Complex Lower Extremity Trauma: A Case Study of Gustilo IIIC Open Tibia-Fibula Fracture & Critical Ischemia
Key Takeaway
A Gustilo IIIC open tibia-fibula fracture with critical limb ischemia presents with extensive soft tissue loss, gross contamination, and neurovascular compromise, typically from high-energy trauma. Diagnosis involves detailed clinical exam, including assessing pulses and sensation, alongside urgent plain radiographs and CT angiography to define skeletal and arterial injuries.
Patient Presentation and History
Mechanism of Injury and Prehospital Course
A 42-year-old male, previously fit and active with no significant medical comorbidities, presented to the emergency department following a high-energy industrial crush injury involving a large hydraulic press. The mechanism involved the right lower extremity being trapped for approximately 45 minutes under extreme compressive forces. Pre-hospital care provided by emergency medical services included initial hemorrhage control with direct pressure and the application of a combat application tourniquet to the proximal thigh. Intravenous access was established with two large-bore peripheral lines, and the patient received initial fluid resuscitation with one liter of warmed crystalloid solution during transport.
The prolonged extrication time of 45 minutes is a critical factor in this presentation, establishing a significant baseline for warm ischemia time and severe crush syndrome pathophysiology. The combination of direct mechanical tissue destruction and subsequent ischemic hypoxia creates a complex local and systemic physiological insult, predisposing the patient to extensive rhabdomyolysis, systemic inflammatory response syndrome, and subsequent acute kidney injury if not aggressively managed.
Demographics and Medical History
Upon arrival, the patient was hemodynamically stable after initial resuscitation, though tachycardic at 110 beats per minute, with a blood pressure of 105/60 mmHg. He reported severe, unremitting pain localized to his right lower leg, poorly controlled by pre-hospital analgesia. A rapid medical history revealed no significant prior illnesses, no history of peripheral vascular disease, diabetes mellitus, or coagulopathies. There was no history of alcohol or recreational drug use, and he was a non-smoker, which is a favorable prognostic indicator for potential microvascular reconstructive efforts. His tetanus status was confirmed to be up-to-date. Social history was pertinent for a physically demanding occupation as a heavy machine operator, establishing a high functional demand for his baseline activities of daily living and occupational requirements.
Clinical Examination Findings
Advanced Trauma Life Support Primary Survey
The primary survey was conducted strictly adhering to Advanced Trauma Life Support protocols to rule out life-threatening concomitant injuries typical of high-energy industrial trauma.
Airway assessment confirmed a patent and protected airway with no evidence of maxillofacial trauma or inhalation injury. Breathing was spontaneous with symmetrical chest rise, and auscultation revealed clear breath sounds bilaterally without evidence of pneumothorax or hemothorax. Circulation assessment noted a sinus tachycardia of 110 beats per minute, a borderline hypotensive blood pressure of 105/60 mmHg, and a central capillary refill time of less than two seconds. The tourniquet remained in place on the right thigh. Radial and carotid pulses were strong and palpable bilaterally. Disability assessment demonstrated a Glasgow Coma Scale of 15, with pupils equal, round, and reactive to light. Exposure involved a full-body examination in a trauma bay heated to prevent hypothermia; no other significant injuries, seatbelt signs, or occult bleeding sources were identified apart from the right lower extremity.
Secondary Survey of the Right Lower Extremity
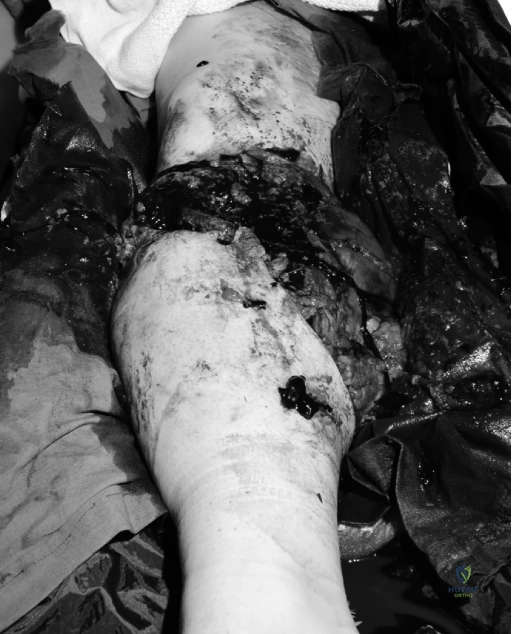
Inspection of the right lower extremity revealed a grossly deformed leg with extensive soft tissue avulsion, severe degloving injuries, and exposed, devitalized cortical bone fragments. A large open wound was noted circumferentially around the mid-shaft tibia and fibula, measuring approximately 15 centimeters in length. Significant muscle crush, necrosis, and gross contamination with industrial grease, particulate debris, and clothing fragments were visually evident within the wound bed.
