Masterclass: Surgical Management of Simple Elbow Dislocation & LUCL Repair
Key Takeaway
This masterclass provides an exhaustive, immersive guide to managing simple elbow dislocations. Fellows will learn intricate pathoanatomy, preoperative planning, and a granular, step-by-step intraoperative execution of Lateral Ulnar Collateral Ligament (LUCL) repair. We cover crucial anatomical landmarks, neurovascular precautions, instrument usage, and advanced techniques for restoring elbow stability, along with comprehensive postoperative protocols.
Comprehensive Introduction and Patho-Epidemiology
The management of the simple elbow dislocation represents a fundamental pillar of upper extremity trauma surgery. By strict orthopaedic definition, a "simple" dislocation refers to a complete disruption of the ulnohumeral articulation in the absence of concomitant fractures to the radial head, olecranon, or coronoid process. However, the nomenclature is inherently deceptive; the soft tissue derangement required to permit a complete dislocation is profound, and the subsequent biomechanical consequences are anything but simple. The elbow is the second most commonly dislocated major joint in the adult population, trailing only the glenohumeral joint, and the most frequently dislocated joint in the pediatric demographic. Mastering the precise management of these injuries is paramount for any orthopaedic surgeon, as failure to restore the delicate interplay between osseous congruity and ligamentous tension invariably leads to chronic instability, early-onset post-traumatic osteoarthritis, or debilitating stiffness.
The primary objective of this masterclass chapter is to meticulously delineate the pathoanatomy, biomechanics, and surgical management of elbow instability, with a specific focus on the lateral ulnar collateral ligament (LUCL). The LUCL is the critical restraint against posterolateral rotatory instability (PLRI) and is almost universally disrupted in the setting of a complete dislocation. Our surgical goal is rarely just the acute reduction of the joint; rather, it is the anatomic restoration of the highly sophisticated biomechanics that govern elbow kinematics. An intimately reduced and stable elbow allows for the early implementation of active range of motion, which is the singular most effective strategy for mitigating post-traumatic stiffness—the historical bane of elbow trauma management.
Most simple elbow dislocations are sustained via a fall on an outstretched hand (FOOSH). The classic mechanism involves a highly specific, sequential application of forces: an axial load transmitted through the forearm, coupled with supination and a valgus bending moment. This triad of forces drives the radial head posteriorly relative to the capitellum, initiating a cascade of soft tissue failure that predictably begins on the lateral side of the joint and propagates medially. This concept, often referred to as the Horii circle, dictates that the lateral collateral ligamentous complex (specifically the LUCL) is the first structure to fail, followed by the anterior and posterior joint capsule, and finally culminating in the disruption of the medial collateral ligament (MCL) if the deforming forces persist.
Understanding this sequence is critical for both non-operative and operative decision-making. While many simple dislocations can be managed non-operatively if concentric stability is achieved and maintained through a functional arc of motion, a significant subset of patients will demonstrate persistent instability. This is particularly true when the LUCL fails to heal isometrically or when iatrogenic injury to the lateral complex occurs during procedures such as tennis elbow debridement or radial head excision. In these scenarios, the surgeon must be prepared to execute a flawless LUCL repair or reconstruction to prevent the insidious progression of PLRI, which can severely compromise the patient's functional independence and quality of life.
Detailed Surgical Anatomy and Biomechanics
The stability of the ulnohumeral and radiocapitellar articulations relies upon a highly sophisticated, synergistic relationship between static osseous architecture, static ligamentous restraints, and dynamic muscular forces. Understanding this hierarchy of stability is the foundation of successful surgical reconstruction.
Primary Stabilizers: Osseous and Ligamentous
The primary stabilizers of the elbow joint are the osseous articulation of the ulnohumeral joint, the medial collateral ligament (MCL) complex, and the lateral collateral ligament (LCL) complex. The highly congruent, deep articulation of the greater sigmoid notch of the ulna with the spool-shaped trochlea of the humerus provides immense inherent structural stability, particularly in the anteroposterior plane and during terminal flexion and extension. The coronoid process acts as a critical anterior buttress, preventing posterior translation of the ulna.
