Meniscal Allograft Transplantation: A Masterclass in Restorative Knee Surgery
Key Takeaway
This masterclass guides fellows through meniscal allograft transplantation, covering essential anatomy, meticulous preoperative planning, and precise intraoperative execution. We detail graft preparation, tibial recipient site creation, and advanced fixation techniques, emphasizing critical pearls and pitfalls. Learn to manage potential complications and optimize postoperative rehabilitation, restoring function in symptomatic meniscal-deficient knees.
Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues, to the operating theater and to this definitive masterclass. Today, we are tackling a procedure that, while technically demanding and fraught with steep learning curves, offers profound relief and long-term joint preservation for a highly select cohort of patients: Meniscal Allograft Transplantation (MAT). This intervention is not merely a structural repair; it is a fundamental biomechanical restoration. Our primary objective is to mitigate the devastating, progressive effects of meniscal deficiency, thereby altering the natural history of the post-meniscectomized knee and delaying the onset of end-stage osteoarthritis.
To fully appreciate the gravity of this procedure, we must first ground ourselves in the fundamental patho-epidemiology that brings these patients to our surgical tables. Annually, approximately 850,000 meniscal procedures are performed in the United States alone. This staggering volume underscores the sheer prevalence of meniscal injuries and, consequently, the massive population at risk for post-meniscectomy syndrome. Meniscal pathology generally bifurcates into two broad, clinically distinct categories: acute traumatic tears and degenerative lesions, each presenting unique challenges regarding preservation and subsequent deficiency.
Acute traumatic tears typically manifest in younger, highly active patients, often under the age of 35, who possess previously robust, healthy meniscal tissue. These injuries frequently occur in conjunction with an acute Anterior Cruciate Ligament (ACL) rupture or other multiligamentous trauma. While our primary, unwavering goal in orthopedic sports medicine is always meniscus preservation via meticulous repair techniques, large, complex, or chronically retracted irreparable tears often unfortunately necessitate partial or subtotal meniscal excision. Consequently, many of these young patients become profoundly symptomatic in the meniscal-deficient compartment. This symptomatology is driven by a radical increase in articular cartilage contact stresses, leading to progressive, accelerated cartilage deterioration. Traumatic tears often present as unstable longitudinal tears within the vascular zone, making them ideal candidates for primary repair; however, for those with severe, irreparable loss, allograft transplantation becomes a vital, joint-saving option.
Conversely, degenerative tears represent a more complex, insidious pathophysiological process, typically observed in patients over the age of 35. In these scenarios, a relatively minor, seemingly innocuous event often acts as the catalyst, propagating a complex tear pattern through already desiccated, degenerative meniscal fibrocartilage. Unlike traumatic tears in young tissue, these degenerative lesions are generally not amenable to primary repair due to poor tissue quality and diminished healing capacity. This underscores the critical need for alternative, restorative solutions like transplantation when these patients become acutely symptomatic following inevitable resection. The natural history of meniscectomy is stark: resecting the meniscus can decrease the contact area by up to 75% and increase joint contact stresses by over 200%. This biomechanical catastrophe invariably leads to premature cartilage deterioration, subchondral bone overload, and the ultimate development of unicompartmental osteoarthritis.
Detailed Surgical Anatomy and Biomechanics
Before we make any incision or establish our arthroscopic portals, a profound, three-dimensional understanding of meniscal anatomy and its biomechanical interplay is absolutely paramount. The menisci are semilunar, wedge-shaped fibrocartilaginous discs, composed predominantly of highly organized Type I collagen arranged in a circumferential pattern. This specific architectural arrangement is critical, as it allows the meniscus to translate axial compressive loads into circumferential "hoop stresses," thereby protecting the underlying articular cartilage. Water, accounting for roughly 70% of their composition, is trapped within a dense extracellular matrix by negatively charged glycosaminoglycans, providing the menisci with their unique viscoelastic and shock-absorbing properties.
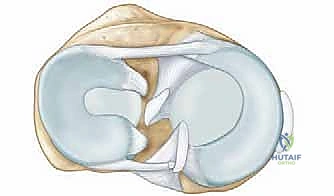
Crucially, the vascularity of the meniscus is highly limited and topographically specific. Only the peripheral 10% to 30% of the meniscus is vascularized, receiving its blood supply via the perimeniscal capillary plexus, which is fed by contributions from the superior and inferior medial and lateral geniculate arteries. The most robust vascular supply is located adjacent to the popliteal hiatus laterally and the posteromedial capsule medially. This limited, peripheral vascularity dictates our repair strategies, explains why central tears inevitably fail to heal, and underscores the necessity of achieving secure peripheral fixation during allograft transplantation to encourage host tissue integration and revascularization of the allograft rim.
