Corticosteroid-Induced Avascular Necrosis (AVN) of the Humeral Head: Etiology, Pathophysiology, and Clinical Insights

Key Takeaway
Avascular necrosis (AVN) of the humeral head, or ONFH, is bone cell death due to interrupted blood supply. Corticosteroid use is a significant non-traumatic risk factor. Pathophysiology involves fat embolization, hypercoagulability, and increased intraosseous pressure. Diagnosis uses systems like Ficat, Steinberg, and ARCO, with treatment guided by these stages.
Introduction and Epidemiology
Osteonecrosis of the humeral head (ONFH), commonly referred to as avascular necrosis (AVN), represents a debilitating condition characterized by the death of osteocytes and marrow cells due to an interruption of the blood supply to the humeral head. This ischemic insult leads to subchondral bone collapse, articular cartilage incongruity, and ultimately, glenohumeral osteoarthritis. While multifactorial in etiology, corticosteroid use is a prominent and well-recognized risk factor, significantly contributing to the incidence of non-traumatic ONFH.
The pathophysiology of corticosteroid-induced ONFH is complex, involving multiple proposed mechanisms. These include fat embolization leading to microvascular occlusion, corticosteroid-induced hypercoagulability, increased intraosseous pressure secondary to marrow adipogenesis, direct cytotoxicity to osteocytes, and impairment of reparative angiogenesis. At the cellular level, glucocorticoids induce the differentiation of mesenchymal stem cells into adipocytes rather than osteoblasts via the upregulation of peroxisome proliferator-activated receptor gamma (PPAR-gamma). This adipocytic shift leads to marrow hypertrophy within the rigid confines of the proximal humerus, elevating intraosseous pressure, compressing venous outflow, and precipitating a compartment syndrome of the bone. High-dose and prolonged corticosteroid administration, particularly systemic corticosteroids, are strongly implicated. While a direct dose-response relationship is challenging to quantify definitively across all patients due to individual variability, cumulative dose, duration of therapy, and underlying conditions requiring steroid treatment are all critical considerations.
Epidemiologically, ONFH is less common than osteonecrosis of the femoral head, but its prevalence is increasing, partly due to the wider use of corticosteroids in various medical conditions and improved diagnostic imaging. It can affect individuals across a broad age spectrum, though corticosteroid-associated cases tend to manifest in adults, frequently in the third to fifth decades of life. This presents a unique challenge, as these patients are often younger and more active than the typical primary osteoarthritis demographic. Other significant risk factors include heavy alcohol consumption, trauma (e.g., proximal humerus fractures disrupting vascularity), sickle cell disease, Gaucher's disease, systemic lupus erythematosus, HIV infection, organ transplantation, chemotherapy, radiation therapy, and diving-related decompression sickness. Approximately 15-20% of cases are idiopathic.
The natural history of ONFH is often characterized by progressive deterioration. Initially, patients may be asymptomatic or experience mild, intermittent shoulder pain. As the disease progresses, subchondral bone collapse ("crescent sign") develops, leading to increasingly severe pain, loss of range of motion, and functional impairment. Without intervention, joint destruction and secondary osteoarthritis are common sequelae.
Several classification systems are utilized to stage ONFH, guiding prognosis and treatment decisions. The Cruess classification, specifically adapted for the shoulder from the Ficat and Arlet hip classification, remains widely utilized in orthopedic literature.
* Stage I Normal radiographs, changes visible only on MRI (edema, double-line sign).
* Stage II Sclerosis, osteopenia, or cystic changes on plain radiographs without subchondral collapse.
* Stage III Subchondral fracture resulting in the pathognomonic crescent sign, indicating early mechanical failure.
* Stage IV Flattening and collapse of the articular surface.
* Stage V Degenerative changes extending to the glenoid, representing end-stage secondary osteoarthritis.
Contemporary staging also relies heavily on the ARCO (Association Research Circulation Osseous) Classification, an international system that emphasizes MRI findings for early diagnosis and stratifies patients for appropriate management pathways based on the volumetric extent of necrosis.
Surgical Anatomy and Biomechanics
A thorough understanding of the surgical anatomy of the proximal humerus and glenohumeral joint, particularly its vascular supply, is paramount for both diagnosis and surgical management of ONFH.