The distal extremity and foot appeared severely mottled and were cool to the touch distally to the ankle joint. The pre-hospital tourniquet was transiently released under direct visualization to assess for the presence of active bleeding and to evaluate the distal vascular tree. This maneuver revealed brisk, pulsatile arterial bleeding from multiple small unnamed vessels within the zone of injury, alongside significant venous oozing, necessitating immediate reapplication and conversion to a more proximal pneumatic tourniquet inflated to 300 mmHg. There was obvious clinical shortening and external rotation of the distal limb segment. Notably, compartment swelling was significant and tense in the proximal leg, demonstrating that acute compartment syndrome can easily coexist with, and indeed be exacerbated by, obvious open wounds distally.
Palpation elicited intense, intractable tenderness over the entire mid-to-distal tibia and fibula. Significant crepitus was elicited with even the most gentle manipulation, confirming gross skeletal instability. Range of motion of the tibia and fibula was grossly unstable, demonstrating a flail limb segment. Ankle and foot range of motion could not be actively assessed due to pain, instability, and the underlying neurological deficits.
Neurological and Vascular Assessment
A comprehensive neurological and vascular assessment of the distal extremity is paramount in high-energy open fractures to determine limb viability and guide the reconstructive algorithm.
Motor examination revealed absolute paralysis, with no voluntary movement of the ankle or toes in either dorsiflexion or plantarflexion. Sensory examination demonstrated profound deficits. There was complete anesthesia in the distribution of the common peroneal nerve, affecting the dorsum of the foot and the lateral aspect of the leg. Sensation in the distribution of the superficial peroneal nerve was absent. Hypoesthesia was noted in the distribution of the tibial nerve along the plantar aspect of the foot. Crucially, deep peroneal nerve sensation, tested at the first dorsal web space, was completely absent.
Vascular assessment confirmed the absence of both dorsalis pedis and posterior tibial pulses on manual palpation. Handheld continuous-wave Doppler ultrasonography was utilized and confirmed a complete absence of biphasic or triphasic acoustic signals in both the anterior tibial and posterior tibial arterial distributions at the level of the ankle. An Ankle Brachial Index could not be reliably obtained due to the extent of the open injury and the severe distal ischemia. The foot remained cool, pale, and mottled, presenting a classic clinical picture of critical limb ischemia secondary to catastrophic vascular disruption.
Soft Tissue and Compartment Evaluation
The initial assessment of the soft tissue envelope strongly indicated a Gustilo Anderson Type IIIC open fracture of the tibia and fibula. This classification is justified by the presence of a high-energy segmental fracture, extensive soft tissue stripping and loss, gross industrial contamination, severe muscle crush injury, and, most critically, an arterial injury requiring mandatory surgical repair for limb salvage. The proximal leg compartments, specifically the anterior and deep posterior compartments, were palpably tense, raising immediate concern for impending or established acute compartment syndrome proximal to the zone of the open fracture.
Imaging and Diagnostics
Initial Plain Radiography Findings
Immediate imaging protocols focused on defining the skeletal injury architecture and assessing for concurrent occult trauma. Standard anteroposterior and lateral plain radiographs of the right tibia and fibula were obtained in the trauma bay.
The radiographs demonstrated a highly comminuted, segmental open fracture of the mid-diaphyseal tibia. The fracture pattern extended proximally into the metaphyseal region, presenting a complex zone of injury with significant cortical bone loss and severe displacement of the primary fracture fragments. Multiple butterfly fragments were visualized, displaced entirely from the periosteal envelope. Associated highly comminuted fractures of the fibula were present at multiple levels, indicating the massive kinetic energy imparted to the limb. The "missing bone" sign was evident, correlating with the extruded cortical fragments noted during the clinical inspection. There was no evidence of intra-articular extension into the tibial plateau or the tibial plafond, though the proximal metaphyseal extension complicated potential definitive intramedullary nailing strategies.