The ligamentous complexes provide critical stability against varus, valgus, and rotatory stresses. The anterior band of the medial collateral ligament (aMCL) originates from the anterior inferior surface of the medial epicondyle and inserts broadly onto the sublime tubercle of the proximal ulna. It is the primary restraint to valgus stress across the elbow, particularly functioning between 30 and 120 degrees of flexion. Conversely, the lateral collateral ligament complex consists of the radial collateral ligament, the annular ligament, and the lateral ulnar collateral ligament (LUCL). The LUCL is the undisputed primary restraint to posterolateral rotatory instability. It originates from a precise isometric point on the lateral epicondyle—specifically, the center of the capitellar axis of rotation—and courses distally and posteriorly, blending with the annular ligament to insert onto the supinator crest of the proximal ulna.
Secondary Stabilizers and Dynamic Constraints
When the primary stabilizers are compromised, the joint becomes highly reliant on secondary stabilizers. The radial head is the most critical secondary stabilizer, providing up to 30% of the resistance to valgus stress in an intact elbow, but becoming the primary valgus buttress in the setting of an aMCL deficiency. Excision of a fractured radial head in the presence of an unrecognized MCL tear will predictably result in catastrophic valgus instability. The anterior joint capsule also serves as a secondary restraint to hyperextension and valgus stress when the elbow is in full extension.
Dynamic stability is conferred by the muscular forces crossing the joint. The flexor-pronator mass medially and the extensor-supinator mass laterally generate compressive forces across the joint surfaces, effectively increasing the congruency and stability of the articulation during active motion. The anconeus and triceps also play a role in dynamic stabilization against varus and posterolateral rotatory forces. During surgical approaches, meticulous preservation of these dynamic muscular origins is essential to avoid compounding the inherent instability of the traumatized joint.

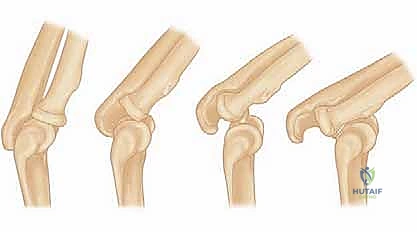
FIG 1 • A. Posterolateral rotatory instability follows a typical progression of disruption, allowing the joint to become perched and then dislocate as soft tissue injury progresses.
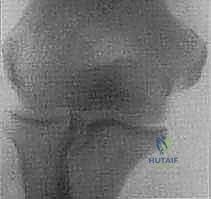
FIG 1 • B. Intraoperative photograph demonstrating avulsion of the origin of the lateral ulnar collateral ligament (LUCL) after traumatic dislocation of the elbow. The origin of the LUCL and the extensor muscles are avulsed as one layer, held by the forceps.
Pathophysiology and the Spectrum of Instability
Posterolateral Rotatory Instability (PLRI)
The concept of posterolateral rotatory instability (PLRI), brilliantly elucidated by O’Driscoll, describes a three-stage spectrum of pathoanatomy that dictates the clinical presentation of elbow instability. Stage 1 involves isolated disruption of the LUCL, resulting in subluxation of the radial head posterior to the capitellum. Clinically, this manifests as a positive pivot-shift test but rarely presents as a frank dislocation. Stage 2 involves progression of the tear anteriorly and posteriorly through the capsule, allowing the coronoid to "perch" on the trochlea.
Stage 3 is a complete dislocation. Stage 3A involves disruption of all soft tissues except the anterior band of the MCL, allowing the elbow to pivot on the intact aMCL. Stage 3B involves tearing of the aMCL, resulting in severe, multi-directional instability. Stage 3C represents a catastrophic stripping of the entire distal humerus, including the flexor-pronator mass. Understanding these stages is vital; a patient with a Stage 1 injury may only complain of vague lateral elbow pain and clicking when pushing up from a chair, whereas a Stage 3 injury presents as a gross deformity in the emergency department.
Complex Instability Variants: PMRI and the Terrible Triad
While this chapter focuses on simple dislocations, the surgeon must be hyper-vigilant in ruling out complex instability patterns. The "Terrible Triad"—a combination of an elbow dislocation, a radial head fracture, and a coronoid fracture—is notoriously difficult to manage and practically guarantees recurrent instability if treated non-operatively.
Furthermore, Posteromedial Rotatory Instability (PMRI) is an increasingly recognized variant characterized by an avulsion of the LCL complex combined with a fracture of the anteromedial facet of the coronoid. This injury occurs via an axial load combined with a varus and posteromedial rotatory force. Because the radial head is typically intact, plain radiographs may only show a subtle loss of joint space or a tiny fleck of bone anterior to the joint. Missing a PMRI pattern will lead to rapid, destructive varus-induced osteoarthritis of the ulnohumeral joint.
