Mastering Axillary Space Resections: An Intraoperative Guide to Complex Tumor Excision and Neurovascular Preservation
Key Takeaway
Welcome, fellows, to an immersive masterclass on axillary space exploration and tumor resection. This complex region, housing vital neurovascular structures, demands meticulous technique. We'll navigate the intricate anatomy, from muscular boundaries to the brachial plexus and axillary vessels, ensuring precise tumor excision while preserving limb function. Our focus: comprehensive preoperative planning, exact patient positioning, and a granular, step-by-step intraoperative execution, emphasizing critical pearls and pitfalls for successful limb-sparing outcomes.
Welcome to the definitive guide on axillary space exploration and tumor resection. In the realm of orthopaedic oncology, this anatomical region represents one of the most challenging yet profoundly rewarding operative landscapes. This is not merely a technical exercise in tissue removal; it is an intricate, high-stakes dance of anatomical precision, meticulous neurovascular dissection, and critical intraoperative decision-making. The axilla, often a silent harbor for aggressive soft tissue and bone malignancies, demands our utmost respect and a profound understanding of its three-dimensional architecture. Our overarching goal remains steadfast: limb preservation and functional optimization, but never at the expense of oncologic radicality and patient survival.
Comprehensive Introduction and Patho-Epidemiology
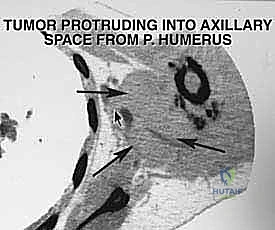
The axillary space is a complex, multi-layered anatomical conduit that serves as the primary gateway for the neurovascular supply to the upper extremity. Pathologies arising within or extending into this space present unique surgical dilemmas. Primary soft tissue sarcomas, such as undifferentiated pleomorphic sarcoma, synovial sarcoma, and liposarcoma, frequently manifest in the deep muscular compartments bordering the axilla. Furthermore, primary bone sarcomas of the proximal humerus or scapula, including osteosarcoma and Ewing sarcoma, often exhibit massive extraosseous components that invade the axillary pyramid.
Historically, aggressive malignancies involving the axillary space were managed with radical forequarter amputations to ensure adequate oncologic margins. However, the modern era of orthopaedic oncology has witnessed a paradigm shift toward limb-sparing surgery. This evolution has been propelled by advancements in neoadjuvant chemotherapy, refined radiation therapy protocols, and the development of sophisticated microsurgical reconstruction techniques. Today, limb salvage is the standard of care, provided that a functional extremity can be maintained and negative margins (R0 resection) can be achieved.
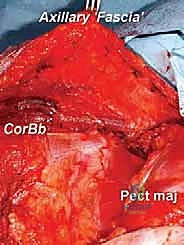
Despite these advancements, the axilla remains an unforgiving anatomical zone. The intimate relationship between expanding tumor masses and the brachial plexus, axillary vessels, and regional lymphatic basins requires an exhaustive preoperative diagnostic workup and a masterful surgical approach. Metastatic disease, particularly from breast carcinoma or melanoma, frequently involves the axillary lymph nodes, creating large, matted, and highly vascular masses that necessitate careful en bloc resection.
The surgical philosophy dictating axillary resections must balance the aggressive eradication of microscopic disease with the preservation of critical neurovascular structures. Surgeons must be prepared for complex reconstructive procedures, including vascular grafting and regional muscle flap transfers, to restore continuity and provide adequate soft-tissue coverage. Mastery of this space requires not only technical dexterity but also a deep, spatial understanding of the axillary boundaries and the variations in its neurovascular anatomy.
Detailed Surgical Anatomy and Biomechanics
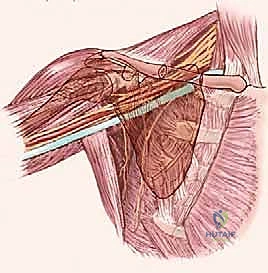
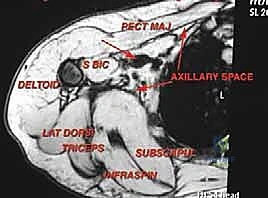
To execute a safe and oncologically sound resection, the surgeon must possess an eidetic understanding of the axillary architecture. Conceptualize the axillary space as a truncated pyramid, with its apex pointing superiorly toward the cervicoaxillary canal and its base opening inferiorly toward the arm. This three-dimensional space is bounded by complex myofascial layers that dictate tumor containment and guide our surgical approach.