Computed Tomography Angiography Indications and Findings
Given the hard signs of vascular injury—absent distal pulses, absent Doppler signals, active pulsatile bleeding upon tourniquet release, and a profoundly ischemic distal limb—a Computed Tomography Angiography of the bilateral lower extremities with runoff was immediately indicated. In the setting of a Gustilo IIIC fracture, CTA is the gold standard for delineating the exact anatomical level of arterial transection, evaluating the presence of intimal flaps or in-situ thrombosis, and assessing the patency of the distal runoff vessels, which is critical for planning the vascular reconstruction or determining the viability of a distal bypass target.

The CTA confirmed a complete, abrupt occlusion of the anterior tibial artery approximately 4 centimeters distal to the tibioperoneal trunk. The posterior tibial artery demonstrated a long segment of severe vasospasm and irregular contrast opacification, with a focal area of contrast extravasation indicative of a partial transection or severe avulsion injury within the zone of the highly comminuted tibial fracture. The peroneal artery was non-opacified through the zone of injury. Crucially, delayed phase imaging showed faint, sluggish reconstitution of the dorsalis pedis and distal posterior tibial arteries via collateral microcirculation, confirming that the distal vascular bed remained patent but critically under-perfused.
Preoperative Templating and Surgical Planning
Preoperative planning required a synchronized, multidisciplinary approach involving Orthopedic Trauma, Vascular Surgery, and Plastic and Reconstructive Surgery. Radiographic templating was performed to determine the appropriate spanning external fixation construct. A delta frame or a biplanar construct was planned to maximize biomechanical rigidity while allowing unimpeded access to the soft tissue wounds for serial debridements, vascular repair, and eventual free tissue transfer. The length of the tibial defect was estimated at 6 centimeters, indicating that primary shortening or immediate bone grafting would be impossible, and a staged induced membrane technique or bone transport would eventually be required if limb salvage was pursued.
Differential Diagnosis
In the context of severe lower extremity crush trauma presenting with ischemia and extensive tissue damage, the diagnosis of a Gustilo IIIC open fracture is clinically apparent. However, the differential diagnosis process in this scenario focuses on distinguishing the primary cause of the ischemia and evaluating the viability of limb salvage versus the necessity of primary amputation.
| Diagnostic Consideration | Key Clinical Features | Imaging and Diagnostic Findings | Management Strategy Implications |
|---|---|---|---|
| Gustilo Anderson Type IIIC Open Fracture | High-energy open fracture, massive soft tissue destruction, hard signs of vascular injury (absent pulses, ischemia), gross contamination. | Plain films show comminuted/segmental fractures. CTA confirms arterial transection or occlusion requiring repair. | Immediate operative intervention. Damage control orthopedics, vascular shunting/repair, prophylactic fasciotomies, serial debridements. |
| Gustilo Anderson Type IIIB Open Fracture with Severe Spasm | High-energy open fracture, extensive soft tissue stripping requiring flap coverage. Pulses may be diminished but limb is not critically ischemic. | Plain films show severe fractures. CTA shows patent vessels, potentially with localized vasospasm, but intact distal runoff without transection. | Urgent operative debridement and skeletal stabilization. Vascular repair is not mandated. Soft tissue coverage planned within 3-5 days. |
| Isolated Acute Compartment Syndrome | Tense, woody compartments, pain out of proportion to injury, pain with passive stretch. Pulses are typically PRESENT until very late stages. | Intracompartmental pressure monitoring > 30 mmHg or Delta P < 30 mmHg. Arterial imaging is usually normal unless concomitant injury exists. | Emergent four-compartment fasciotomies. Skeletal stabilization if associated with a fracture. |
| Mangled Extremity Requiring Primary Amputation | Unsalvageable soft tissue, complete disruption of the posterior tibial nerve, warm ischemia > 6 hours, severe systemic instability. | Severe skeletal destruction on plain films. CTA shows extensive, multi-level vascular destruction with no viable distal targets. | Resuscitation, life-saving primary amputation (e.g., through-knee or above-knee) to prevent lethal systemic complications. |
Surgical Decision Making and Classification
Gustilo Anderson Classification and Prognostic Implications
The injury was definitively classified as a Gustilo Anderson Type IIIC open tibia and fibula fracture. This classification carries immense prognostic weight. Type IIIC injuries are defined not merely by the size of the soft tissue defect, but by the presence of an arterial injury that necessitates surgical repair to ensure limb viability, regardless of the soft tissue wound dimensions. Historically, these injuries carry a high risk of amputation, deep infection, non-union, and severe long-term functional impairment. The presence of profound contamination and crush mechanism further elevates the risk of clostridial myonecrosis and osteomyelitis.