The humeral head receives its primary blood supply from the ascending branch of the anterior humeral circumflex artery (AHCA) and branches from the posterior humeral circumflex artery (PHCA). The ascending branch of the AHCA, often termed the arcuate artery of Laing, traditionally was considered the dominant blood supply to the humeral head. It ascends in the bicipital groove, giving off branches to the lesser and greater tuberosities before penetrating the head.
However, modern quantitative anatomical and microangiographic studies have fundamentally shifted this paradigm. Research demonstrates that the PHCA actually provides the majority of the blood supply to the humeral head (up to 64%), particularly the critical superior and posterior quadrants. The PHCA travels through the quadrangular space with the axillary nerve and sends robust intraosseous anastomotic branches into the posteromedial aspect of the surgical neck. This intraosseous anastomotic network is critical; however, the terminal subchondral vessels remain vulnerable to ischemic insults. In corticosteroid-induced AVN, the microvascular occlusion predominantly affects the superior-central aspect of the humeral head, which is subjected to the highest biomechanical contact forces.
Biomechanically, the glenohumeral joint is characterized by a profound osseous mismatch, prioritizing mobility over stability. The humeral head represents approximately one-third of a sphere, articulating with the shallow glenoid fossa. Normal humeral head version is typically 20 to 30 degrees of retroversion relative to the transepicondylar axis, with a neck-shaft inclination angle of approximately 130 to 140 degrees.
When corticosteroid-induced osteonecrosis progresses to Stage III or IV, the structural integrity of the subchondral bone is compromised. The collapse alters the radius of curvature of the humeral head. This geometric distortion eliminates the normal congruent articulation, leading to point-loading and dramatically increased contact stresses on the opposing glenoid cartilage. This altered kinematic profile accelerates the degradation of the glenoid, propelling the disease from isolated humeral head pathology (Stage IV) to bipolar glenohumeral arthritis (Stage V). Understanding these biomechanical consequences dictates the surgical algorithm, particularly the decision between joint-preserving procedures, humeral-sided replacements, and total joint arthroplasty.
Indications and Contraindications
The management of corticosteroid-induced humeral head AVN is dictated by the stage of the disease, the extent of articular involvement, patient age, functional demands, and medical comorbidities. The primary objective is to alleviate pain and restore function, utilizing joint-preserving techniques in early stages and arthroplasty in advanced stages with structural collapse.
Operative Versus Non Operative Management
| Management Strategy | Indications | Contraindications |
|---|---|---|
| Non-Operative (Observation, NSAIDs, Physical Therapy) | Asymptomatic Stage I or II; Poor surgical candidates; Patients with limited functional demands. | Rapidly progressive pain; Structural collapse (Stage III/IV) in active patients; Severe mechanical symptoms. |
| Core Decompression (with or without bone grafting) | Symptomatic Stage I or II (pre-collapse); Small to medium necrotic lesions (< 50% head involvement). | Stage III (crescent sign) or higher; Large lesions (> 50% involvement); Active infection. |
| Hemiarthroplasty | Stage III or IV (collapse) with an intact, pristine glenoid cartilage surface; Young, high-demand patients where glenoid wear is a concern. | Stage V (glenoid arthritis); Coracoacromial arch insufficiency; Unrepairable rotator cuff tear. |
| Total Shoulder Arthroplasty (TSA) | Stage IV or V with concurrent glenoid cartilage degeneration; Intact rotator cuff. | Active infection; Neuropathic joint; Unrepairable rotator cuff tear; Deltoid paralysis. |
| Reverse Total Shoulder Arthroplasty (rTSA) | Stage IV or V with concomitant massive, unrepairable rotator cuff tear; Severe glenoid bone loss precluding standard TSA. | Functioning deltoid is required; Active infection; Axillary nerve palsy. |
Non-operative management is rarely successful in halting the progression of corticosteroid-induced AVN, given the systemic and irreversible nature of the microvascular insult. Therefore, surgical intervention is frequently required. Core decompression aims to reduce intraosseous pressure and stimulate reparative angiogenesis, often augmented with orthobiologics or structural bone grafts. Arthroplasty remains the gold standard for post-collapse disease, providing predictable pain relief and functional restoration.
Pre Operative Planning and Patient Positioning
Thorough preoperative evaluation is critical for optimizing surgical outcomes in the setting of humeral head osteonecrosis.
