Growing Rod Instrumentation: A Masterclass in Early-Onset Scoliosis Management
Key Takeaway
This masterclass provides an exhaustive guide to growing rod instrumentation for early-onset scoliosis. We delve into comprehensive surgical anatomy, meticulous preoperative planning, and granular, real-time intraoperative execution, from incision to hardware placement. Fellows will gain critical insights into neurovascular risks, precise instrument use, and strategies for managing potential pitfalls, ensuring optimal outcomes for our youngest scoliosis patients.
Comprehensive Introduction and Patho-Epidemiology
The management of early-onset scoliosis (EOS) represents one of the most formidable and intellectually demanding challenges in the field of pediatric orthopedic surgery. Unlike adolescent idiopathic scoliosis, where the primary objective is the three-dimensional correction of a spinal deformity in a nearly mature skeleton, the treatment of EOS requires a delicate, dynamic balance. We are tasked not merely with correcting a curve, but with actively preserving and harnessing the vital growth potential of the developing spine, thorax, and pulmonary system. Growing rod instrumentation has emerged as a cornerstone in this endeavor, offering a mechanical solution to a complex biological problem.
Defining Early-Onset Scoliosis
To establish a rigorous clinical baseline, early-onset scoliosis is definitively characterized by the diagnosis of a coronal plane spinal deformity of 10 degrees or greater presenting at or before the age of 5 years. This specific chronological demarcation is not arbitrary; it is fundamentally rooted in the critical phases of human somatic and pulmonary development. The age cutoff dictates our entire treatment philosophy, shifting the paradigm from definitive arthrodesis—which would disastrously stunt truncal height and lung volume—to growth-sparing techniques. Intervening appropriately during this window is essential to prevent the cascade of cardiopulmonary morbidities associated with severe, unchecked early spinal deformity.
Etiologies and Pathogenesis
The etiologic landscape of EOS is markedly heterogeneous, and a precise understanding of the underlying diagnosis is paramount for formulating a comprehensive, patient-specific surgical strategy. The causes broadly stratify into four main categories, each with distinct prognostic implications:
* Congenital Anomalies: These arise from failures of vertebral formation (e.g., hemivertebrae, wedge vertebrae) or failures of segmentation (e.g., unilateral unsegmented bars, block vertebrae). These structural defects demand meticulous radiographic assessment, as their growth potential is inherently asymmetrical and highly predictable in its progression. Furthermore, they are frequently associated with intraspinal anomalies such as syringomyelia, diastematomyelia, or a tethered spinal cord.
* Neuromuscular Disorders: Pathologies such as cerebral palsy, spinal muscular atrophy (SMA), myelodysplasia, and various muscular dystrophies frequently present with early, rapidly progressive, and relentless spinal curves. These deformities are often characterized by long, sweeping "C-shaped" curves that encompass the pelvis, compounded by profound hypotonia or spasticity, making implant fixation exceptionally challenging.
* Syndromic Associations: A multitude of connective tissue and genetic syndromes feature EOS as a primary phenotypic manifestation. Neurofibromatosis type 1 (NF1) can present with dystrophic curves notorious for rapid progression and pseudoarthrosis. Marfan syndrome, Ehlers-Danlos syndrome, and various trisomies also require tailored approaches due to inherent osteopenia and ligamentous laxity.
* Idiopathic Causes: Infantile idiopathic scoliosis (IIS) is diagnosed when curves appear between birth and 3 years of age without an identifiable structural, neuromuscular, or syndromic cause. While the precise etiology remains elusive, current literature points toward a multifactorial origin involving genetic predisposition, intrauterine molding, and postnatal positioning. Interestingly, IIS has a documented association with breech presentation, plagiocephaly, and, particularly in male infants, premature birth.
Natural History and Thoracic Insufficiency Syndrome
The natural history of EOS is inextricably linked to its specific etiology, but the overarching threat remains the same: the devastating impact of progressive spinal deformity on the developing thorax. While a subset of mild infantile idiopathic curves may spontaneously resolve, congenital and neuromuscular curves almost universally progress. This progression is not merely a cosmetic or orthopedic concern; it is a profound physiological threat.
