Hemiarthroplasty for Proximal Humerus: What You Need to Know
Key Takeaway
Learn more about Hemiarthroplasty for Proximal Humerus: What You Need to Know and how to manage it. Hemiarthroplasty for proximal humerus is a surgical option for complex or severely displaced proximal humerus fractures, which account for 4-5% of all fractures. This procedure replaces the damaged articular segment of the humerus with a prosthetic implant, while preserving the shoulder's socket. It's often chosen when poor bone quality or compromised blood supply increases osteonecrosis risk, particularly in older patients.
Comprehensive Introduction and Patho-Epidemiology
Proximal humerus fractures (PHFs) represent a complex and highly variable spectrum of orthopaedic trauma, accounting for approximately 4% to 5% of all fractures presenting to emergency departments. These injuries involve isolated or combined disruptions to the four primary anatomical segments of the proximal humerus: the greater tuberosity, the lesser tuberosity, the articular segment (humeral head), and the proximal humeral shaft. The incidence of these fractures follows a bimodal distribution. In younger demographics, they are typically the result of high-energy trauma, such as motor vehicle collisions or significant athletic injuries, often presenting with severe comminution and concomitant soft tissue disruption. Conversely, the vast majority of PHFs occur in the elderly population, driven by the increasing prevalence of osteoporosis and associated with low-energy mechanisms, such as a fall from a standing height.

The pathogenesis of proximal humerus fractures in the geriatric population is inextricably linked to declining bone mineral density and frailty. Risk factors for the development of these fractures include advanced age, female sex, lack of hormone replacement therapy, a history of previous fragility fractures, smoking, and the presence of three or more chronic medical comorbidities. Furthermore, the orthopaedic surgeon must maintain a high index of suspicion for pathologic fractures secondary to primary bone malignancies or metastatic disease, particularly in patients presenting with atypical fracture patterns or a history of antecedent shoulder pain prior to the traumatic event. The differential diagnosis for acute shoulder pain and dysfunction following trauma should also include acute hemorrhagic bursitis, traumatic rotator cuff tears, simple glenohumeral dislocations, acromioclavicular separations, and acute calcific tendinitis.

The natural history of displaced, complex proximal humerus fractures treated nonoperatively is historically poor. Neer’s seminal 1970 study evaluating the outcomes of nonoperative management versus hemiarthroplasty for three- and four-part displaced proximal humerus fractures demonstrated universally unsatisfactory results in the nonoperative cohort. These failures were primarily attributed to inadequate fracture reduction, high rates of nonunion and malunion, and the devastating complication of humeral head osteonecrosis with subsequent articular collapse. Subsequent randomized trials, including those by Stableforth and Olerud, reaffirmed these findings, demonstrating that patients with displaced complex fractures treated nonoperatively suffered from significantly worse pain, restricted range of motion, and profound limitations in activities of daily living compared to those undergoing prosthetic replacement.

Despite the historical reliance on hemiarthroplasty for complex fractures, the management paradigm has evolved significantly. While nonoperative treatment remains the gold standard for minimally displaced fractures (which constitute nearly 80% of all PHFs), the surgical management of displaced three- and four-part fractures requires a nuanced approach. The decision to proceed with hemiarthroplasty hinges on patient-specific variables, including physiologic age, bone quality, cognitive status, and the ability to comply with a rigorous postoperative rehabilitation protocol. Moribund individuals or those with profound cognitive impairments (e.g., advanced dementia, severe closed head injuries) are generally poor candidates for prosthetic reconstruction, as the success of a hemiarthroplasty is critically dependent on structured, deliberate postoperative physical therapy to ensure functional tuberosity healing and dynamic joint stability.
Detailed Surgical Anatomy and Biomechanics
A profound understanding of the surgical anatomy of the proximal humerus is the foundation upon which successful hemiarthroplasty is built. As conceptualized by Neer, the proximal humerus is divided into four distinct segments: the articular surface (anatomical head), the greater tuberosity, the lesser tuberosity, and the humeral diaphysis. The spatial relationship between these segments is paramount for prosthetic reconstruction. On average, the most cephalad aspect of the articular segment sits 8 mm superior to the superior tip of the greater tuberosity. Failure to restore this critical height relationship during hemiarthroplasty leads to either subacromial impingement (if the tuberosities are placed too high) or profound deltoid and rotator cuff mechanical disadvantage (if the stem is placed too low). Furthermore, native humeral version averages approximately 30 degrees of retroversion relative to the epicondylar axis of the distal humerus, though this can range widely from 10 to 55 degrees among individuals.