Imaging and Templating
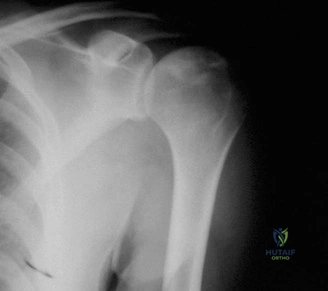
Standard radiographic evaluation must include a true anteroposterior (Grashey) view, an axillary lateral view, and a scapular Y view. These plain films are assessed for the presence of sclerosis, cystic changes, the crescent sign, humeral head flattening, and glenoid wear.
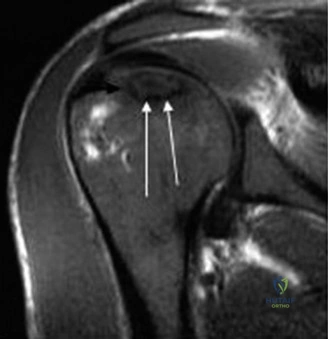
Magnetic Resonance Imaging (MRI) is the gold standard for early detection, staging, and volumetric assessment of the necrotic lesion. T1-weighted images typically demonstrate a well-demarcated band of low signal intensity separating the necrotic segment from viable bone. T2-weighted images may reveal the classic "double-line sign," representing the hypervascular granulation tissue interface at the border of the infarct. MRI is essential for identifying early, pre-radiographic Stage I disease, which is highly amenable to core decompression.
Computed Tomography (CT) without contrast is highly recommended for advanced stages (Stage III-V) when arthroplasty is considered. CT provides superior detail regarding the geometry of the humeral head collapse, subchondral cyst formation, and, critically, glenoid morphology. 3D reconstruction and preoperative templating software allow the surgeon to precisely plan humeral head resection levels, determine the appropriate component size, and assess glenoid version and available bone stock for baseplate or peg fixation.
Patient Positioning and Anesthesia
Surgery is typically performed under regional anesthesia (interscalene block) combined with general endotracheal anesthesia to ensure complete muscle relaxation and postoperative pain control.
The patient is placed in the beach chair position, flexed to approximately 30 to 45 degrees at the hips and knees to prevent sliding. The operative shoulder must be positioned completely off the edge of the table to allow for unrestricted extension and adduction of the arm, which is critical for humeral preparation during arthroplasty. The head is secured in a specialized headrest, maintaining neutral cervical alignment. The arm is prepped and draped free, often utilizing a sterile hydraulic arm positioner (e.g., SPIDER or Trimano arm) to assist with dynamic positioning and retraction during the procedure. Careful padding of all bony prominences, particularly the contralateral ulnar nerve and bilateral fibular heads, is mandatory to prevent perioperative neuropraxias.
Detailed Surgical Approach and Technique
The standard approach for both core decompression and shoulder arthroplasty in the setting of AVN is the deltopectoral approach. This utilizes an internervous plane, minimizing denervation to the dynamic stabilizers of the shoulder.
The Deltopectoral Approach
An incision is made starting from the tip of the coracoid process and extending distally and laterally toward the deltoid insertion. Subcutaneous tissue is incised, and the cephalic vein is identified, marking the interval between the deltoid (axillary nerve) and the pectoralis major (medial and lateral pectoral nerves). The cephalic vein is typically retracted laterally with the deltoid to preserve its primary venous tributaries, though medial retraction is acceptable based on surgeon preference and venous anatomy.
The deltopectoral interval is developed bluntly. The clavipectoral fascia is incised lateral to the conjoined tendon (coracobrachialis and short head of the biceps). The conjoined tendon is retracted medially. Care must be taken to avoid aggressive medial retraction, which places the musculocutaneous nerve at risk as it enters the coracobrachialis typically 3-5 cm distal to the coracoid. The axillary nerve is palpated at the inferior border of the subscapularis, traversing the quadrangular space, and protected throughout the procedure.
Core Decompression Technique
For pre-collapse disease (Stage I/II), core decompression is performed. Following the deltopectoral exposure, the anterior humeral circumflex vessels ("three sisters") at the inferior border of the subscapularis are identified and managed. Under direct fluoroscopic guidance, a guide pin is introduced into the lateral humeral cortex, distal to the greater tuberosity, and advanced into the center of the necrotic lesion within the humeral head.