Progressive curves during the first five years of life critically impede the volumetric expansion of the thorax, leading to Thoracic Insufficiency Syndrome (TIS). TIS is defined as the inability of the thorax to support normal respiration or lung growth. Scoliosis in this vulnerable age group severely inhibits the multiplication and hypertrophy of both alveoli and pulmonary arterioles. The earlier the onset of the deformity, the more hypoplastic the lung parenchyma becomes, resulting in an irreversible diminution of alveolar surface area that far exceeds what would be expected from a simple lack of intrathoracic space. This leads to severe ventilation-perfusion mismatches, restrictive lung disease, pulmonary hypertension, and eventual cor pulmonale.
Detailed Surgical Anatomy and Biomechanics
A profound mastery of pediatric spinal anatomy and growth biomechanics is the absolute prerequisite for safe and effective growing rod instrumentation. The surgeon must navigate a landscape that is continuously changing, working with structures that are largely cartilaginous and highly vulnerable to iatrogenic injury.
Spinal Growth Dynamics
The human spine undergoes two distinct periods of accelerated growth velocity, both of which are intimately linked to the risk of rapid curve progression. The primary period of concern in EOS is from birth to 5 years of age. During this phase, the growth velocity from T1 to L5 is at its zenith, averaging approximately 2 centimeters per year (roughly 1 cm in the thoracic spine and 1 cm in the lumbar spine). By the age of 5, a child has already achieved approximately two-thirds of their final sitting height.
Following this rapid infantile phase, the growth rate plateaus significantly between the ages of 5 and 10, before surging again during the adolescent growth spurt until skeletal maturity. The fundamental biomechanical objective of growing rod instrumentation is to harness and safely direct this early, explosive growth without permanently tethering the spinal column. Premature fusion in this age group not only severely limits truncal height but also induces the "crankshaft phenomenon," where continued anterior vertebral body growth in the presence of a solid posterior fusion mass leads to progressive rotational deformity and lordosis.
Thoracic Development and Pulmonary Function
The development of the thoracic cage and the underlying pulmonary parenchyma occurs in parallel with spinal growth. At birth, thoracic volume is merely 5% of its final adult capacity. It expands to 30% by age 5, reaches 50% by age 10, and completes the remaining 50% of its volumetric expansion during the adolescent years. Crucially, the functional development of the lungs—specifically, the bulk of alveolar multiplication and the completion of the respiratory bronchial tree branching—occurs rapidly in the first year of life and is largely complete by age 8.
Patients afflicted with progressive EOS inevitably develop a restrictive pattern of pulmonary dysfunction. The compliance of the thoracic cage is drastically reduced, leading to a proportional decrease in total lung capacity (TLC) and vital capacity (VC). Unlike late-onset adolescent idiopathic scoliosis, where pulmonary compromise is rarely clinically significant unless curves exceed 80-100 degrees, the severity of scoliosis in EOS is directly and linearly proportional to the severity of restrictive lung disease. This physiological restriction leads to chronic hypoventilation, secondary pulmonary vasoconstriction, and ultimately, right-sided heart failure—complications that can be severely disabling or fatal in early adulthood.
Neurovascular Risks and Muscular Intervals
Posterior spinal surgery in the pediatric population demands an acute awareness of delicate neurovascular structures and the preservation of muscular envelopes to prevent spontaneous autofusion.
* Spinal Cord and Nerve Roots: The pediatric spinal cord is highly sensitive to mechanical manipulation and ischemic insult. The placement of pedicle screws, particularly in the dysplastic, narrow, or largely cartilaginous pedicles frequently encountered in congenital anomalies, carries a substantial risk of cortical breach and direct neurological injury. Continuous, multimodality intraoperative neuromonitoring (somatosensory and motor evoked potentials) is an absolute, non-negotiable standard of care.