The intertubercular (bicipital) groove serves as a critical anatomical landmark during surgical reconstruction. It lies between the greater and lesser tuberosities, housing the long head of the biceps tendon as it transitions from its intra-articular origin at the superior glenoid tubercle to the distal arm. The tuberosities themselves attach to the articular segment at the anatomic neck and serve as the insertion sites for the rotator cuff musculature. The greater tuberosity features three distinct facets—superior, middle, and inferior—accommodating the insertions of the supraspinatus, infraspinatus, and teres minor tendons, respectively. The lesser tuberosity provides the singular insertion site for the subscapularis tendon. Distal to the surgical neck, the deltoid, pectoralis major, and latissimus dorsi insert onto the humeral shaft. These massive muscle groups act as primary deforming forces during a fracture, with the pectoralis major pulling the shaft medially and anteriorly, while the rotator cuff retracts the tuberosities superiorly and posteriorly.

The vascular anatomy of the proximal humerus dictates the risk of osteonecrosis following a fracture. Historically, the anterolateral branch of the anterior humeral circumflex artery—often referred to as the arcuate artery of Laing—was considered the primary blood supply to the humeral head. This vessel courses parallel to the lateral aspect of the long head of the biceps tendon, penetrating the bone at the proximal terminus of the intertubercular groove. However, modern microvascular studies have revolutionized this understanding, demonstrating that the posterior humeral circumflex artery plays a much more substantial, if not dominant, role in the perfusion of the articular segment. Preservation of the posteromedial capsular hinge is therefore critical in valgus-impacted four-part fractures, as it often maintains adequate vascular inflow from the posterior circulation, potentially allowing for joint-preserving fixation rather than arthroplasty.

The biomechanics of a proximal humeral hemiarthroplasty rely entirely on the successful healing of the tuberosities to the prosthetic stem and the humeral shaft. Unlike total hip or knee arthroplasty, where inherent prosthetic constraint and static ligamentous balancing provide stability, the shoulder is a highly unconstrained joint dependent on the dynamic concavity compression provided by the rotator cuff. If the tuberosities fail to heal, resorb, or migrate postoperatively, the patient will be left with a functionally pseudoparalytic shoulder, characterized by superior migration of the prosthetic head and an inability to actively elevate the arm above the horizontal plane. Therefore, the surgical technique must prioritize meticulous handling of the tuberosity fragments, rigid cerclage fixation, and the use of autologous cancellous bone graft to optimize the biologic environment for osteointegration.
Exhaustive Indications and Contraindications
The decision to perform a hemiarthroplasty for a proximal humerus fracture requires a careful synthesis of patient-specific physiological factors and fracture morphology. While the advent of reverse total shoulder arthroplasty (RTSA) has significantly altered the treatment algorithm, particularly in the elderly, hemiarthroplasty remains a vital operation for specific indications. The primary goal of any surgical intervention in this setting is to anatomically reconstruct the glenohumeral joint, restore humeral height, replicate appropriate prosthetic retroversion, and establish secure, mechanically sound tuberosity fixation to allow for early rehabilitation.