Once the pin position is confirmed on both AP and axillary fluoroscopic views, a cannulated reamer or core drill (typically 6mm to 8mm) is passed over the wire to decompress the lesion. The necrotic bone is curetted. The resulting void can be backfilled with autologous cancellous bone graft (often harvested from the proximal humerus or iliac crest), allograft, or demineralized bone matrix mixed with bone marrow aspirate concentrate (BMAC) to provide osteoconductive and osteoinductive properties. The tract is then sealed, and the wound is closed in layers.
Total Shoulder Arthroplasty Technique
For post-collapse disease (Stage IV/V), arthroplasty is indicated.

Subscapularis Management
Access to the glenohumeral joint requires mobilization of the subscapularis. This can be achieved via a lesser tuberosity osteotomy (LTO), a subscapularis peel off the bone, or a tenotomy. In patients with corticosteroid-induced AVN, the bone quality of the lesser tuberosity may be compromised. If the bone is structurally sound, an LTO provides excellent bone-to-bone healing. If the bone is severely osteopenic, a tenotomy or peel followed by robust transosseous repair may be preferred. The biceps tendon is typically tenodesed to the pectoralis major or the bicipital groove.
Humeral Preparation
The shoulder is dislocated via external rotation and extension. The humeral head resection is performed using an oscillating saw. The cut is planned based on preoperative templating, aiming to replicate the patient's native retroversion (typically 20-30 degrees) and inclination. The humeral canal is sequentially broached to accept the humeral stem. In AVN, the metaphyseal bone may be cystic and soft; thus, care must be taken to avoid varus malalignment or cortical perforation. Depending on bone quality, a press-fit or cemented stem is utilized.
Glenoid Preparation
If a TSA is planned, the humerus is retracted posteriorly with a Fukuda retractor to expose the glenoid. The labrum is excised, and the glenoid surface is reamed to correct any version abnormalities and create a concentric surface. A keeled or pegged polyethylene glenoid component is cemented into place using a modern cementing technique with pulsatile lavage and pressurization.
Implantation and Closure
The definitive humeral stem and modular head are impacted. The joint is reduced, and stability is assessed. Translatory testing (anterior, posterior, and inferior) should demonstrate approximately 50% translation with spontaneous relocation. The subscapularis is meticulously repaired using heavy non-absorbable sutures. The deltopectoral interval is closed, followed by subcutaneous tissue and skin.
Complications and Management
Surgical intervention for corticosteroid-induced AVN carries specific risks, exacerbated by the underlying etiology and the systemic effects of chronic corticosteroid use, such as immunosuppression and poor tissue healing.
Common Complications and Salvage Strategies
| Complication | Estimated Incidence | Etiology / Risk Factors | Management and Salvage Strategy |
|---|---|---|---|
| Subscapularis Failure | 2% - 8% | Poor tendon/bone quality due to chronic steroids; Non-compliance with postoperative restrictions. | Early recognition is key. Operative repair if acute. If chronic and irreparable with functional deficit, consider pectoralis major transfer or revision to rTSA. |
| Glenoid Component Loosening | 5% - 15% (Long-term) | Eccentric loading; Polyethylene wear; Progression of underlying bone disease. | Revision arthroplasty. May require bone grafting of the glenoid vault and conversion to a reverse total shoulder arthroplasty (rTSA). |
| Periprosthetic Infection | 1% - 3% | Immunosuppression from corticosteroids; Diabetes; Rheumatologic conditions. | Acute (< 4 weeks): Irrigation and debridement with modular component exchange (DAIR). Chronic: Two-stage revision with antibiotic spacer and prolonged IV antibiotics. |
| Progressive Glenoid Erosion | 10% - 20% (Hemiarthroplasty) | Hard metallic head articulating against native cartilage; Altered biomechanics. | Conversion from hemiarthroplasty to Total Shoulder Arthroplasty (TSA) or rTSA depending on rotator cuff integrity. |
| Disease Progression (Post-Decompression) | 20% - 40% | Failure to arrest ischemia; Continued high-dose corticosteroid use. | Conversion to hemiarthroplasty or total shoulder arthroplasty once structural collapse occurs. |
| Instability | 1% - 3% | Component malposition (e.g., excessive retroversion); Soft tissue imbalance. | Identify etiology via CT. Revision of malpositioned components; Soft tissue plication or reconstruction. |
Patients with corticosteroid-induced AVN require meticulous preoperative optimization. Tapering of corticosteroids, if medically permissible by the prescribing specialist, can improve soft tissue healing and reduce infection risk. However, acute adrenal insufficiency must be avoided, and perioperative stress-dose steroids are frequently required.