Mangled Extremity Severity Score Evaluation
To objectify the decision between limb salvage and primary amputation, scoring systems such as the Mangled Extremity Severity Score are frequently utilized, albeit with an understanding of their limitations.
Evaluating this patient:
1. Skeletal / Soft Tissue Injury: High energy, crush injury (Score: 3).
2. Limb Ischemia: Pulseless, paresthetic, diminished capillary refill. Given the 45-minute extrication and transport time, warm ischemia was approaching 2-3 hours at the time of surgical decision making (Score: 2).
3. Shock: Transiently hypotensive in the field, currently hemodynamically stable but tachycardic (Score: 1).
4. Age: 42 years old (Score: 1).
The calculated Mangled Extremity Severity Score was 7. A score of 7 or higher historically suggests that amputation is likely, and a score of 8 is highly predictive of amputation. However, modern literature, specifically the Lower Extremity Assessment Project study, has demonstrated that no scoring system is perfectly predictive of functional outcomes or the absolute necessity of amputation.
Multidisciplinary Limb Salvage Versus Amputation Discussion
The decision to proceed with limb salvage was made following a rapid multidisciplinary consultation. Factors favoring salvage included the patient's relatively young age, excellent baseline physiological reserve, absence of medical comorbidities, and the fact that the warm ischemia time was still under the critical 6-hour threshold, albeit narrowing rapidly. Factors favoring amputation included the severe crush mechanism, complete loss of deep peroneal and tibial nerve sensation (though this can be due to ischemia rather than anatomical transection), and the extensive bone loss.
Following a frank discussion with the patient regarding the high likelihood of multiple complex surgeries, prolonged rehabilitation, the risk of chronic pain, and the possibility of delayed secondary amputation if salvage failed, informed consent was obtained for an aggressive limb salvage attempt, including damage control orthopedics, vascular reconstruction, and potential soft tissue free flap transfer, with the explicit understanding that intraoperative findings might still dictate a primary amputation to save his life.
Surgical Technique and Intervention
Patient Positioning and Operating Room Setup
The patient was transported emergently to the operating room. Under general endotracheal anesthesia, he was positioned supine on a radiolucent trauma table. A bump was placed under the ipsilateral hip to correct external rotation and allow access to the lateral compartments. The entire right lower extremity, from the iliac crest to the toes, was prepped and draped in a standard sterile fashion. The contralateral left lower extremity was also prepped and draped circumferentially to serve as a donor site for a reversed great saphenous vein graft. A sterile pneumatic tourniquet was positioned on the proximal right thigh but remained uninflated initially to allow continuous assessment of tissue perfusion during debridement.
Initial Debridement and Damage Control Orthopedics
The paramount initial step in managing a Gustilo IIIC fracture is radical, aggressive debridement of all devitalized tissue. The zone of injury was extended longitudinally to fully expose the compromised compartments.
Skin and subcutaneous tissues were sharply excised until healthy, bleeding edges were encountered. The deep fascia was opened extensively. Muscle viability was assessed using the classic "4 C's" criteria: Color, Consistency, Contractility, and Capacity to bleed. The crush mechanism had resulted in severe myonecrosis of the anterior tibialis and extensor digitorum longus muscles, which were radically excised back to bleeding, contractile tissue. The deep posterior compartment was explored, and while contused, the flexor hallucis longus and tibialis posterior demonstrated partial viability and were preserved.
Skeletal debridement involved the removal of all avascular cortical bone fragments devoid of soft tissue attachments. The fractured bone ends were curetted and irrigated until punctate cortical bleeding (the "paprika sign") was observed, indicating viable bone. This radical skeletal debridement resulted in a definitive diaphyseal bone defect measuring approximately 6.5 centimeters. Copious irrigation was performed using 9 liters of normal saline delivered via low-pressure gravity flow to mechanically remove industrial grease and particulate debris.