Hemiarthroplasty is classically indicated for severe, displaced four-part proximal humerus fractures, particularly in patients under the age of 65 to 70 who possess adequate bone stock to support tuberosity healing. It is also the treatment of choice for head-splitting fractures where the articular cartilage is irreparably comminuted, and for chronic anterior or posterior fracture-dislocations where more than 40% of the articular surface is damaged (e.g., severe Hill-Sachs or reverse Hill-Sachs lesions). In three-part fractures occurring in severely osteoporotic bone where open reduction and internal fixation (ORIF) carries an unacceptably high risk of hardware cutout and failure, hemiarthroplasty serves as a reliable alternative. Furthermore, several studies have demonstrated that primary hemiarthroplasty performed in the acute setting yields functional outcomes vastly superior to delayed reconstruction performed for post-traumatic arthritis or malunion.

Contraindications to hemiarthroplasty must be strictly respected to avoid catastrophic failure. Absolute contraindications include active local or systemic infection, a neuropathic (Charcot) joint, and the presence of a non-functioning deltoid muscle (e.g., due to a pre-existing axillary nerve palsy). Relative contraindications involve patient factors that preclude participation in the demanding postoperative rehabilitation protocol, such as severe cognitive impairment, active substance abuse, or profound medical comorbidities rendering the patient unfit for surgery. Additionally, in elderly patients (typically >70 years) with massive, irreparable rotator cuff tears or profound tuberosity comminution where healing is deemed highly unlikely, RTSA is now widely considered the superior surgical option, as it relies on deltoid tension rather than rotator cuff integrity for overhead function.
Indications and Contraindications Summary
| Category | Specific Conditions |
|---|---|
| Primary Indications | Displaced 4-part fractures in physiologically young patients (<65-70 yrs) Head-splitting fractures with irreparable articular comminution Anatomic neck fractures with high risk of avascular necrosis Fracture-dislocations with >40% articular surface impression defects |
| Relative Indications | 3-part fractures in severe osteoporotic bone precluding ORIF Delayed presentation of complex fractures without established malunion |
| Absolute Contraindications | Active glenohumeral or systemic infection Neuropathic arthropathy (Charcot shoulder) Pre-existing deltoid paralysis / axillary nerve transection |
| Relative Contraindications | Extreme physiological age with poor bone stock (RTSA preferred) Inability to comply with strict postoperative rehabilitation Pre-existing massive, irreparable rotator cuff arthropathy |

Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning is the cornerstone of a successful proximal humeral hemiarthroplasty. The clinical evaluation must begin with a meticulous neurovascular examination of the injured extremity. The axillary nerve is particularly vulnerable in anterior fracture-dislocations, and its function must be documented by assessing sensation over the lateral deltoid and, when possible, isometric deltoid contraction. A comprehensive history should elucidate the patient's premorbid functional baseline, hand dominance, occupational demands, and any history of malignancy that might suggest a pathologic etiology.

Standard radiographic evaluation requires a complete trauma series of the shoulder, including true anteroposterior (Grashey), scapular "Y", and axillary lateral views. If patient discomfort precludes a standard axillary radiograph, a Velpeau trauma axillary view can be obtained with the patient remaining in a sling. Advanced imaging with fine-cut Computed Tomography (CT) with 3D reconstructions is highly recommended for all complex proximal humerus fractures. CT imaging allows the surgeon to precisely map the fracture lines, quantify the degree of tuberosity comminution, assess glenoid version and morphology, and definitively diagnose articular head-splitting components that might be occult on plain radiographs. Preoperative digital templating should be performed using a true AP radiograph of the contralateral, uninjured shoulder to estimate the native humeral head diameter, radius of curvature, and the distance from the superior margin of the pectoralis major insertion to the top of the humeral head.

Anesthetic management typically involves a combination of regional and general anesthesia. An ultrasound-guided interscalene brachial plexus block provides excellent perioperative analgesia, significantly reducing volatile anesthetic requirements and postoperative opioid consumption. Endotracheal intubation is strongly recommended over a laryngeal mask airway (LMA) to ensure a secure airway during dynamic positioning and to allow for profound pharmacological muscle relaxation, which is essential for mobilizing retracted tuberosity fragments and reducing the joint without excessive force.

