Comprehensive Introduction and Patho-Epidemiology
The medial collateral ligament (MCL) complex is the most frequently injured ligamentous structure of the knee joint, representing a significant portion of both isolated and combined knee injuries encountered in orthopedic practice. Historically, the prevailing dogma dictated that nearly all MCL injuries, regardless of severity, could be managed non-operatively with bracing and early mobilization. While this remains true for the vast majority of isolated Grade I and Grade II sprains due to the robust vascular supply and intrinsic healing capacity of the medial capsuloligamentous envelope, our understanding of complex knee biomechanics has evolved. We now recognize that specific injury patterns—particularly high-grade tibial avulsions, multi-ligamentous knee injuries (MLKIs), and chronic valgus instability—demand meticulous surgical intervention to restore joint kinematics and prevent rapid articular degeneration.
The pathophysiology of MCL injuries typically involves a valgus force applied to a partially flexed knee, frequently compounded by an external rotational moment. This mechanism is ubiquitous in contact sports such as American football, rugby, and alpine skiing. When the applied force exceeds the tensile strength of the ligamentous complex, sequential failure occurs, typically beginning with the superficial MCL (SMCL), progressing to the deep MCL (dMCL), and ultimately compromising the posteromedial corner (PMC) and the posterior oblique ligament (POL). The location of the tear is prognostically significant; femoral-sided avulsions generally possess a superior healing environment compared to mid-substance tears, while distal tibial avulsions may become entrapped superficial to the pes anserinus tendons—a phenomenon clinically analogous to the Stener lesion of the thumb—rendering anatomic non-operative healing impossible.
In the context of multi-ligamentous knee injuries, the presence of an unaddressed Grade III MCL tear significantly compromises the success of concurrent anterior cruciate ligament (ACL) or posterior cruciate ligament (PCL) reconstructions. The persistent valgus and rotatory laxity places deleterious stress on the central pivot grafts, dramatically increasing the risk of premature graft elongation and clinical failure. Consequently, the contemporary orthopedic surgeon must possess a nuanced understanding of when to abandon conservative management in favor of acute repair or chronic reconstruction.
This chapter serves as an intraoperative masterclass, detailing the exhaustive surgical anatomy, precise biomechanical principles, and step-by-step surgical techniques required to master medial knee reconstruction. Whether performing a primary repair with suture anchors or a complex anatomic double-bundle reconstruction using allograft tissue, the principles of anatomic footprint restoration, meticulous soft-tissue handling, and rigid fixation remain paramount to achieving optimal patient outcomes.
Detailed Surgical Anatomy and Biomechanics
A profound, three-dimensional understanding of the medial knee anatomy is the absolute foundation of successful surgical intervention. The medial aspect of the knee is not a single ligamentous band but a highly orchestrated, multi-layered complex. The classic anatomical description by Warren and Marshall elegantly organizes the medial knee into three distinct layers, a framework that remains indispensable for the operating surgeon navigating this region.
Layer I: Superficial Fascia and Retinaculum
Layer I represents the most superficial fascial investment of the medial knee, primarily comprising the crural fascia. Proximally, this layer is continuous with the deep fascia of the thigh and the medial patellar retinaculum. Distally, it firmly invests the sartorius muscle and blends with the periosteum of the proximal medial tibia. The sartorius itself serves as a critical surgical landmark; its anterior border dictates the fascial incision required to access the deeper ligamentous structures. During surgical dissection, elevating Layer I from the underlying structures requires meticulous care to avoid inadvertent damage to the superficial medial collateral ligament, which lies immediately deep to it.
Layer II: Superficial Medial Collateral Ligament and MPFL
Layer II houses the primary static stabilizers of the medial knee: the Superficial Medial Collateral Ligament (SMCL) and the Medial Patellofemoral Ligament (MPFL). The SMCL is the undisputed workhorse of medial stability. Quantitative anatomical studies by LaPrade et al. have precisely defined its footprints. The femoral origin is located approximately 3.2 mm proximal and 4.8 mm posterior to the medial epicondyle. From this origin, the SMCL courses distally to form two distinct tibial attachments: a proximal attachment primarily to soft tissue and a broad, robust distal fascial attachment located approximately 6 cm distal to the joint line, situated deep to the pes anserinus tendons.