Muscular Boundaries and Aponeurotic Framework
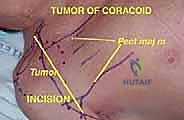
The walls of the axillary pyramid are formed by a complex interplay of muscles and fascial investments, each serving as a potential origin, boundary, or conduit for tumor extension. The anterior wall is dominated by the robust pectoralis major muscle. Its broad tendinous insertion into the lateral lip of the bicipital groove of the humerus is a critical landmark; its release is frequently required for adequate exposure of the deep axillary contents. Deep to the pectoralis major lies the pectoralis minor, enveloped by the clavipectoral fascia, which attaches superiorly to the clavicle and inferiorly blends with the suspensory ligament of the axilla.
The posterior wall is a formidable barrier composed of the subscapularis, teres major, and latissimus dorsi muscles. The latissimus dorsi, with its broad origin and distinct tendinous insertion into the floor of the intertubercular sulcus, serves as a vital inferior landmark. The subscapularis forms the bulk of the posterior wall, and its anterior surface is intimately related to the deep axillary neurovascular structures. The medial boundary is formed by the lateral thoracic wall, specifically the serratus anterior muscle overlying the upper ribs and intercostal spaces. Tumor extension into this medial boundary significantly escalates operative complexity, often necessitating a multidisciplinary approach involving thoracic surgery for en bloc rib resection and chest wall reconstruction.
The lateral wall, or the apex of the pyramid's base, is narrow and defined by the intertubercular sulcus of the humerus, the coracobrachialis, and the short head of the biceps brachii. These muscles, particularly the coracobrachialis, are intimately associated with the musculocutaneous nerve and serve as the lateral gateway to the neurovascular bundle. Understanding these muscular boundaries is critical for planning the surgical approach, as tumors will often displace these structures rather than invade them directly, unless the fascia is breached by prior biopsy or aggressive tumor biology.
Neurovascular Topography: The Brachial Plexus and Axillary Sheath
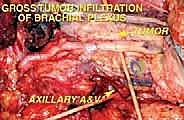
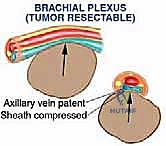
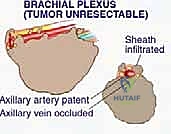
The true challenge, and indeed the beauty, of axillary surgery lies in navigating the major branches of the infraclavicular brachial plexus and the axillary vessels. These structures run in a common, albeit complex, neurovascular sheath, making their precise identification, mobilization, and preservation paramount to a successful limb-sparing outcome. The infraclavicular brachial plexus gives rise to five major terminal nerves at the level of the pectoralis minor, and their relationship to the axillary vascular sheath is relatively constant.

The Lateral Cord derivatives include the musculocutaneous nerve and the lateral root of the median nerve. The musculocutaneous nerve is often the first major neural structure identified during exploration. It courses along the medial aspect of the conjoint tendon, innervating the coracobrachialis and biceps. Finding it in the superficial axillary fat, just inferior to the coracoid process, provides a reliable "Rosetta Stone" for deciphering the rest of the plexus. The Posterior Cord gives rise to the axillary and radial nerves. The axillary nerve dives deep, passing inferior to the glenohumeral joint through the quadrangular space, while the radial nerve travels posterior to the neurovascular sheath, eventually spiraling around the posterior humerus.
The Medial Cord derivatives are particularly vulnerable. The ulnar nerve is positioned along the most medial aspect of the vascular sheath and exits distally. Due to this medial, dependent position, it is the nerve most frequently involved or compressed by tumors arising inferior to the brachial plexus. The axillary artery, a direct continuation of the subclavian artery, is divided into three parts by the pectoralis minor. Surgeons must be prepared to meticulously ligate its major branches—the thoracoacromial, lateral thoracic, subscapular, and circumflex humeral arteries—to mobilize the main trunk away from the tumor mass. The axillary vein, thin-walled and easily compressible, is highly susceptible to tumor infiltration; venous occlusion on preoperative imaging is a grave indicator of neurovascular encasement.