Patient positioning is executed meticulously in the beach-chair configuration. The patient is placed on a specialized operating table with the backrest elevated to approximately 45 to 60 degrees. The head and neck are secured in a neutral position using a Mayfield headrest or specialized foam positioner, ensuring all pressure points are heavily padded and the eyes are protected. The operative arm must be completely free to allow for unrestricted extension, adduction, and rotation. A sterile articulating arm holder (e.g., Spider or Trimano) is highly advantageous, providing stable, static positioning during canal preparation and cementing. Intraoperative C-arm fluoroscopy must be positioned coming in from the contralateral side or over the head of the bed, ensuring orthogonal views of the proximal humerus can be obtained effortlessly without compromising the sterile field.
Step-by-Step Surgical Approach and Fixation Technique
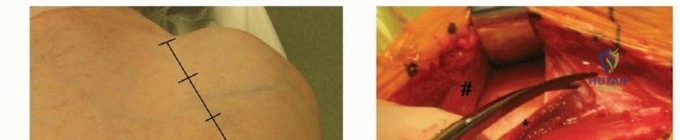
The surgical execution of a hemiarthroplasty for a proximal humerus fracture is universally performed via the classic deltopectoral approach. This internervous plane between the deltoid (axillary nerve) and the pectoralis major (medial and lateral pectoral nerves) provides extensile access to the anterior shoulder without denervating the critical abductor musculature. The skin incision begins just superior and lateral to the coracoid process and extends distally and laterally along the deltopectoral groove toward the deltoid tuberosity. Subcutaneous tissues are divided, and the cephalic vein is identified. The vein is typically retracted laterally with the deltoid to preserve its major tributary branches, though medial retraction can be utilized based on surgeon preference and venous anatomy.

Deep dissection involves mobilizing the pectoralis major medially and the deltoid laterally. If exposure to the inferior capsule or proximal shaft is limited, the proximal 1 to 2 centimeters of the pectoralis major tendon insertion may be released and tagged for later repair. The clavipectoral fascia is then incised lateral to the conjoint tendon, exposing the fracture hematoma, the bursa, and the tuberosity fragments. At this stage, the axillary nerve should be palpated digitally at the anteroinferior border of the subscapularis as it courses posteriorly through the quadrangular space. Gentle external rotation of the arm relaxes the capsule and reduces tension on the nerve.

Tuberosity mobilization is the most critical and technically demanding aspect of the procedure. The long head of the biceps (LHB) tendon is identified within the bicipital groove and traced proximally to the rotator interval. The LHB serves as the definitive landmark separating the greater tuberosity (posterior/superior) from the lesser tuberosity (anterior). The rotator interval is opened, and the coracohumeral ligament is released to allow for independent mobilization of the tuberosities. Heavy, nonabsorbable traction sutures (e.g., #2 or #5 FiberWire or 1-mm cottony Dacron) are placed directly through the tendon-bone interface of the rotator cuff insertions. Typically, two to three sutures are passed through the subscapularis, and three to four sutures are passed through the supraspinatus and infraspinatus tendons. These sutures provide absolute control over the fragments and will later be utilized for definitive cerclage fixation.

Once the tuberosities are controlled and retracted, the fractured articular head is extracted. The native head is measured using a caliper to select the appropriate prosthetic head size, aiming to recreate the native radius of curvature. Crucially, the extracted head is taken to the back table, where all available cancellous bone is harvested using a rongeur and curette; this autograft will be packed around the stem and between the tuberosities to stimulate osteointegration. The humeral canal is then sequentially broached or reamed to accommodate the prosthetic stem. Modern fracture-specific stems often feature proximal bone windows, reduced proximal profiles to prevent tuberosity overstuffing, and specialized suture holes or collars to facilitate anatomic tuberosity reconstruction.

Determining the correct height and version of the prosthesis is paramount. Height can be referenced using the superior border of the pectoralis major tendon (typically 5.6 cm from the top of the prosthetic head) or by
Clinical & Radiographic Imaging Archive



