The SMCL is functionally divided into anterior and posterior bands. The anterior fibers are taut in knee flexion, while the posterior fibers become taut in extension. This reciprocal tensioning mechanism ensures valgus stability throughout the entire arc of motion. Biomechanically, the SMCL provides approximately 78% of the restraining force against valgus stress at 25 degrees of knee flexion. The MPFL, also located in Layer II, originates in the saddle region between the medial epicondyle and the adductor tubercle, fanning out to insert on the superomedial border of the patella. It acts as the primary restraint to lateral patellar translation.
Layer III: Deep Medial Collateral Ligament and Posteromedial Corner
Layer III is the deepest strata, consisting of the true joint capsule and its specialized thickenings. The deep MCL (dMCL) is fundamentally a thickening of the medial joint capsule, divided into meniscofemoral and meniscotibial (coronary) ligaments. These structures firmly anchor the medial meniscus to the femur and tibia, respectively. The meniscotibial ligament is shorter and thicker, making it less prone to injury than its meniscofemoral counterpart. The dMCL acts as a secondary restraint to valgus stress but plays a critical role in stabilizing the medial meniscus and preventing its extrusion.
Moving posteriorly within Layer III, we encounter the Posteromedial Corner (PMC), a complex network providing vital rotational stability. The defining structure of the PMC is the Posterior Oblique Ligament (POL). The POL is a fan-shaped fibrous expansion originating near the adductor tubercle and branching into three distinct fascial arms: superficial, tibial, and capsular. The POL is the primary restraint to internal rotation of the tibia when the knee is near full extension. Furthermore, the semimembranosus tendon intimately interacts with the PMC, sending five distinct expansions that dynamically tension the posteromedial capsule and the POL during knee flexion, actively resisting anteromedial rotatory instability (AMRI).
Neurovascular Considerations
Navigating the medial knee requires an acute awareness of the regional neurovascular anatomy. The saphenous nerve, the largest cutaneous branch of the femoral nerve, exits the adductor canal and pierces the deep fascia between the sartorius and gracilis tendons. It then courses inferiorly along the medial aspect of the tibia. Its infrapatellar branch, which provides sensation to the anteromedial knee, is highly variable in its course and is exquisitely vulnerable during the initial surgical incision and superficial dissection.
The great saphenous vein ascends superficially along the medial aspect of the leg and thigh, generally running posterior to the primary surgical field but remaining at risk during extensive posterior retraction. Iatrogenic injury to the saphenous nerve or its branches can result in debilitating neuromas, chronic regional pain syndrome (CRPS), or distressing areas of anesthesia. Surgeons must employ meticulous blunt dissection techniques within the subcutaneous tissues and maintain a hyper-vigilant awareness of these structures when elevating fascial flaps.
Exhaustive Indications and Contraindications
The decision-making process regarding the surgical management of MCL injuries requires a sophisticated synthesis of clinical examination findings, imaging data, and patient-specific functional demands. While the historical pendulum swung heavily toward non-operative management, contemporary literature supports a more aggressive surgical approach for specific, high-risk injury patterns to prevent chronic instability and articular degradation.
Surgical intervention is unequivocally indicated in the setting of a multi-ligamentous knee injury (e.g., combined ACL/MCL or PCL/MCL tears) where the MCL exhibits Grade III valgus laxity. In these scenarios, failing to restore medial competence places unacceptable stress on cruciate ligament reconstructions, predisposing them to early failure. Another absolute indication for acute surgical repair is a distal tibial avulsion of the SMCL where the ligament end is flipped superficial to the pes anserinus (the "Stener-like" lesion of the knee). This anatomical interposition prevents the ligament from healing to its footprint, guaranteeing chronic instability if left unaddressed.