* Segmental Vessels: The segmental arteries, originating directly from the aorta, course transversely across the mid-portion of the vertebral bodies to supply the paraspinal musculature and, critically, the spinal cord via radicular branches. Aggressive lateral dissection or errant anterior screw trajectory can lacerate these vessels, leading to catastrophic hemorrhage or spinal cord ischemia, particularly if the vital artery of Adamkiewicz (typically located between T8 and L2 on the left) is compromised.
* Muscular Intervals and Soft Tissue Handling: The surgical approach for growing rods is uniquely tailored to minimize tissue trauma. We typically utilize a midline skin incision but restrict deep subperiosteal dissection exclusively to the planned proximal and distal foundation sites. The intervening paraspinal musculature (the erector spinae group) is left meticulously intact over the apical segments. This subfascial passage of the rods is critical to prevent iatrogenic spontaneous autofusion of the spine, which would defeat the entire purpose of a growth-sparing construct.
* Osteology of the Immature Spine: The surgeon must adapt to the biomechanics of immature bone. Pediatric pedicles are significantly smaller, possess a thicker cartilaginous endochondral cap, and exhibit different spatial trajectories compared to adult vertebrae. The laminae are thin and pliable. Consequently, the fixation strength of anchors (screws or hooks) is inherently lower, predisposing the construct to a higher risk of implant pullout or failure during the repetitive stresses of subsequent lengthening procedures.
Exhaustive Indications and Contraindications
The decision to proceed with growing rod instrumentation is never taken lightly. It represents a commitment to a prolonged, multi-year surgical journey fraught with potential complications. Patient selection must be rigorous, balancing the immediate need for deformity control against the long-term biological costs.
| Category | Specific Parameters | Clinical Rationale |
|---|---|---|
| Absolute Indications | Documented curve progression > 10° despite maximal conservative management (serial casting/bracing). | Indicates failure of non-operative measures; untreated progression will lead to TIS. |
| Absolute Indications | Cobb angle > 50° in a child under 8 years of age with significant remaining growth potential. | Curves of this magnitude in EOS are biomechanically unstable and will inevitably progress, threatening pulmonary function. |
| Absolute Indications | Phase 2 rib-vertebral relationship or RVAD > 20° combined with documented progression. | Radiographic markers strongly predictive of relentless, malignant curve progression in infantile idiopathic scoliosis. |
| Relative Indications | Severe, rigid congenital kyphoscoliosis unamenable to localized hemivertebra excision. | Requires long-segment control to prevent global truncal decompensation while allowing vertical growth. |
| Absolute Contraindications | Medically unstable patient deemed at profound risk for general anesthesia. | The procedure requires prolonged anesthesia; severe existing cor pulmonale may preclude safe intubation/extubation. |
| Absolute Contraindications | Active, untreated systemic or deep surgical site infection. | Implantation of extensive hardware in the presence of bacteremia guarantees catastrophic biofilm formation. |
| Relative Contraindications | Profound osteopenia or metabolic bone disease (e.g., severe Osteogenesis Imperfecta). | High probability of immediate implant pullout; requires specialized anchor techniques (e.g., sublaminar bands) or pre-operative bisphosphonate optimization. |
| Relative Contraindications | Extremely poor soft tissue coverage or severe malnutrition. | High risk of wound dehiscence and hardware prominence; requires nutritional optimization prior to intervention. |
Pre-Operative Planning, Templating, and Patient Positioning
Surgical success in EOS is dictated long before the first incision is made. Meticulous preoperative planning is the blueprint that guides the surgeon through the anatomical complexities of the pediatric spine.
Patient History and Physical Findings
A comprehensive medical history is the foundation of our evaluation. We must meticulously document prenatal, birth, and family histories. Developmental milestones are critical; a delay in motor or cognitive development coupled with a progressive spinal curve should raise immediate suspicion for an underlying neuroaxial or syndromic etiology.
The physical examination requires a systematic and highly detailed approach. The surgeon must observe the child's gait mechanics, respiratory effort at rest, and global truncal and pelvic balance. A thorough cutaneous examination is necessary to identify stigmata of neurofibromatosis (café-au-lait spots, axillary freckling) or spinal dysraphism (hairy patches, sacral dimples). Neurological assessment must include motor strength, sensory pathways, deep tendon reflexes, and the presence of superficial abdominal reflexes. Asymmetric abdominal reflexes are a strong clinical indicator of central nervous system pathology, such as a syrinx, mandating advanced neuroimaging.