When evaluating the specific meniscal characteristics, we must differentiate between the medial and lateral compartments. The medial meniscus is C-shaped and covers a smaller percentage of the medial tibial plateau compared to its lateral counterpart. Medial meniscal injuries are frequently associated with chronic ACL insufficiency, acting as a critical secondary stabilizer to anterior tibial translation. A key anatomical detail for surgical planning is that a portion of the ACL's tibial insertion footprint lies directly between the anterior and posterior horn attachment sites of the medial meniscus. This distinct interposition dictates that medial meniscal transplants are typically performed using a two-bone-plug technique rather than a single bone bridge, to avoid iatrogenic injury to the native ACL footprint.
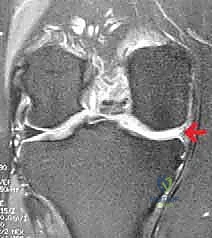
Conversely, the lateral meniscus is more circular, covers a relatively larger percentage of the articular surface in its compartment, and is highly mobile. Its anterior horn attaches adjacent to the ACL, and its posterior horn attachment is located posterior to the intercondylar eminence. Critically for our surgical approach, the anterior and posterior horn attachments of the lateral meniscus are anatomically proximate to each other, without a major ligamentous insertion footprint interposed between them. This unique anatomical arrangement makes the lateral meniscus highly amenable to a single bone bridge (slot) transplantation technique, which provides superior biomechanical fixation and is the gold standard for lateral compartment restoration.
Neurovascular Risks and Muscular Intervals
When we perform our open approaches for graft insertion and peripheral fixation, we must remain acutely vigilant of the surrounding neurovascular structures. The lateral approach, utilized for lateral meniscal transplantation, places the common peroneal nerve at significant risk. This nerve courses posterolaterally, wrapping around the fibular neck; therefore, our posterolateral exposure for inside-out suturing must be meticulous. We typically navigate the interval between the biceps femoris and the iliotibial band, carefully identifying and retracting the common peroneal nerve to prevent iatrogenic palsy. Furthermore, while the popliteal artery and vein lie deep in the popliteal fossa, excessive posterior dissection or errant drill penetration during tunnel creation can result in catastrophic vascular injury.
For the medial approach, the saphenous nerve and its infrapatellar branch, along with the greater saphenous vein, are the primary structures at risk. These structures course medially, superficial to the sartorius fascia. Our medial parapatellar arthrotomy and the posteromedial repair approach require careful superficial dissection, identification, and gentle retraction. We typically employ a vertical incision posterior to the medial collateral ligament (MCL), carefully dissecting the interval between the medial head of the gastrocnemius and the posterior capsule. Retracting the gastrocnemius posteriorly acts as a physical shield for the deep neurovascular bundle in the popliteal fossa, ensuring safe passage for our inside-out suture needles.
Exhaustive Indications and Contraindications
Patient selection is arguably the most critical determinant of success in Meniscal Allograft Transplantation. This is not a prophylactic procedure for asymptomatic individuals, nor is it a salvage operation for end-stage arthritis. The ideal candidate is typically a younger patient, generally under the age of 40—though an upper limit of 50 is acceptable for highly active, physiologically young patients who are not yet appropriate candidates for unicompartmental or total knee arthroplasty. These patients must present with an absent or functionally deficient meniscus and complain of localized, symptomatic pain directly attributable to this deficiency, such as activity-related joint line ache or pain associated with barometric pressure changes.
A comprehensive clinical history and physical examination form the blueprint for our preoperative planning. We must meticulously assess prior operative reports and arthroscopic images to determine the exact degree of previous meniscal resection and the documented condition of the articular cartilage. On physical examination, we focus on localizing joint line tenderness, assessing ligamentous stability (particularly the ACL, as stability is a prerequisite for a successful transplant), and evaluating overall mechanical alignment. It is imperative to differentiate meniscal insufficiency pain—typically a dull, deep ache—from the sharp, catching pain that might indicate a recurrent meniscal tear, a loose body, or an unstable chondral flap.