Vascular Exploration and Reconstruction
Following the initial damage control debridement, the vascular surgery team proceeded with exploration of the neurovascular bundle. The anterior tibial artery was found to be completely avulsed and thrombosed over a 5-centimeter segment within the zone of maximum skeletal injury. The posterior tibial artery was intact but severely contused with an intimal tear and localized thrombosis.
Given the critical ischemia, a temporary intraluminal vascular shunt (Argyle shunt) was immediately placed into the posterior tibial artery to restore distal perfusion rapidly and limit further warm ischemia time. Once distal flow was re-established, attention turned to definitive repair. The severely damaged segment of the posterior tibial artery was resected. A reversed great saphenous vein graft was harvested from the contralateral left thigh. An interposition vein graft was then anastomosed end-to-end to the native posterior tibial artery using 6-0 Prolene sutures under loupe magnification. The anterior tibial artery was deemed unsalvageable and was ligated proximally and distally. Upon release of the vascular clamps, excellent pulsatile flow was observed through the vein graft, and the distal foot immediately demonstrated improved color and robust capillary refill.
Temporary Skeletal Stabilization with External Fixation
With vascular flow restored, rigid skeletal stabilization was absolutely critical to protect the delicate microvascular anastomosis and prevent further soft tissue trauma. Due to the massive contamination and the need for serial debridements, internal fixation was strictly contraindicated.
A spanning knee-to-ankle external fixator was applied. Two 5.0mm hydroxyapatite-coated Schanz pins were placed percutaneously into the proximal tibial metaphysis, taking care to avoid the joint capsule and the common peroneal nerve. Two additional 5.0mm Schanz pins were placed into the medial cuneiform and the calcaneal tuberosity to span the ankle joint and control the distal segment.
The fracture was reduced longitudinally to restore anatomical length, alignment, and rotation, utilizing the contralateral leg as a visual reference. The external fixator was assembled using a delta frame configuration with carbon fiber rods to maximize multiplanar stability. The rigid construct successfully neutralized bending and torsional forces, effectively protecting the posterior tibial vein graft from mechanical disruption.
Prophylactic Fasciotomies and Soft Tissue Management
Despite the massive open wound, the proximal compartments remained at high risk for acute compartment syndrome, particularly following the ischemia-reperfusion injury subsequent to the vascular repair.
A standard dual-incision, four-compartment fasciotomy of the right lower leg was performed. A lateral incision was utilized to decompress the anterior and lateral compartments, ensuring the superficial peroneal nerve was identified and protected. A medial incision was utilized to decompress the superficial and deep posterior compartments. The soleus bridge was carefully detached from the tibia to ensure complete release of the deep posterior compartment.
The massive open wound, now encompassing the traumatic defect and the fasciotomy incisions, was managed with advanced wound care. A Negative Pressure Wound Therapy system was applied. Reticulated open-cell polyurethane foam was carefully tailored to fit the defect. A non-adherent contact layer was placed directly over the exposed neurovascular bundle and the vein graft to prevent catastrophic erosion or disruption by the negative pressure sponge. The system was sealed and set to -125 mmHg continuous pressure to manage exudate, reduce interstitial edema, and promote microvascular angiogenesis.
Definitive Fixation Strategy and Flap Coverage
The patient returned to the operating room at 48 hours and 96 hours post-injury for planned, serial debridements. During these "second look" and "third look" procedures, marginal tissue necrosis was further excised. By the third procedure, the wound bed was deemed clean, with healthy granulation tissue and no evidence of purulence or progressive myonecrosis.
At day 7 post-injury, the patient underwent definitive soft tissue coverage. Due to the extensive size of the defect and the exposed tibia and vascular graft, local rotational flaps (such as a gastrocnemius or soleus flap) were inadequate. The plastic and reconstructive surgery team performed a free tissue transfer utilizing an Anterolateral Thigh myocutaneous flap harvested from the contralateral leg. The descending branch of the lateral circumflex femoral artery was anastomosed end-to-side to the proximal patent segment of the anterior tibial artery, and the venae comitantes were anastomosed to the deep venous system.
The external fixator was maintained as the definitive stabilization construct for the initial phase of healing, as the 6.5-centimeter bone defect would require a staged reconstruction. The plan was established to utilize the Masquelet technique (induced membrane technique). During the flap coverage procedure, a polymethylmethacrylate cement spacer, impregnated with vancomycin and tobramycin, was placed into the segmental bone defect to maintain the space and induce the formation of a biologically active pseudosynovial membrane.