Lymphatic Drainage and Regional Node Basins
The axilla is remarkably rich in lymphatic tissue, serving as the primary drainage basin for the upper extremity, the superficial chest wall, and the breast. Major clusters of lymph nodes are anatomically segregated into groups: the pectoral (anterior) nodes along the lateral thoracic vessels, the subscapular (posterior) nodes along the subscapular vessels, the humeral (lateral) nodes along the axillary vein, and the central and apical nodes deeper within the fat pad.
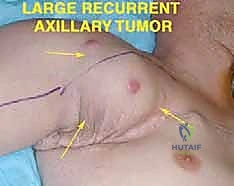
Metastatic disease, particularly from breast cancer, melanoma, or aggressive soft tissue sarcomas (like epithelioid sarcoma or clear cell sarcoma, which have a propensity for lymphatic spread), frequently involves these nodes. When lymphatic metastasis is present, the nodes often coalesce into large, matted masses that obliterate normal tissue planes. In these scenarios, the surgeon must perform a meticulous en bloc regional lymphadenectomy, sweeping the lymphatic contents away from the preserved neurovascular structures while maintaining oncologic integrity.
Exhaustive Indications and Contraindications
The decision to proceed with a complex axillary space resection requires a rigorous, multidisciplinary evaluation. The primary indication for surgical intervention is the presence of a primary bone or soft tissue sarcoma localized to the axillary region, where an R0 (microscopically negative) margin can be achieved while preserving a functional upper extremity. Additionally, isolated bulky metastatic lymphadenopathy that is symptomatic or progressing despite systemic therapy may warrant palliative or local-control resection.
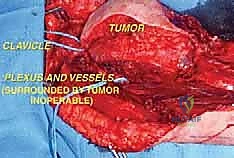
Contraindications are primarily dictated by the extent of neurovascular and chest wall involvement. Absolute contraindications to limb salvage include circumferential encasement of the entire brachial plexus and axillary vessels that precludes vascular reconstruction or results in a completely flail, insensate limb. In such devastating scenarios, a forequarter amputation remains the only oncologically sound option. Massive invasion into the thoracic cavity with pleural or pulmonary involvement may also preclude resection, depending on the patient's physiological reserve and the feasibility of complex chest wall reconstruction.
Relative contraindications include the presence of fungating, ulcerated tumors that heavily contaminate the surgical field, severe patient comorbidities that prohibit prolonged anesthesia or complex reconstructive procedures, and extensive distant metastatic disease where the morbidity of the surgery outweighs the palliative benefit. The surgical team must engage in candid, detailed discussions with the patient regarding the high risks of nerve palsy, vascular compromise, and the potential need for intraoperative conversion to amputation.
Table: Indications and Contraindications for Axillary Limb-Sparing Resection
| Category | Specific Criteria | Clinical Considerations |
|---|---|---|
| Absolute Indications | Primary soft tissue sarcoma (STS) of the axilla | Curative intent; requires R0 margins and functional limb preservation. |
| Absolute Indications | Primary bone sarcoma with axillary extension | e.g., Proximal humerus osteosarcoma; requires en bloc resection of bone and extraosseous mass. |
| Relative Indications | Isolated metastatic axillary lymphadenopathy | Palliative or local control; often seen in melanoma or breast carcinoma. |
| Relative Indications | Benign aggressive tumors | e.g., Desmoid fibromatosis causing severe neurovascular compression or pain. |
| Absolute Contraindications | Complete encasement of the Brachial Plexus | Results in a flail, insensate limb post-resection; forequarter amputation indicated. |
| Absolute Contraindications | Unreconstructable vascular encasement | Inability to bypass or graft the axillary artery/vein safely. |
| Relative Contraindications | Massive chest wall/pleural invasion | Requires complex multidisciplinary thoracic reconstruction; depends on patient reserve. |
| Relative Contraindications | Fungating, infected tumor mass | High risk of sepsis and flap failure; requires aggressive preoperative optimization. |
Pre-Operative Planning, Templating, and Patient Positioning
The success of an axillary resection is inextricably linked to the meticulousness of the preoperative planning phase. This is the stage where the surgical strategy is formulated, anatomical challenges are anticipated, and the precise boundaries of the oncologic resection are defined. A multidisciplinary tumor board review, incorporating insights from medical oncology, radiation oncology, musculoskeletal radiology, and reconstructive microsurgery, is mandatory.