Unique physical examination techniques tailored for the EOS population include:
* Thumb Excursion Test: A clinical assessment of dynamic thoracic expansion and chest wall compliance during maximal inspiration.
* Sitting Height Measurement: The most accurate anthropometric indicator of spinal growth, crucial for tracking the efficacy of growth-sparing interventions over time.
* Flexibility Assessment: Evaluated via manual traction applied gently through the cervical spine or via three-point bending at the curve apex to differentiate structural from compensatory deformities.
Imaging and Diagnostic Studies
Radiographic assessment is the absolute cornerstone of preoperative templating. The interpretation of these studies dictates anchor placement, rod contouring, and the overall surgical strategy.
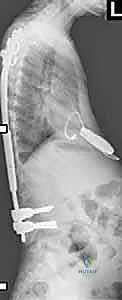
- Full-Length Standing Radiographs: High-quality, 36-inch AP and lateral radiographs from the cervical spine to the pelvis, encompassing the entire thoracic cage, are mandatory. For non-ambulatory neuromuscular patients, supine or seated films are utilized to establish the baseline deformity.
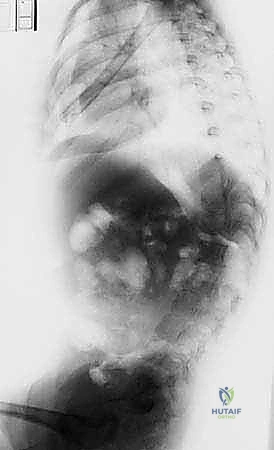
FIG 1 • A. Lateral radiograph of a neuromuscular patient with early-onset scoliosis, demonstrating profound sagittal plane collapse and hyperkyphosis typical of hypotonic disorders.
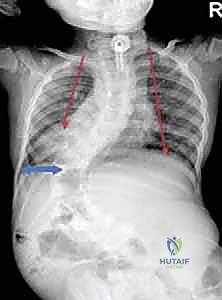
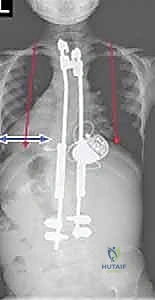
FIG 1 • B. AP radiograph detailing the space available for the lung (SAL) in a patient with early-onset scoliosis. Note the severe pelvic obliquity and "windswept" thoracic cage.
- Traction or Side-Bending Radiographs: These are essential to delineate the true flexibility of the curve, identify the structural apex, and determine the appropriate upper and lower instrumented vertebrae (UIV and LIV).
- Cobb Angle Measurement: The standard metric for quantifying curve magnitude in both coronal and sagittal planes. Surgeons must acknowledge a standard 5-6 degree inter- and intra-observer variability.
- Mehta Rib-Vertebral Angle Difference (RVAD): Described by Min Mehta in 1972, this measurement is a critical prognosticator for infantile idiopathic scoliosis, quantifying apical vertebral rotation.
- The angle is measured between a line drawn perpendicular to the endplate of the apical thoracic vertebra and a line bisecting the neck and head of the corresponding concave and convex ribs.
- Prognostic Significance: An RVAD ≤ 20 degrees suggests an 85-90% probability of spontaneous curve resolution. An RVAD ≥ 21 degrees is a highly reliable predictor of relentless curve progression requiring intervention.
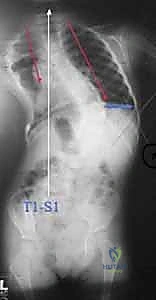
FIG 2 • The rib-vertebral angle difference (RVAD) measures the angle of a line drawn perpendicular to the apical thoracic vertebra endplate and a line drawn down the center of the concave and convex ribs. The difference is calculated by subtracting the convex from the concave angle. An RVAD of 20 degrees or less indicates a curve that is likely to resolve; an RVAD of 21 degrees or greater often is associated with curves that will progress. The phase of the rib head notes the position of the convex rib head on the apical vertebra. A “phase 1” relationship indicates no overlap of the rib head or neck on the apical vertebra. In cases that have a phase 1 relationship, the RVAD may be calculated and used to determine the likelihood of progression. In a phase 2 relationship, the head of the rib on the convex side of the apical vertebra overlaps with the vertebra, and the curve is likely to progress.