Crucially, the status of the articular cartilage must be rigorously evaluated. MAT is indicated in the setting of Outerbridge Grade I or II chondral changes. While Grade III changes are acceptable, the outcomes are less predictable. Widespread Grade IV articular cartilage loss (bone-on-bone arthritis) is an absolute contraindication to isolated MAT, as the allograft will rapidly fail in a severely arthritic milieu. However, if a focal Grade IV osteochondral lesion exists, MAT can be performed concurrently with a cartilage restoration procedure, such as Osteochondral Allograft Transplantation (OCA) or Matrix-Induced Autologous Chondrocyte Implantation (MACI), provided the overall joint space is preserved.
Furthermore, mechanical alignment and ligamentous stability are non-negotiable prerequisites. A meniscal transplant placed into a malaligned, overloaded compartment (e.g., a medial transplant in a varus knee) or an unstable joint (e.g., an ACL-deficient knee) is doomed to mechanical failure. Therefore, any concurrent varus or valgus malalignment must be corrected via a high tibial osteotomy (HTO) or distal femoral osteotomy (DFO), either in a staged fashion or concurrently. Similarly, ACL or other ligamentous deficiencies must be reconstructed concomitantly to protect the meniscal allograft from excessive shear forces.
| Parameter | Indications for Meniscal Allograft Transplantation | Absolute Contraindications | Relative Contraindications |
|---|---|---|---|
| Age | Typically < 40 years (up to 50 in highly active patients) | Advanced physiological age | Age > 50 with moderate activity |
| Symptoms | Unicompartmental pain related to activity/weight-bearing | Asymptomatic meniscectomy | Diffuse, unlocalized knee pain |
| Meniscal Status | Prior subtotal or total meniscectomy; functionally absent | Repairable meniscal tear | Partial meniscectomy with adequate rim |
| Cartilage Status | Outerbridge Grade I, II, or focal III | Diffuse Outerbridge Grade IV (bone-on-bone) | Diffuse Grade III changes |
| Alignment | Normal mechanical axis | Uncorrected malalignment | Mild, asymptomatic malalignment |
| Stability | Ligamentously stable knee | Uncorrected ligamentous instability | Chronic, low-grade laxity |
| Systemic Factors | Healthy, compliant patient | Inflammatory arthritis, active infection, obesity (BMI > 35) | Smoking, poor surgical compliance |
Pre-Operative Planning, Templating, and Patient Positioning
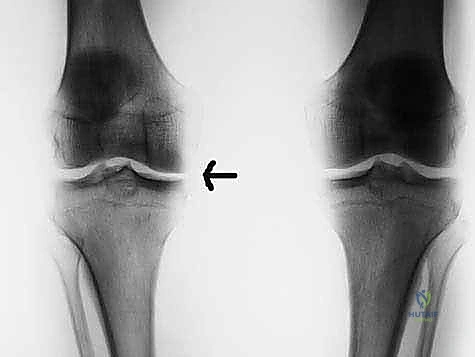
Comprehensive imaging is the cornerstone of accurate diagnosis, meticulous preoperative planning, and precise graft sizing. Plain radiography remains our first-line modality. We require weight-bearing Anteroposterior (AP) views in full extension, but more importantly, weight-bearing 45-degree flexion Posteroanterior (PA) views (Rosenberg views). These flexion views are hyper-critical for detecting subtle joint space narrowing in the posterior aspect of the condyles, which is frequently the first radiographic sign of post-meniscectomy arthrosis. We also obtain Merchant views to assess the patellofemoral joint and long-leg alignment films to quantify any mechanical axis deviation that may necessitate a concurrent osteotomy.

High-resolution Magnetic Resonance Imaging (MRI) is absolutely indispensable. It provides invaluable, non-invasive detail regarding the extent of the meniscal deficiency, allowing us to quantify the remaining peripheral rim. More importantly, MRI allows for a rigorous assessment of the articular cartilage and subchondral bone. We look for subchondral edema (bone marrow lesions), which often correlates with the patient's pain and indicates mechanical overload. MRI also aids in evaluating the integrity of the cruciate and collateral ligaments, ensuring no missed concurrent pathologies exist that could compromise our transplant.
Graft sizing is a meticulous process where precision is paramount. While the exact biomechanical tolerance for size mismatch is debated, an oversized graft can lead to extrusion and altered kinematics, while an undersized graft will fail to adequately reduce contact stresses, leading to premature failure. We utilize the method described by Pollard et al., utilizing AP and lateral radiographs. For coronal sizing (width), we measure the distance from the peak of the tibial eminence to the periphery of the tibial plateau. For sagittal sizing (length), we measure the AP dimension of the tibial plateau on the lateral radiograph. These measurements, corrected for radiographic magnification, are provided to the tissue bank to secure an appropriately size-matched, fresh-frozen, non-irradiated allograft.

