parameters is crucial for managing patient and parental expectations. The surgeon must clearly articulate that while guided growth will correct the knock-knees or bowlegs, it will not directly untwist the bone. If the torsional deformity is the primary driver of the patient's functional deficit, guided growth may only serve as a temporizing measure or a preliminary stage before definitive derotational osteotomy.
Furthermore, if the patient has a history of trauma, physeal fracture, or severe localized infection, the integrity of the physis must be interrogated before attempting guided growth. An MRI with specific physeal mapping sequences or a fine-cut CT scan is utilized to detect the presence, location, and cross-sectional area of a physeal bar. As previously stated, attempting guided growth across a completely tethered physis is futile and will only exacerbate limb-length discrepancy.
Operating Room Setup and Patient Positioning
Meticulous preparation of the operating room environment ensures a smooth, efficient, and safe surgical execution. The patient is positioned supine on a radiolucent operating table. Proper positioning is critical; the patient must be placed far enough down the table to allow the C-arm fluoroscopy unit unimpeded access to the entire lower extremity, particularly the knee joint, without the table pedestal obstructing the beam. All pressure points, including the heels and sacrum, are generously padded to prevent decubitus ulcers or neurapraxias during the anesthetic period.
Fluoroscopy is the surgeon's intraoperative "eye" and its setup must be flawless. The C-arm is brought into the sterile field, typically from the contralateral side of the operative limb. Before prepping and draping, a "dry run" is performed to confirm that perfect, true AP and lateral fluoroscopic views of the targeted physes (distal femur, proximal tibia, or both) can be obtained effortlessly. A true AP view is confirmed when the femoral condyles are symmetric and the fibular head is bisected by the lateral tibial cortex. A true lateral view is confirmed by perfect superimposition of the posterior aspects of the medial and lateral femoral condyles.
A sterile pneumatic tourniquet is routinely applied high on the proximal thigh. The use of a tourniquet provides a completely bloodless surgical field, which is invaluable for identifying delicate fascial planes, protecting superficial nerves, and ensuring precise hardware placement without the obscuration of continuous hemorrhage. The entire lower extremity, from the level of the tourniquet down to the toes, is meticulously prepped with a chlorhexidine or iodine-based antiseptic solution and draped in a sterile, free-draped fashion. This free-draping allows the surgeon to manipulate the limb through a full range of motion, facilitating optimal C-arm positioning and dynamic assessment of joint stability.
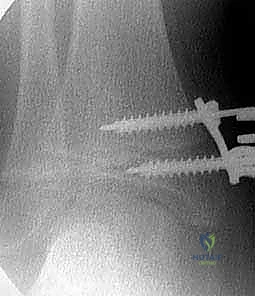
Step-by-Step Surgical Approach and Fixation Technique
Incision, Dissection, and Physeal Identification
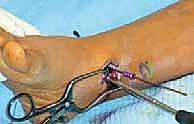
With the patient prepped, draped, and the tourniquet inflated, the surgical masterclass commences. We will focus on the application of the tension-band eight-Plate, the gold standard for this procedure. Under direct fluoroscopic guidance, a radiopaque marker (such as a Kirschner wire or a hemostat) is laid over the skin to precisely localize the targeted physis (e.g., the medial distal femoral physis for genu valgum). A meticulously planned, 2 to 3-centimeter longitudinal skin incision is made directly over this fluoroscopically confirmed site.
The dissection proceeds through the subcutaneous adipose tissue. Meticulous hemostasis is maintained, and superficial veins are cauterized or retracted. As we reach the deep fascia, we employ the specific anatomic intervals detailed previously (e.g., elevating the vastus medialis or splitting the ITB). It is imperative to perform this deep dissection bluntly whenever possible to avoid inadvertent injury to the periosteum or the perichondrial ring. The periosteum must absolutely not be stripped or elevated. Stripping the periosteum can stimulate unpredictable local bone overgrowth or, worse, induce a localized physeal arrest, completely undermining the objective of the procedure.
Once the bone surface is exposed, the precise location of the cartilaginous physis must be tactilely and radiographically confirmed. A straight Keith needle or a smooth 1.6mm Kirschner wire is introduced perpendicular to the bone surface. As the needle penetrates the cortex and enters the physis, the surgeon will experience a distinct, yielding "bar of soap" or "rubber band" sensation, confirming placement within the unossified cartilaginous growth plate. The needle is advanced slightly, and its position strictly within the center of the physis is verified on both true AP and true lateral fluoroscopic views. This needle serves as the critical central fulcrum for the subsequent plate application.