- Rib Head Phase: A critical assessment of the spatial relationship between the convex rib head and the apical vertebral body.
- Phase 1: No overlap is present. The RVAD dictates prognosis.
- Phase 2: The convex rib head visually overlaps the vertebral body on the AP radiograph. This indicates severe rotational deformity and a high risk of malignant progression, regardless of the RVAD value.
- Nash-Moe Method: A radiographic grading system for evaluating vertebral rotation by assessing the position of the pedicles relative to the lateral margins of the vertebral body. Grade 0 indicates no rotation, while Grade 4 indicates the convex pedicle has rotated past the midline.
- Space Available for the Lung (SAL): A vital ratio calculated by dividing the distance from the apex of the cephalad rib to the highest point of the hemidiaphragm on the concave side by the same distance on the convex side. A progressively decreasing SAL is a dire indicator of worsening thoracic insufficiency.
- Advanced Imaging (MRI and CT): A total spine MRI is absolutely indicated for any EOS patient with neurological abnormalities, atypical curve patterns (e.g., left thoracic curve), or curves > 20 degrees to definitively rule out intraspinal pathology (syrinx, tethered cord, Chiari malformation). High-resolution CT scanning with 3-D volumetric reconstructions is invaluable for delineating complex bony architecture, mapping dysplastic pedicles in congenital scoliosis, and preoperatively templating pedicle screw trajectories.
- Pulmonary Function Tests (PFTs): Whenever the child is of sufficient age and cognitive ability to cooperate, baseline PFTs are essential to quantify the degree of restrictive lung disease and establish a baseline for post-operative monitoring.
Nonoperative Management and Optimization
Surgical intervention is rarely the first step. A rigorous trial of nonoperative management is indicated for curves that are flexible, mild, or expected to progress slowly, allowing the child to gain critical truncal height and age before committing to instrumentation.
- Serial Derotational Casting: Often performed under general anesthesia on a specialized casting frame, this is the gold standard for delaying surgery in progressive IIS. Casts are meticulously molded to apply specific derotational forces over the rib prominences and are changed every 6-12 weeks to accommodate growth.

FIG 3 • A custom-molded thoracolumbosacral orthosis (TLSO) utilized in the non-operative management of early-onset scoliosis, designed to provide three-point pressure to control curve progression.
- Bracing: Following a successful period of casting, or for curves that are less aggressive, a rigid orthosis (such as a Milwaukee brace or a custom TLSO) may be utilized. Bracing requires strict compliance (typically 23 hours per day) and aims to maintain alignment while the child grows.
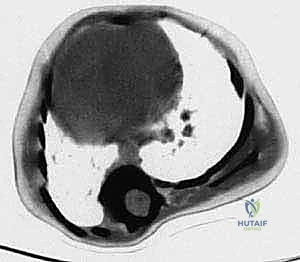
FIG 4 • Application of an elongation-derotation-flexion (EDF) cast under general anesthesia, a cornerstone technique for delaying surgical intervention in infantile idiopathic scoliosis.
Step-by-Step Surgical Approach and Fixation Technique
When conservative measures fail and curve progression threatens pulmonary function, surgical intervention with growing rod instrumentation becomes imperative. The modern standard is the dual growing rod (DGR) construct, which provides superior biomechanical stability and deformity control compared to historical single-rod systems.
Patient Positioning and Neuromonitoring
The patient is brought to the operating theater and placed under general endotracheal anesthesia. Total intravenous anesthesia (TIVA) is strictly utilized to prevent the suppression of neuromonitoring signals. Baseline Somatosensory Evoked Potentials (SSEPs) and Motor Evoked Potentials (MEPs) are obtained prior to positioning.