Patient positioning in the operating room must facilitate seamless transition between arthroscopic visualization and open extra-articular dissection. The patient is placed supine on the operating table. A high thigh tourniquet is applied but typically left uninflated unless visualization becomes severely compromised. The operative leg is placed in a standard leg holder, allowing for full, unobstructed range of motion from 0 to 120 degrees of flexion, and enabling the application of valgus or varus stress to open the respective compartments. The contralateral leg is placed in a well-padded gynecological stirrup to prevent neurovascular compression. A thorough examination under anesthesia (EUA) is performed to confirm preoperative stability assessments before prepping and draping in a standard sterile fashion.
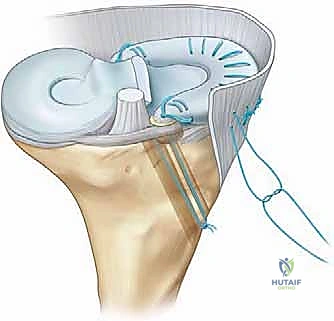
Step-by-Step Surgical Approach and Fixation Technique
Arthroscopic Preparation of the Host Bed
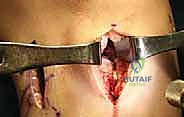
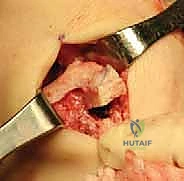
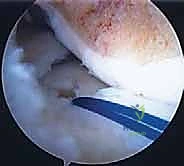
The procedure commences with a comprehensive diagnostic arthroscopy utilizing standard anterolateral and anteromedial portals. We systematically evaluate all compartments, meticulously documenting the status of the articular cartilage to confirm it meets our preoperative criteria (Outerbridge Grade III or less). Any unstable chondral flaps are carefully debrided to stable, vertical margins. We then turn our attention to the meniscal-deficient compartment. The remnant meniscal tissue is carefully excised using a combination of arthroscopic biters and motorized shavers.

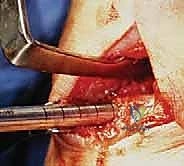
Crucially, we must preserve a 1 to 2-millimeter peripheral rim of the native meniscus. This remnant rim acts as a vascularized host bed, essential for suturing the allograft and facilitating subsequent biological integration and revascularization. The peripheral capsule and the preserved meniscal rim are then aggressively rasped and abraded using a meniscal rasp or a motorized burr without suction. This intentional induction of localized trauma creates a bleeding bed, stimulating a robust healing response and optimizing the biological milieu for allograft incorporation.
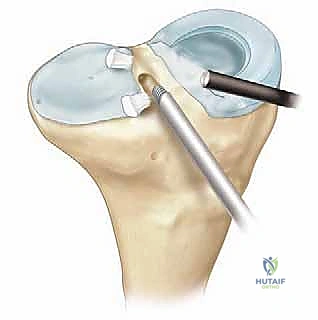
For lateral meniscal transplants, we must clearly define the anterior and posterior horn attachment sites. The anterior horn footprint is located adjacent to the ACL insertion, while the posterior horn footprint lies posterior to the intercondylar eminence. We use an arthroscopic electrocautery device to precisely mark the center of these footprints, as this will dictate the trajectory of our osseous slot. For medial transplants, the footprints are similarly identified and marked, noting the interposition of the ACL footprint between them, which necessitates the two-bone-plug technique.
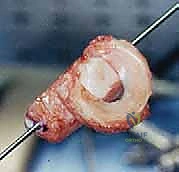
Allograft Preparation on the Back Table
While the host bed is being prepared arthroscopically, the surgical assistant or a second surgeon meticulously prepares the fresh-frozen meniscal allograft on the back table. The allograft is thawed in warm saline. For a lateral meniscal transplant utilizing the bone bridge (slot) technique, the attached tibial bone block is sculpted into a precise rectangular bridge. We typically aim for a bone bridge measuring 7 to 8 millimeters in width and 10 millimeters in depth.