Post Operative Protocol and Rehabilitation
Intensive Care Management and Flap Monitoring
Postoperatively, the patient was transferred to the Surgical Intensive Care Unit. The primary focus was aggressive hemodynamic monitoring to ensure optimal perfusion of the free flap and the repaired posterior tibial artery. Mean arterial pressure was maintained strictly above 65 mmHg, and vasopressors were avoided whenever possible to prevent peripheral vasoconstriction.
Flap monitoring was conducted continuously using an implantable venous Doppler probe and clinical checks every hour for the first 48 hours, assessing flap color, temperature, capillary refill, and turgor. The patient was placed on a strict warming protocol to prevent hypothermia-induced vasospasm.
Due to the massive crush injury and subsequent ischemia-reperfusion, the patient was at extreme risk for severe rhabdomyolysis. Intravenous fluids were titrated to maintain a urine output of greater than 100 mL/hour. Urine myoglobin levels and serum creatine kinase were monitored serially. Alkalinization of the urine with sodium bicarbonate infusions was initiated to prevent precipitation of myoglobin in the renal tubules, successfully averting acute kidney injury.
Weight Bearing Status and Physical Therapy
The patient was maintained on strict non-weight-bearing status for the right lower extremity. Deep vein thrombosis prophylaxis was initiated with low molecular weight heparin once surgical hemostasis was assured, given the high risk of thromboembolism in complex lower extremity trauma.
Physical therapy was consulted on postoperative day 1. Early rehabilitation focused on aggressive range of motion of the bilateral upper extremities and the uninjured left lower extremity to prevent deconditioning. Once the free flap was deemed stable (postoperative day 5), gentle, passive range of motion of the right knee and toes was initiated. The external fixator spanning the ankle precluded ankle range of motion, but isometric quadriceps and hamstring exercises were encouraged to maintain muscle tone.
Long Term Follow Up and Bone Grafting Considerations
The patient was discharged to an acute inpatient rehabilitation facility on postoperative day 21. His long-term reconstructive algorithm is complex.
At approximately 8 to 10 weeks post-injury, once the soft tissue envelope is fully healed and stable, the patient will return to the operating room for the second stage of the Masquelet technique. The external fixator may be exchanged for a definitive locking intramedullary nail if the proximal fracture extension allows, or a circular Ilizarov frame may be applied. The antibiotic cement spacer will be carefully removed, preserving the induced biological membrane. The 6.5-centimeter defect will then be filled with copious autologous cancellous bone graft, typically harvested from the bilateral anterior and posterior iliac crests using the Reamer-Irrigator-Aspirator system to maximize graft volume and osteogenic potential.
Following the bone grafting, the patient will face a prolonged period of protected weight-bearing and aggressive physical therapy. The ultimate functional outcome remains guarded, as the initial ischemic insult and nerve damage may result in persistent neuropathic pain, foot drop, and altered gait mechanics, potentially requiring a future ankle-foot orthosis or delayed tendon transfers.
Clinical Pearls and Pitfalls
Key Takeaways for High Energy Open Fractures
- Time is Tissue in Vascular Compromise: In Gustilo IIIC injuries, the clock starts at the time of injury, not at presentation. Rapid identification of vascular disruption and minimizing warm ischemia time via temporary shunting is paramount to limb survival.
- Radical Debridement is the Foundation of Success: Inadequate debridement is the primary cause of deep infection and reconstructive failure. The "steak test" (evaluating muscle color, consistency, contractility, and bleeding) must be applied ruthlessly. It is better to create a larger defect of healthy tissue than to leave a smaller defect containing necrotic nidus.
- Multidisciplinary Collaboration is Mandatory: Complex limb salvage cannot be managed in a silo. Early, simultaneous involvement of Orthopedic Trauma, Vascular Surgery, and Reconstructive Microsurgery ensures a cohesive strategy that addresses bone, blood flow, and soft tissue coverage concurrently.
- Rigid Fixation Protects the Anastomosis: The primary role of the external fixator in the acute phase of a IIIC injury is not just fracture alignment, but absolute mechanical protection of the delicate microvascular repairs and vein grafts.
Common Errors in Gustilo IIIC Management
*