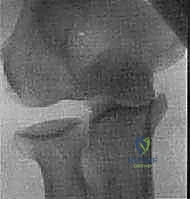
FIG 1 • C. Posteromedial rotatory instability is a variant of elbow instability in which the elbow dislocates, rupturing the LUCL, and the medial coronoid sustains an impaction fracture.
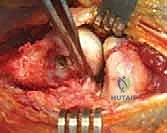
FIG 1 • E. Impaction fracture can be seen on the 3D CT reconstruction.
Exhaustive Indications and Contraindications
The decision to proceed with operative intervention for a simple elbow dislocation hinges upon the clinical assessment of post-reduction stability. The vast majority of acute simple dislocations can be managed non-operatively with a brief period of immobilization followed by early, protected range of motion. However, surgical intervention is absolutely mandated when concentric stability cannot be maintained within a functional arc of motion.
Indications for acute surgical repair include an elbow that remains subluxated or dislocated despite a technically sound closed reduction. Furthermore, if the elbow re-dislocates or subluxates in greater than 30 to 45 degrees of flexion (even when the forearm is fully pronated to tension the medial structures), the lateral sided restraints are completely incompetent and require surgical fixation. Entrapment of soft tissue or neurovascular structures within the joint space, preventing concentric reduction, is an absolute indication for immediate open exploration. In the chronic setting, symptomatic recurrent PLRI that has failed a dedicated course of physical therapy (focused on supinator and extensor strengthening) is a strong indication for LUCL reconstruction.
Contraindications to surgical intervention are generally limited to systemic factors or severe local tissue compromise. Active local infection, severe medical comorbidities precluding anesthesia, or a profoundly non-compliant patient who will not adhere to strict post-operative rehabilitation protocols represent absolute contraindications. Relative contraindications include severe osteopenia, which may compromise suture anchor fixation, and massive soft tissue degloving injuries, which may necessitate delayed reconstruction once the soft tissue envelope has declared itself.
| Category | Indications for Surgical Intervention | Contraindications to Surgery |
|---|---|---|
| Acute Setting | Non-concentric reduction post-manipulation | Active local or systemic infection |
| Instability > 30° flexion with forearm pronated | Severe medical comorbidities (ASA IV/V) | |
| Entrapped intra-articular loose body or soft tissue | Poor soft tissue envelope / blistering | |
| Post-reduction neurovascular deficit (e.g., Ulnar nerve) | Patient non-compliance / psychiatric instability | |
| Chronic Setting | Symptomatic recurrent PLRI | Painless, functional instability (low demand) |
| Positive lateral pivot-shift test under anesthesia | Severe, end-stage post-traumatic osteoarthritis | |
| Failure of > 6 months of conservative physiotherapy | Isolated, asymptomatic radiographic subluxation |
Pre-Operative Planning, Templating, and Patient Positioning
Clinical and Radiographic Evaluation
Meticulous pre-operative planning begins in the trauma bay. A rigorous neurovascular examination is non-negotiable. The ulnar nerve is particularly vulnerable to traction neuropraxia during a posterolateral dislocation, and its status must be documented prior to any reduction attempt. Following closed reduction, standard orthogonal anteroposterior (AP) and lateral radiographs must be scrutinized. The surgeon must look for the "drop sign"—an increase in the ulnohumeral distance on the lateral radiograph—which indicates profound ligamentous incompetence.
If there is any suspicion of a subtle coronoid or radial head fracture, a non-contrast Computed Tomography (CT) scan with 3D reconstructions is mandatory. Magnetic Resonance Imaging (MRI) is rarely indicated in the acute setting, as the diagnosis of ligamentous disruption is inferred from the dislocation itself; however, MRI is highly valuable in the chronic setting to map the quality of the LUCL remnants and assess for chondral damage prior to reconstruction.
Examination Under Anesthesia (EUA) and Fluoroscopy
The true magnitude of instability is often masked by muscular guarding in the awake patient. Therefore, an Examination Under Anesthesia (EUA) coupled with dynamic fluoroscopy is the most critical step prior to making the final surgical incision. The surgeon must systematically assess the joint through its entire flexion-extension arc.
The lateral pivot-shift maneuver is the hallmark test for PLRI. With the patient's arm overhead, the surgeon applies a supination torque, a valgus stress, and an axial load while slowly flexing the elbow from full extension. In an LUCL-deficient elbow, the radial head will subluxate posterolaterally. As flexion reaches roughly 40 degrees, the tension in the intact triceps and medial structures will suddenly pull the joint back into anatomic alignment with a palpable and visible "clunk." This maneuver should be captured on live fluoroscopy to confirm the diagnosis and document the pathology.