Clinical and Neurological Assessment
Before any advanced imaging is reviewed, a thorough, documented clinical history and physical examination are paramount. The surgeon must meticulously assess the distal neurovascular status of the affected limb. Palpation of the radial and ulnar pulses, coupled with Allen's test, establishes the baseline vascular supply. Inspection for signs of venous congestion, collateral vein engorgement, or swelling in the ipsilateral limb provides clues regarding axillary vein compression or thrombosis.
A comprehensive neurological examination is critical. The surgeon must test the motor strength and sensory dermatomes of the axillary, radial, median, musculocutaneous, and ulnar nerves. It is a vital clinical axiom that loss of nerve function is typically a very late finding. If a patient presents with a profound motor deficit, it is highly indicative of major, likely infiltrative, tumor involvement of the brachial plexus. This finding immediately raises the question of limb-sparing feasibility and must be discussed extensively with the patient. Furthermore, the biopsy history must be critically reviewed. A poorly placed biopsy tract can contaminate critical neurovascular structures or tissue planes, potentially necessitating a much more radical resection, or even a forequarter amputation, to encompass the contaminated tract.
Advanced Imaging Modalities
Three-dimensional, multi-modality imaging is the cornerstone of accurate tumor localization and surgical templating. Plain radiography, while basic, remains an important starting point to assess for bony erosion, periosteal reactions, or matrix mineralization indicative of bone sarcomas extending into the soft tissues.
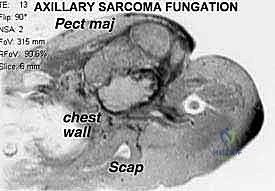
Computed Tomography (CT) with intravenous contrast is invaluable for demonstrating the relationship of the tumor to major vascular structures and outlining the muscular planes. CT is particularly sensitive for evaluating the bony walls of the axilla, specifically the proximal humerus, glenohumeral joint, and scapula. Crucially, the chest wall must be inspected to rule out tumor involvement of the rib cage and pleural cavity. If significant rib involvement is suspected, preoperative planning must include a double-lumen endotracheal tube to allow for selective lung deflation during en bloc chest wall resection.
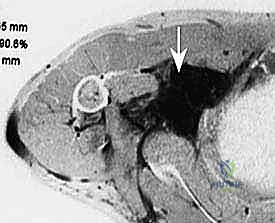
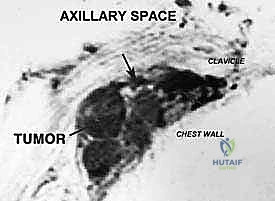
Magnetic Resonance Imaging (MRI) is the gold standard for visualizing the soft tissue contents of the axillary space. Multiplanar MRI precisely defines the anatomic extent of the tumor, its relationship to the fascial compartments, and the presence of peritumoral edema. While the individual cords of the brachial plexus can be difficult to visualize directly when distorted by a large mass, their consistent anatomic relationship to the axillary vessels allows the surgeon to infer their location. MRI is also critical for assessing lymphatic involvement and planning the extent of the soft tissue resection margin.
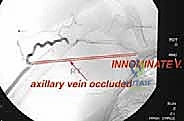
Operative Setup and Patient Positioning
Optimal patient positioning is essential for unhindered access to the axillary space. The patient is typically positioned supine or in a modified beach-chair position, depending on the posterior extent of the tumor. A bump is placed under the ipsilateral scapula to elevate the shoulder girdle and allow the arm to fall posteriorly, opening the anterior axillary approach.
The entire forequarter, chest wall, and upper extremity must be prepped and draped free. Draping the arm free is a critical step; it allows the surgical assistant to manipulate the limb through a full range of motion—abduction, adduction, and rotation—which dynamically alters the tension on the neurovascular structures and dramatically facilitates deep dissection. If a vascular graft (e.g., reversed saphenous vein) is anticipated, the ipsilateral leg must also be prepped and draped. A sterile tourniquet may be placed high on the arm, though its utility is often limited by the proximal extent of the tumor.
Step-by-Step Surgical Approach and Fixation Technique
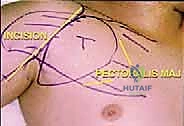
The surgical approach to the axilla requires a methodical, layered dissection, prioritizing the early identification and protection of the neurovascular bundle before any attempt is made to mobilize the tumor mass. The utilitarian deltopectoral approach, extended inferiorly into the axilla, provides the most versatile and comprehensive exposure.