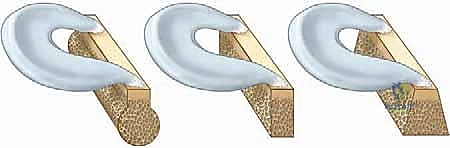
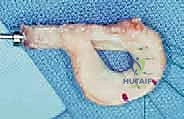
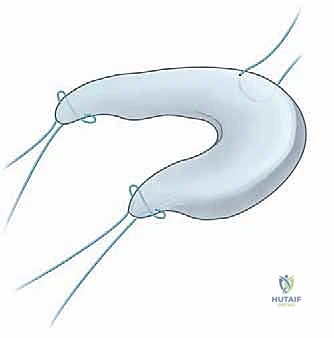
Using a specialized sizing block and a sagittal saw, the bone bridge is fashioned to match the dimensions of the slot that will be created in the patient's tibia. A traction suture (typically a #2 high-strength suture) is passed through the posterior aspect of the bone bridge; this will be utilized to pull the graft into the tibial slot. Additionally, a vertical mattress traction suture is placed through the posterior horn of the meniscal tissue itself to assist with intra-articular manipulation and reduction.
For a medial meniscal transplant, the preparation differs significantly. The allograft is prepared with two distinct cylindrical bone plugs—one for the anterior horn and one for the posterior horn—typically measuring 7 to 8 millimeters in diameter. Using a core reamer, these plugs are carefully harvested from the allograft tibial plateau, ensuring the meniscal root attachments remain pristine and securely attached to the bone. Traction sutures are placed through the base of each bone plug to facilitate their later passage into the corresponding tibial tunnels.
Osseous Tunnel and Slot Preparation
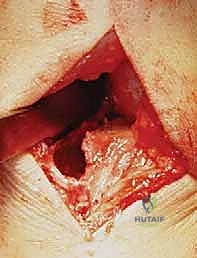
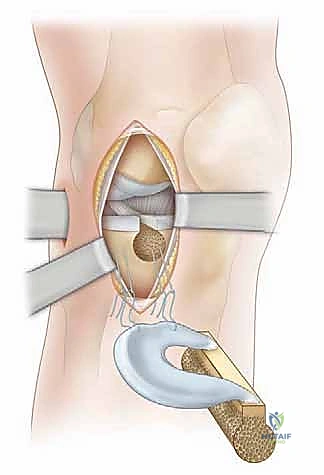
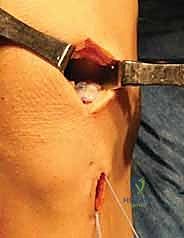
Returning to the patient, we proceed with the creation of the osseous fixation points. For the lateral bone bridge technique, a specialized drill guide is introduced through the anteromedial portal and positioned precisely over the previously marked anterior and posterior horn footprints. A guide pin is drilled from the anteromedial tibia, exiting at the posterior horn footprint. A cannulated reamer (matching the width of our prepared bone bridge, typically 7 or 8 mm) is then passed over the guide pin to create the posterior aspect of the slot.
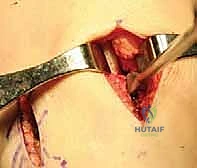
This process is repeated for the anterior horn footprint. Once both the anterior and posterior cylindrical tunnels are established, a box cutter or a specialized rasp is utilized to connect the two tunnels, creating a continuous, rectangular trough or "slot" in the sagittal plane of the lateral tibial plateau. The depth of the slot is carefully calibrated to match the 10-millimeter depth of our allograft bone bridge. The slot is then meticulously cleared of any bony debris using a shaver and a rasp to ensure smooth graft insertion.
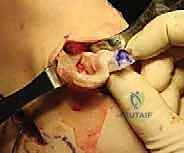
For the medial two-bone-plug technique, individual tunnels are drilled. A standard ACL tibial drill guide is used to target the posterior horn footprint of the medial meniscus. A guide pin is drilled, followed by a reamer that matches the diameter of the posterior bone plug (typically 7 or 8 mm). A separate tunnel is then drilled for the anterior horn footprint. Careful attention must be paid to the trajectory of these tunnels to avoid convergence and to ensure they do not compromise the native ACL tibial footprint.