FIG 2 • A. The lateral pivot-shift maneuver is performed with the patient’s arm positioned overhead, and a supination–valgus stress is applied.
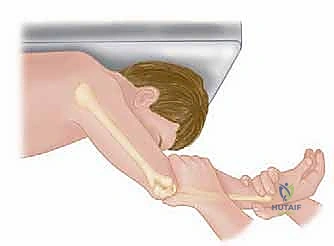
FIG 2 • B. Fluoroscopic image demonstrating posterior subluxation of the radial head relative to the capitellum during the lateral pivot-shift maneuver.
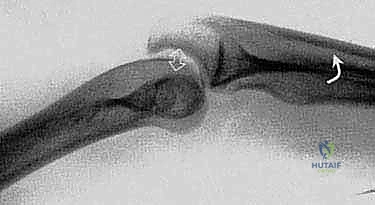
FIG 2 • C. Fluoroscopic image demonstrating concentric reduction of the radiocapitellar joint after the "clunk" is felt during continued flexion.
Patient Positioning and Setup
Surgical positioning must allow for unencumbered access to the lateral aspect of the elbow and unimpeded fluoroscopic imaging. The patient is typically positioned supine. A non-sterile tourniquet is applied high on the brachium. The arm is draped free and brought across the patient's chest. A sterile bump or a specialized arm positioner can be utilized to hold the elbow in flexion. Alternatively, the lateral decubitus position with the arm draped over a radiolucent post provides excellent exposure for both the lateral and medial sides of the elbow, though it requires more setup time. The C-arm should be brought in parallel to the operating table, allowing for easy transitions between AP and lateral imaging without moving the patient's arm excessively.
Step-by-Step Surgical Approach and Fixation Technique
The Surgical Approach
Surgical access to the lateral collateral ligament complex can be achieved via the Kocher or the Kaplan interval. The Kocher approach utilizes the internervous plane between the extensor carpi ulnaris (ECU, innervated by the posterior interosseous nerve) and the anconeus (innervated by the radial nerve). This is the traditional and most commonly utilized approach for LUCL repair. The skin incision is centered over the lateral epicondyle and extended obliquely towards the supinator crest of the ulna.
Alternatively, the Kaplan approach exploits the interval between the extensor digitorum communis (EDC) and the extensor carpi radialis brevis (ECRB). While this provides more direct access to the anterior aspect of the radiocapitellar joint, it places the posterior interosseous nerve (PIN) at slightly higher risk if the dissection strays distally. Regardless of the chosen interval, the common extensor origin is often found to be partially or completely avulsed from the lateral epicondyle in the setting of a traumatic dislocation, providing immediate, albeit pathologic, access to the underlying ligamentous complex.
FIG 3 • A. Surgical exposure of the lateral elbow demonstrating the avulsed LUCL and common extensor origin from the lateral epicondyle.
Identification and Preparation of the Footprint
Upon entering the joint, the surgeon must evacuate the hematoma and thoroughly irrigate the articulation to remove any osteochondral debris. The LUCL is meticulously inspected. In acute traumatic dislocations, the LUCL is almost universally avulsed from its humeral origin on the lateral epicondyle, often taking a small fleck of bone or the common extensor tendon with it. Intrasubstance tears or ulnar avulsions are distinctly less common.
The anatomic footprint of the LUCL on the lateral epicondyle must be identified and debrided of soft tissue to create a bleeding bony bed that will promote robust ligamentous healing. The precise isometric point is located at the center of the capitellar arc of rotation, which is typically just anterior and inferior to the apex of the lateral epicondyle. Identifying this precise isometric point is the single most critical technical step in the operation. If the repair is placed too anteriorly, the ligament will be tight in flexion and loose in extension; if placed too posteriorly, it will be tight in extension and loose in flexion, leading to a profound loss of motion or recurrent instability.
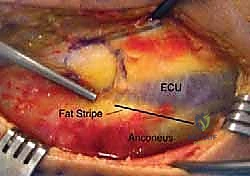
FIG 3 • B. Placement of a suture anchor at the isometric point on the lateral epicondyle for LUCL repair.