Post Operative Rehabilitation Protocols
Rehabilitation following surgery for humeral head AVN is procedure-specific. A delicate balance must be struck between protecting the surgical repair (particularly the subscapularis in arthroplasty) and preventing adhesive capsulitis.
Core Decompression Rehabilitation
For patients undergoing core decompression without structural alteration of the joint, rehabilitation progresses relatively rapidly.
* Phase I (0-2 Weeks): Sling for comfort. Immediate initiation of pendulum exercises, passive range of motion (PROM), and active-assisted range of motion (AAROM) in all planes as tolerated. Scapular retractions and distal extremity active range of motion (AROM) are encouraged.
* Phase II (2-6 Weeks): Discontinue sling. Progress to full AROM. Initiate gentle isometric strengthening of the rotator cuff and deltoid.
* Phase III (6-12 Weeks): Advance to isotonic strengthening, focusing on periscapular stabilizers and rotator cuff endurance. Return to light functional activities.
* Phase IV (3+ Months): Unrestricted activity. Heavy lifting and impact activities are gradually reintroduced based on radiographic evidence of bone healing and absence of pain.
Arthroplasty Rehabilitation
Rehabilitation following hemiarthroplasty or TSA is heavily dictated by the subscapularis repair.
* Phase I (0-4 Weeks): Strict sling immobilization. PROM only. Forward elevation is typically limited to 130 degrees. Strict restriction: External rotation is limited to 0 to 30 degrees (based on intraoperative safe zone testing) to protect the subscapularis repair. No active internal rotation.
* Phase II (4-8 Weeks): Wean from sling. Begin AAROM and progress to AROM in forward elevation. Gradually increase external rotation parameters. Continue to avoid active internal rotation against resistance or stretching into extreme external rotation.
* Phase III (8-12 Weeks): Initiate gentle strengthening. Begin subscapularis isometrics and progress to light isotonic internal rotation. Advance deltoid and periscapular strengthening.
* Phase IV (12+ Weeks): Progressive heavy strengthening. Return to recreational activities (e.g., golf, swimming) is typically permitted between 4 to 6 months postoperatively, contingent upon the restoration of functional strength and dynamic stability.
Summary of Key Literature and Guidelines
The management of humeral head AVN has evolved significantly, guided by long-term outcome studies.
Historically, core decompression was widely applied to all early-stage lesions. However, literature by Mont et al. demonstrated that while core decompression yields excellent results (up to 80% success) in Ficat/Cruess Stage I and II disease, its efficacy drops precipitously once any structural collapse (Stage III) is present. Consequently, contemporary guidelines strictly reserve decompression for pre-collapse pathology. The addition of orthobiologics (BMAC) to core decompression remains an area of active investigation, with early studies suggesting improved survivorship of the humeral head, though high-level randomized controlled trials are still needed to establish definitive superiority over decompression alone.
For post-collapse disease, the choice between hemiarthroplasty and TSA has been a subject of considerable debate. Because patients with corticosteroid-induced AVN are often younger, surgeons historically favored hemiarthroplasty to avoid the risk of long-term glenoid component loosening. However, longitudinal studies by Feeley et al. and Schoch et al. have demonstrated that hemiarthroplasty in the setting of AVN is associated with a high rate of progressive glenoid erosion and subsequent pain, often necessitating revision.
Current academic consensus and guidelines from the American Academy of Orthopaedic Surgeons (AAOS) suggest that Total Shoulder Arthroplasty (TSA) provides more predictable, durable pain relief and superior functional outcomes compared to hemiarthroplasty for Stage IV and V disease, provided the rotator cuff is intact and sufficient glenoid bone stock exists. Modern cross-linked polyethylene glenoid components and improved cementing techniques have significantly reduced the rates of aseptic loosening, making TSA a viable and preferred option even in the younger AVN demographic. Reverse Total Shoulder Arthroplasty (rTSA) is strictly reserved as a salvage procedure for cases with concomitant massive rotator cuff tears, severe glenoid retroversion/bone loss, or revision scenarios following failed anatomic arthroplasty.