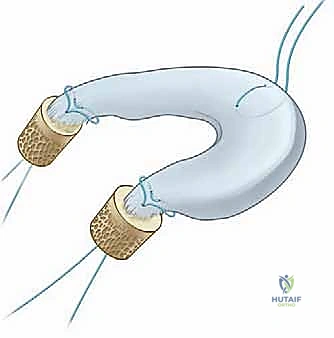
Allograft Insertion and Final Fixation
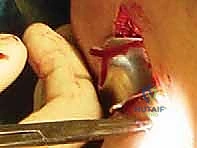
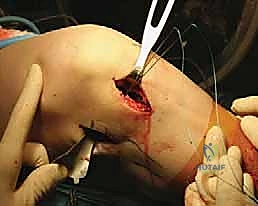
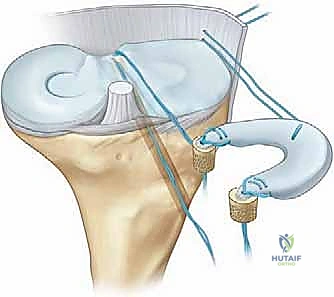
With the host bed and osseous tunnels prepared, we proceed to the critical phase of graft insertion. For the lateral meniscal transplant, an accessory anterolateral mini-arthrotomy is often required to accommodate the bone bridge. A passing suture is introduced through the tibial slot and retrieved out the anterolateral portal. This suture is tied to the traction suture on the allograft bone bridge. Applying gentle, sustained tension on the passing suture while simultaneously pushing the graft through the portal, the bone bridge is carefully docked into the tibial slot.
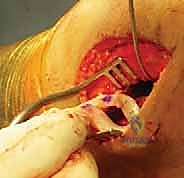
Once the bone bridge is fully seated, we must secure the peripheral meniscal tissue to the host capsule. This is achieved using a combination of inside-out and all-inside suturing techniques. An open posterolateral approach is performed, navigating between the IT band and biceps femoris, to protect the common peroneal nerve and retrieve the inside-out needles. We systematically place vertical mattress sutures, starting from the posterior horn and working anteriorly, securing the allograft to the bleeding, prepared host rim.
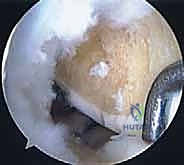
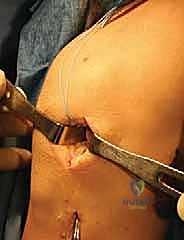
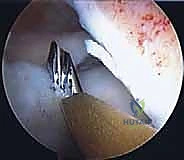
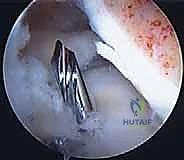
The anterior horn is typically secured using outside-in or direct open suturing techniques. Finally, the bone bridge is secured within the slot. This can be accomplished with an interference screw placed adjacent to the bone bridge, or by tying the traction sutures over a cortical button or bone bridge on the anterior tibial cortex. Thorough probing confirms the stability of the graft, ensuring there is no excessive mobility or extrusion.
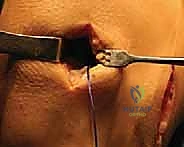
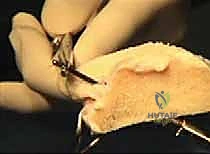
For the medial transplant, the posterior bone plug traction sutures are passed through the posterior tunnel, and the anterior plug sutures through the anterior tunnel. The graft is reduced into the joint, and the bone plugs are seated into their respective sockets. Peripheral fixation is then achieved via a posteromedial approach, protecting the saphenous nerve and retrieving inside-out sutures to secure the posterior and middle thirds of the medial meniscus. The bone plug sutures are then tied over a cortical bridge on the anteromedial tibia.
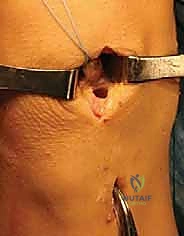
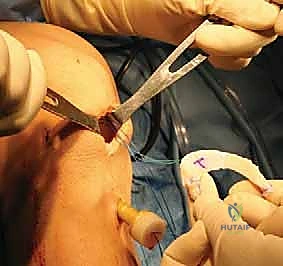
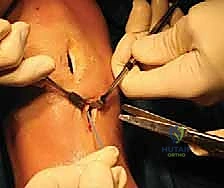
Before closing, we perform a final arthroscopic sweep, dynamically ranging the knee to ensure the graft tracks smoothly without impingement or subluxation. The arthrotomy and portal sites are closed in layers, and the knee is placed in a hinged brace locked in full extension.
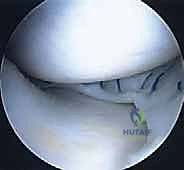
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique and stringent patient selection, Meniscal Allograft Transplantation is a complex biological procedure with a recognized complication profile. Understanding these potential pitfalls is critical for informed patient consent and prompt postoperative management. Complications can be broadly categorized into intraoperative technical errors, early postoperative biological failures, and late mechanical deterioration.
Intraoperative complications primarily