Fixation Technique: Acute Repair
For acute repairs where the native tissue is robust, primary repair using suture anchors is the gold standard. A double-loaded suture anchor (typically 3.5mm to 4.5mm in diameter) is deployed into the prepared isometric point on the lateral epicondyle. The sutures are then passed through the avulsed edge of the LUCL and the common extensor origin using a locked Krackow or grasping whipstitch configuration.
Before committing to the final knots, an isometricity check must be performed. The surgeon pulls tension on the sutures while taking the elbow through a full range of motion. The suture should maintain a constant tension without stretching or going completely slack. Once isometricity is confirmed, the elbow is positioned in 30 to 40 degrees of flexion with the forearm in maximum pronation. This position closes the lateral joint space and takes tension off the repair. The knots are then tied securely, docking the ligamentous tissue firmly against the bleeding bone of the epicondyle.
FIG 4 • A. Passing sutures through the LUCL and common extensor mass utilizing a grasping stitch configuration.
FIG 4 • B. Intraoperative view showing the tensioning of the repair prior to final knot tying.
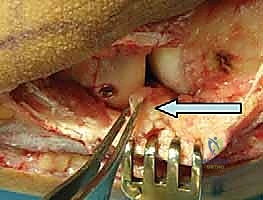
FIG 4 • C. Final knot tying with the elbow held in flexion and pronation to protect the lateral repair.
Fixation Technique: Chronic Reconstruction
In cases of chronic instability or severely attenuated acute tissue, primary repair is destined to fail, and a formal LUCL reconstruction using an autograft or allograft is required. The palmaris longus autograft is the graft of choice, though gracilis or triceps fascia can be utilized.
The reconstruction typically employs a docking technique or a figure-of-eight configuration. Bone tunnels are drilled at the supinator crest of the ulna (the insertion footprint) and at the isometric point on the lateral epicondyle. The graft is passed through the ulnar tunnels, shuttled across the joint to recreate the trajectory of the native LUCL, and docked into the humeral tunnel. Tensioning is performed in the same manner as an acute repair—elbow in 30-40 degrees of flexion and full pronation—before securing the graft with an interference screw or tying it over a bony bridge.
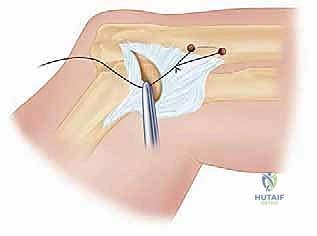
FIG 5 • A. Preparation for LUCL reconstruction utilizing a graft passed through ulnar bone tunnels.
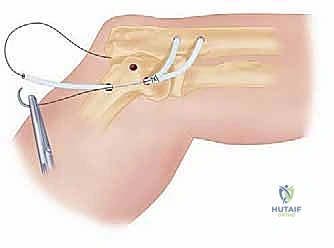
FIG 5 • B. The graft is passed and tensioned across the radiocapitellar joint to restore stability.
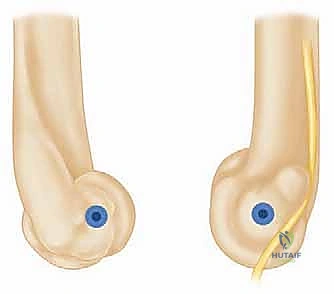
FIG 5 • C. Final appearance of an LUCL reconstruction with the graft secured firmly into the humeral isometric point.
Complications, Incidence Rates, and Salvage Management
Surgical management of elbow instability is technically demanding, and complications are not infrequent. The surgeon must be adept at anticipating, preventing, and managing these adverse events.
Post-traumatic stiffness is the most ubiquitous complication following elbow trauma, affecting up to 30% of patients regardless of whether they are treated operatively or non-operatively. The elbow capsule is exquisitely sensitive to trauma and rapidly forms dense, fibrotic adhesions. The most effective prevention is the achievement of a stable joint that permits early active range of motion. If stiffness persists beyond 6 months despite aggressive therapy, an open or arthroscopic capsular release may be necessary. Heterotopic ossification (HO) is another significant driver of stiffness. Prophylaxis with NSAIDs (e.g., Indomethacin) or single-dose localized radiation therapy should be considered in high-risk patients, particularly those with concomitant head trauma or severe muscular crush injuries.
Recurrent instability occurs in approximately 5-10% of surgically managed cases. This is almost exclusively due to technical errors during surgery, most notably the failure to identify the true isometric point on the lateral epicondyle. If the anchor is placed non-isometrically, the repair will inevitably stretch out or rupture during early mobilization. Salvage for recurrent instability requires a