Chronic medial instability, characterized by persistent valgus laxity and medial-sided pain despite a comprehensive minimum 3-to-6-month trial of targeted physical therapy, is a primary indication for MCL reconstruction. Furthermore, patients demonstrating significant anteromedial rotatory instability (AMRI), indicative of combined SMCL and POL/posteromedial corner incompetence, rarely achieve satisfactory outcomes with conservative care and require surgical reconstruction to restore rotational kinematics.
Conversely, isolated Grade I and Grade II MCL sprains are strict contraindications for acute surgical intervention. These injuries possess an excellent prognosis with functional bracing and structured rehabilitation. Even isolated Grade III injuries, particularly those occurring at the femoral origin or mid-substance, often heal remarkably well non-operatively, provided the patient is compliant with a hinged knee brace locked in slight varus. Absolute contraindications to surgery include active local or systemic infection, severe peripheral vascular disease compromising wound healing, and a medically unstable patient unable to tolerate anesthesia.
| Category | Indications for Surgery | Contraindications for Surgery |
|---|---|---|
| Acute Injuries | - Grade III tear in a multi-ligament knee injury (MLKI) - Tibial avulsion entrapped above pes anserinus - Intra-articular ligament entrapment - Open medial knee injuries |
- Isolated Grade I or II sprains - Isolated Grade III femoral/mid-substance tears (relative) - Active local soft tissue infection |
| Chronic Injuries | - Symptomatic chronic valgus instability - Persistent Anteromedial Rotatory Instability (AMRI) - Failure of >3-6 months of conservative rehab |
- Asymptomatic laxity - Severe, uncorrected mechanical varus/valgus malalignment (requires osteotomy first) - Medically unfit for surgery |
| Patient Factors | - High-demand athletes requiring absolute stability - Concomitant meniscal repairs requiring stable capsular bed |
- Poor compliance with complex postoperative rehabilitation - Severe advanced tricompartmental osteoarthritis |
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the crucible in which surgical success is forged. The process begins with a rigorous clinical examination, ideally performed both in the clinic and subsequently under anesthesia. The cornerstone of the physical exam is the valgus stress test, performed at both 0 degrees and 30 degrees of knee flexion. Asymmetric medial opening at 30 degrees isolates the SMCL, while laxity at 0 degrees indicates a massive injury involving the SMCL, dMCL, POL, and likely the cruciate ligaments. The Slocum test and the anterior drawer test performed in external rotation are critical for identifying anteromedial rotatory instability (AMRI), which dictates the need to address the posteromedial corner during surgery.
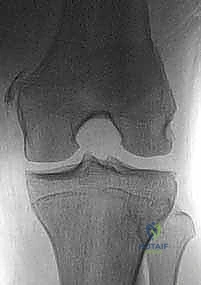
Imaging plays a confirmatory and strategic role. Standard weight-bearing radiographs are essential to evaluate for underlying osteoarthritis, avulsion fractures, or the presence of a Pellegrini-Stieda lesion (calcification at the femoral MCL origin), which signifies chronic, repetitive medial-sided injury. Magnetic Resonance Imaging (MRI) is the gold standard for defining the precise pathoanatomy. The surgeon must systematically interrogate the MRI to determine the exact location of the tear (femoral, mid-substance, or tibial), the integrity of the POL and deep capsular structures, and the presence of concomitant meniscal or chondral pathology.
Examination Under Anesthesia and Patient Positioning
Prior to the first incision, an Examination Under Anesthesia (EUA) is mandatory. Muscular guarding is eliminated, allowing for a pure assessment of ligamentous competence. The surgeon must meticulously document the degree of valgus gapping, the quality of the endpoint, and the presence of any concurrent pivot shift or posterior sag.
Patient positioning must facilitate unhindered access to the medial compartment and allow for dynamic manipulation of the knee through a full range of motion. The patient is positioned supine on the operating table. A standard approach utilizes a well-padded high-thigh tourniquet. The operative leg is typically placed in a specialized arthroscopic leg holder positioned at the level of the proximal thigh. This setup allows the surgeon to stand or sit comfortably, utilizing their own torso to apply varus or valgus stress while freeing both hands for surgical instrumentation.