Incision and Superficial Dissection
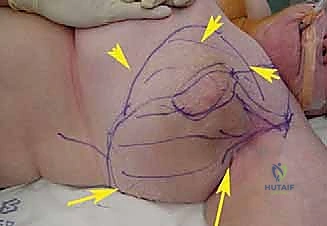
The skin incision begins at the lateral third of the clavicle, courses distally over the coracoid process, and follows the deltopectoral groove. To access the deep axilla, the incision is extended inferiorly along the medial border of the biceps brachii into the axillary fold. If the incision crosses the axillary crease perpendicularly, a Z-plasty should be incorporated during closure to prevent a debilitating scar contracture.
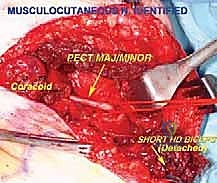
Thick fasciocutaneous flaps are elevated medially and laterally. The cephalic vein is identified within the deltopectoral interval and typically retracted laterally with the deltoid muscle to preserve its venous drainage, although it may be ligated if it impedes exposure or is involved by the tumor. The biopsy tract, which must have been meticulously planned during the preoperative phase, is elliptically excised en bloc with the skin and left attached to the underlying tumor mass to prevent seeding.
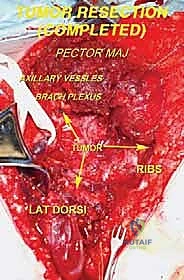
Deep Exposure and Muscular Releases
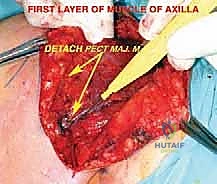
Once the superficial interval is developed, the robust pectoralis major muscle is encountered. For large axillary tumors, adequate exposure is impossible without releasing this muscle. The tendinous insertion of the pectoralis major on the humerus is identified, tagged with heavy non-absorbable sutures for later repair, and sharply divided. The muscle belly is then reflected medially, acting as a retractor and exposing the underlying clavipectoral fascia and pectoralis minor.
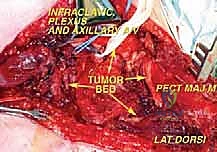
The pectoralis minor, inserting onto the coracoid process, forms the roof over the central portion of the axillary neurovascular bundle. Its insertion is carefully divided and the muscle reflected inferiorly. This critical maneuver unlocks the apex of the axilla, providing direct visualization of the axillary artery, vein, and the surrounding cords of the brachial plexus.
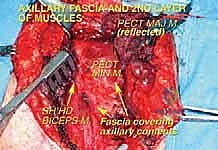
Neurovascular Identification and Mobilization
Dissection within the neurovascular sheath must be performed with meticulous, bloodless technique using loupe magnification and bipolar electrocautery. The golden rule of axillary surgery is to identify normal anatomy in an unviolated area (usually proximally at the apex) and trace it distally toward the tumor.
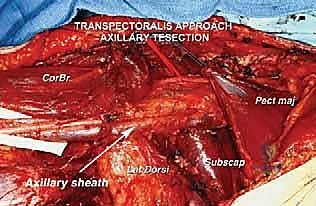
The musculocutaneous nerve is typically the first structure isolated as it enters the coracobrachialis. Tracing this nerve proximally leads the surgeon directly to the lateral cord and the lateral root of the median nerve. Vessel loops are placed around each identified nerve for gentle retraction. The axillary artery is then skeletonized. Its major branches feeding the tumor—often the subscapular or lateral thoracic arteries—are meticulously identified, doubly ligated with silk ties or surgical clips, and divided.
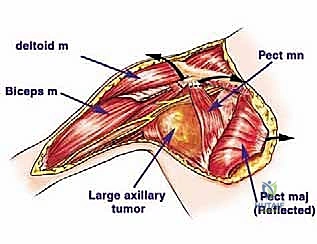
The axillary vein requires extreme caution. It is thin-walled, easily torn, and frequently tethered to the tumor pseudocapsule. Venous tributaries draining the tumor must be securely ligated. If the vein is directly invaded, a segment may need to be resected en bloc with the tumor. While unilateral axillary vein ligation is generally well-tolerated due to robust collateral drainage (e.g., the cephalic vein), primary repair or interposition grafting is preferred if feasible to minimize postoperative lymphedema.