The patient is carefully log-rolled onto a pediatric Jackson frame or a specialized radiolucent pediatric spine table. Meticulous attention is paid to padding all bony prominences (iliac crests, knees, face) to prevent decubitus ulcers and compressive neuropathies. The abdomen must hang completely free to avoid increased intra-abdominal pressure, which directly translates to engorgement of the epidural venous plexus and excessive intraoperative hemorrhage.
Surgical Exposure: The "Foundation" Concept
The surgical philosophy of growing rods relies on establishing robust proximal and distal "foundations" while leaving the intervening apical spine completely undisturbed.
A standard midline longitudinal incision is made. However, deep subperiosteal dissection of the paraspinal musculature is strictly limited to the planned upper instrumented vertebrae (UIV) and lower instrumented vertebrae (LIV).
* Proximal Foundation: Typically spans 2-3 vertebral levels in the upper thoracic spine (e.g., T2-T4). Dissection exposes the laminae, facet joints, and transverse processes.
* Distal Foundation: Typically spans 2-3 levels in the lower lumbar spine or extends to the pelvis in neuromuscular cases.
The intervening soft tissues over the apex of the deformity are left completely intact. A subfascial or submuscular tunnel is created between the proximal and distal foundations using a blunt specialized tunneling device. This critical step prevents iatrogenic autofusion of the growing spine.
Implant Fixation Strategies
The choice of anchors depends heavily on the patient's age, bone quality, and local anatomy.
- Proximal Anchors: Pedicle screws provide the most rigid three-column fixation, but in very young children with small or dysplastic thoracic pedicles, pedicle/transverse process hooks or sublaminar bands may be utilized to minimize the risk of neurological breach.

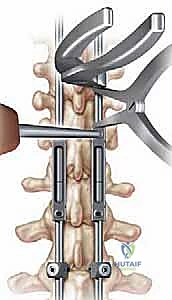
FIG 5 • Intraoperative view demonstrating the meticulous preparation of the proximal foundation. Note the limited subperiosteal exposure restricted only to the anchor sites.
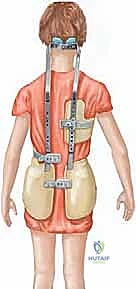
FIG 6 • Placement of proximal anchors. In this instance, a combination of pedicle screws and anatomic hooks are utilized to maximize pullout strength in osteopenic pediatric bone.
- Distal Anchors: Pedicle screws are almost exclusively used in the lumbar spine due to their superior biomechanical pullout strength. In non-ambulatory neuromuscular patients with severe pelvic obliquity, the construct must often be anchored to the pelvis using iliac or S2-alar-iliac (S2AI) screws.
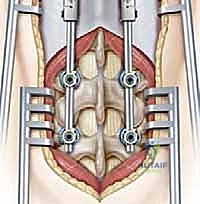
FIG 7 • Preparation of the distal foundation in the lumbar spine. Pedicle screws are placed under fluoroscopic guidance to ensure perfect intraosseous trajectory.
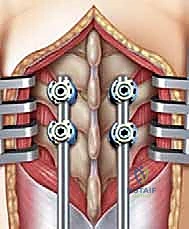
FIG 8 • Final seating of the distal pedicle screws. The intervening paraspinal musculature between the proximal and distal foundations remains completely undisturbed.
Rod Contouring, Insertion, and Distraction
Once the foundations are secure, two titanium or cobalt-chrome rods are carefully contoured to match the desired sagittal profile (restoring physiologic thoracic kyphosis and lumbar lordosis).
The rods are passed blindly through the previously created subfascial tunnels. This requires excellent spatial awareness to ensure the rods do not inadvertently penetrate the pleural cavity or the spinal canal.
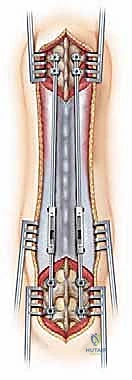
FIG 9 • Subfascial tunneling technique. A blunt passer is used to create a safe tract for the rods, bridging the unexposed apical segments.