Alternatively, the procedure can be performed on a flat radiolucent table with a lateral thigh post and a foot bump to maintain the knee at 70 to 90 degrees of flexion. This flat-table setup is particularly advantageous when concurrent posterior or lateral procedures are anticipated, or when extensive fluoroscopy is required for precise tunnel localization during anatomic reconstructions. Regardless of the setup, the contralateral leg must be carefully padded and secured to prevent pressure-induced neuropathies.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution of an MCL repair or reconstruction demands an uncompromising adherence to anatomic principles and delicate soft-tissue handling. The procedure generally commences with a diagnostic arthroscopy to address intra-articular pathology before transitioning to the open medial approach.
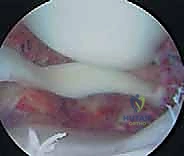
Diagnostic Arthroscopy and Concurrent Pathology Management
Standard anterolateral and anteromedial arthroscopic portals are established. The joint is systematically swept to identify and treat meniscal tears, chondral defects, and cruciate ligament ruptures. In the setting of a high-grade MCL tear, the surgeon will frequently observe a positive "drive-through" sign. This occurs when the medial compartment opens abnormally wide (typically >10 mm) under valgus stress, allowing the arthroscope to pass effortlessly between the medial femoral condyle and the medial tibial plateau into the posteromedial recess.
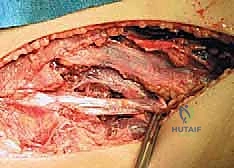
If a concurrent ACL or PCL reconstruction is planned, it is generally advisable to drill the tunnels and pass the central pivot grafts before opening the medial side. However, the final tensioning and fixation of the cruciate grafts should be deferred until the medial side is exposed and ready for fixation, as restoring the medial buttress often re-centers the tibia and alters the optimal tension on the cruciate grafts.
The Mid-Medial Approach and Dissection
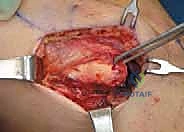
Following arthroscopy, attention is directed to the medial aspect of the knee. A longitudinal, slightly curvilinear incision is made, typically measuring 6 to 8 centimeters, centered over the medial joint line. The incision is placed midway between the medial border of the patella and the posteromedial margin of the tibia.
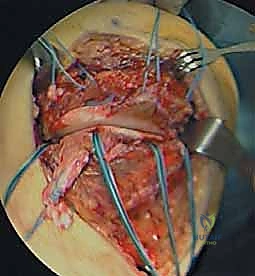
Sharp dissection is carried through the skin, followed by careful blunt dissection through the subcutaneous adipose tissue. The surgeon must remain acutely aware of the saphenous nerve and its infrapatellar branch, sweeping them anteriorly or posteriorly as dictated by the patient's specific anatomy.
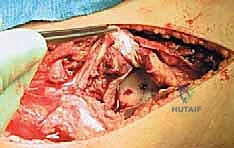
Once the crural fascia (Layer I) is identified, an incision is made along the anterior border of the sartorius muscle. The sartorius is retracted posteriorly, exposing the underlying Superficial MCL (Layer II). The interval between the posterior border of the SMCL and the anterior border of the POL is then identified and developed. This crucial plane allows the surgeon to separate Layer II from Layer III, providing access to the deep MCL, the medial meniscus, and the tibial plateau for anchor placement.
Acute Repair Techniques
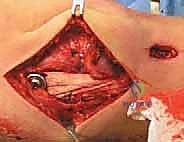
For acute, avulsion-type injuries (typically occurring at the femoral origin or the distal tibial insertion), direct primary repair is the treatment of choice. If the tear involves the deep MCL and meniscotibial ligaments, suture anchors are placed along the articular margin of the proximal tibia.
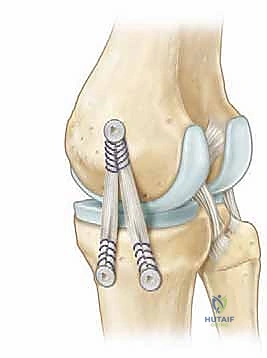
The torn capsular structures are meticulously imbricated and tied down to the bone, restoring the meniscal tether. For superficial MCL avulsions, the anatomic footprint is debrided to bleeding cortical bone. Solid-body or all-suture anchors (typically double-loaded) are placed into the anatomic footprint.
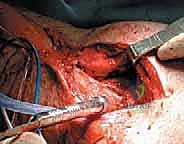
The sutures are passed through the ligamentous stump using a locking Krackow or Mason-Allen configuration. Crucially, the final tensioning and tying of the sutures must be performed with the knee held in 20 to 30 degrees of flexion while a gentle varus force is applied to close the medial joint space. Tying the sutures in full extension risks capturing the ligament in a non-anatomic, overly tight configuration that will severely restrict postoperative flexion.
Chronic Reconstruction Techniques
In cases of chronic instability, mid-substance destruction, or failed primary repairs, a formal reconstruction utilizing autograft (semitendinosus) or allograft tissue is required. Contemporary techniques favor an anatomic reconstruction that addresses both the SMCL and the POL to restore both valgus and rotational stability.

The femoral footprints of the SMCL and POL are identified using fluoroscopic and anatomic landmarks (the medial epicondyle, adductor tubercle, and gastrocnemius tubercle). Guide pins are placed, and sockets are reamed to accommodate the graft. The graft is secured in the femur using an interference screw or a suspensory cortical button.
The graft is then routed distally, deep to the sartorial fascia. The tibial attachments are identified. For the SMCL, a tunnel or anchor is placed approximately 6 cm distal to the joint line. For the POL, fixation is targeted at the posteromedial tibia, near the direct arm of the semimembranosus. The SMCL limb of the graft is tensioned and fixed with the knee at 20-30 degrees of flexion under varus stress. The POL limb is subsequently tensioned and fixed with the knee in full extension to restore its function as an internal rotation restraint.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, complications following medial knee surgery can and do occur. The surgeon must be prepared to identify and manage these issues promptly to optimize clinical outcomes. By far, the most frequent and challenging complication following MCL repair or reconstruction is postoperative stiffness, specifically arthrofibrosis.
The medial capsule is highly vascular and prone to robust, hypertrophic scar formation. If the knee is immobilized for prolonged periods, or if the graft is tensioned excessively in extension, the patient will struggle to regain full range of motion. The incidence of clinically significant stiffness can approach 10-15% in multi-ligamentous reconstructions. Management begins with aggressive, early physical therapy. If conservative measures fail, arthroscopic lysis of adhesions and manipulation under anesthesia (MUA) may be required, typically performed between 8 and 12 weeks postoperatively.
Iatrogenic neurologic injury is another significant concern. The saphenous nerve and its infrapatellar branch are highly vulnerable during the superficial dissection. Injury rates vary in the literature but can occur in up to 5-10% of open medial approaches. While many neuropraxias resolve spontaneously, complete transection or neuroma formation can lead to chronic, debilitating pain. Prevention through meticulous surgical technique is paramount. If a painful neuroma develops and is refractory to conservative management (gabapentinoids, local injections), surgical excision and burying of the nerve stump deep into muscle may be necessary as a salvage procedure.
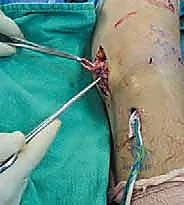
Recurrent instability is a devastating complication, often resulting from non-anatomic tunnel placement, inadequate graft tensioning, failure to address concurrent cruciate pathology, or premature return to aggressive activities. Revision reconstruction is technically demanding, often requiring the use of allograft tissue, hardware removal, and potential staged bone grafting of expanded tunnels.
| Complication | Estimated Incidence | Etiology / Risk Factors | Prevention & Salvage Management |
|---|---|---|---|
| Arthrofibrosis / Stiffness | 10 - 15% | Prolonged immobilization, over-tensioning graft, hypertrophic scarring. | Prevention: Early ROM, anatomic tensioning (SMCL at 30° flexion). Salvage: Aggressive PT, Arthroscopic lysis of adhesions, MUA. |
| Saphenous Nerve Injury | 5 - 10% | Iatrogenic transection or traction during Layer I/II dissection. | Prevention: Blunt superficial dissection, awareness of anatomic variability. Salvage: Neuromodulators, diagnostic blocks, surgical neuroma excision. |
| Recurrent Valgus Instability | 3 - 8% | Non-anatomic tunnel placement, missed MLKI, premature return to sport. | Prevention: Fluoroscopic/anatomic footprint verification, address ACL/PCL. Salvage: Revision anatomic double-bundle reconstruction. |
| Hardware Irritation | 5 - 12% | Prominent femoral interference screws or suture knots under thin medial skin. | Prevention: Countersink screws, use low-profile anchors/buttons. Salvage: Outpatient hardware removal after graft incorporation (>9-12 months). |
Phased Post-Operative Rehabilitation Protocols
The surgical procedure represents only the first half of the therapeutic equation; a rigorously structured, phased rehabilitation protocol is equally critical to achieving a successful outcome. The rehabilitation philosophy must balance the competing demands of protecting the healing soft tissues from deleterious valgus stress while simultaneously preventing the debilitating onset of arthrofibrosis.
Phase I: Protection and Early Mobilization (Weeks 0-4)
Immediately postoperatively, the patient is placed in a hinged knee brace. For isolated MCL repairs or reconstructions, the brace is typically locked in full extension for weight-bearing ambulation to prevent dynamic valgus loading. However, early range of motion (ROM) is instituted immediately to combat stiffness. Patients are encouraged to perform passive and active-assisted ROM exercises from 0 to 90 degrees in a controlled, non-weight-bearing environment. Quadriceps activation is prioritized early through isometric sets and straight leg raises. Cryotherapy and elevation are utilized aggressively to manage edema.
Phase II: Restoring Kinematics and Early Strengthening (Weeks 4-8)
As the graft begins to incorporate and the acute inflammatory phase subsides, the focus shifts to restoring full, symmetric range of motion. The hinged brace is gradually unlocked to allow functional ambulation, provided the patient demonstrates adequate quadriceps control without a lag. Closed kinetic chain exercises, such as mini-squats, leg presses, and stationary cycling, are introduced. These exercises provide axial loading without subjecting the medial structures to harmful varus/valgus shear forces. Proprioceptive training, utilizing balance boards and unstable surfaces, is initiated to retrain the neuromuscular control of the joint.
Phase III: Advanced Strengthening and Dynamic Stability (Weeks 8-16)
During this phase, the patient is typically weaned from the functional brace for activities of daily living. The strengthening regimen becomes more aggressive, incorporating progressive resistance training for the quadriceps, hamstrings, and gluteal musculature. The hamstrings, particularly the semimembranosus and pes anserinus group, act as dynamic medial stabilizers; their robust strength is essential to protect the reconstructed ligaments. Linear running and low-impact plyometrics may be introduced late in this phase, contingent upon the patient demonstrating excellent strength and no effusion.
Phase IV: Return to Sport and High-Level Function (Months 4-6+)
The final phase is highly individualized, tailored to the specific demands of the patient's athletic or occupational pursuits. Advanced plyometrics, cutting, pivoting, and sport-specific agility drills are introduced in a controlled manner. Return to unrestricted sport is not dictated strictly by a timeline, but rather by the achievement of objective functional criteria. These criteria typically include a full, painless range of motion, no clinical effusion, a stable clinical examination (negative valgus stress), and limb symmetry indices (LSI) of greater than 90% on functional hop testing and isokinetic strength evaluations.
Summary of Landmark Literature and Clinical Guidelines
The evolution of medial knee surgery is deeply rooted in a foundation of meticulous anatomical and biomechanical research. The modern orthopedic surgeon stands on the shoulders of giants in this field, and a working knowledge of the landmark literature is essential for evidence-based practice.
The seminal 1979 paper by Warren and Marshall established the three-layer anatomical model of the medial knee, a paradigm that remains the absolute standard for surgical orientation and dissection to this day. Following this, Hughston's foundational work highlighted the critical importance of the posteromedial corner and the posterior oblique ligament in controlling
Detailed Chapters & Topics
Dive deeper into specialized chapters regarding repair-of-acute-and-chronic-knee-medial-collateral-ligament-injuries
