Median Nerve Decompression: Mastering Pronator and Anterior Interosseous Syndromes
Key Takeaway
This masterclass guides fellows through median nerve decompression for pronator and anterior interosseous syndromes. We cover comprehensive surgical anatomy, meticulous intraoperative techniques, and critical pearls to navigate this complex procedure. From initial incision to nerve release and closure, learn precise steps to optimize patient outcomes and manage potential complications, ensuring thorough decompression of all potential impingement sites.
Welcome, colleagues and fellows, to the operating theater. Today, we are tackling a fascinating and clinically challenging domain of upper extremity peripheral nerve surgery: the comprehensive decompression of the median nerve in the proximal forearm. Specifically, we will address the surgical management of Pronator Syndrome and Anterior Interosseous Nerve (AIN) Syndrome. While these represent distinct compression neuropathies with unique clinical presentations, their anatomical proximity and shared surgical corridor often necessitate a combined, extensile approach. Our ultimate objective is a thorough, systematic release of all potential compression points to alleviate the patient's symptoms and restore optimal neurovascular function.
Comprehensive Introduction and Patho-Epidemiology
To master the surgical management of proximal median nerve compression, one must first possess a granular understanding of the underlying pathology and epidemiology. Pronator syndrome is a compression neuropathy of the median nerve proper, occurring in the proximal forearm. It typically presents with an insidious onset of paresthesias in the median nerve distribution of the hand, often accompanied by a deep, aching pain localized to the proximal volar forearm. Unlike carpal tunnel syndrome (CTS), nighttime awakening is relatively uncommon, and the pain is classically exacerbated by activities involving repetitive forearm pronation or heavy gripping. Patients may exhibit mild weakness in median-innervated extrinsic muscles, but sensory symptoms predominate.
Anterior interosseous nerve (AIN) syndrome, conversely, is primarily a motor neuropathy. The AIN is the largest branch of the median nerve, and its compression manifests as diminished motor function or frank paralysis of the muscles it innervates: the flexor pollicis longus (FPL), the flexor digitorum profundus (FDP) to the index and sometimes long fingers, and the pronator quadratus. Patients typically complain of a spontaneous loss of dexterity, classically struggling with fine motor tasks such as picking up small objects or making an "OK" sign due to the inability to flex the interphalangeal (IP) joint of the thumb and the distal interphalangeal (DIP) joint of the index finger. Crucially, because the AIN is devoid of cutaneous sensory fibers, decreased cutaneous sensation is not a feature of pure AIN syndrome, though patients may experience a deep, poorly localized aching pain in the forearm due to proprioceptive sensory fibers.
The epidemiology of these syndromes dictates a high index of suspicion, as they are significantly less common than distal compressions like CTS, leading to frequent misdiagnosis. Pronator syndrome accounts for a mere fraction of all median nerve entrapments, frequently seen in manual laborers, weightlifters, and athletes involved in throwing or racquet sports. AIN syndrome is even rarer and can occur spontaneously, following trauma, or as a result of strenuous exertion. It is vital to differentiate these mechanical compression syndromes from Parsonage-Turner syndrome (neuralgic amyotrophy), an inflammatory mononeuritis multiplex that frequently targets the AIN. Parsonage-Turner typically presents with a prodromal viral-type illness and severe, unrelenting shoulder or proximal arm pain that precedes the onset of profound weakness. Recognizing this distinction is paramount, as Parsonage-Turner syndrome generally resolves with conservative management and time, rendering surgical decompression ineffective and potentially harmful.
Detailed Surgical Anatomy and Biomechanics
The median nerve is the primary workhorse of the volar forearm, and its journey from the brachial plexus through the elbow and into the forearm is fraught with potential anatomical choke points. Understanding this complex three-dimensional anatomy is non-negotiable for the peripheral nerve surgeon. In the distal third of the arm, the median nerve descends medial to the biceps brachii and anterior to the brachialis muscle. It is intimately associated with the brachial artery, typically lying just medial to it. Proximally, surgeons must be vigilant for the presence of a supracondylar process—a bony exostosis arising from the anteromedial aspect of the distal humerus, present in approximately 1% to 3% of the population. When present, the ligament of Struthers, a fibrous band extending from this process to the medial epicondyle, forms a fibro-osseous tunnel through which the median nerve and brachial artery pass, creating a prime site for proximal compression.
As the median nerve traverses the antecubital fossa, it passes deep to the lacertus fibrosus (bicipital aponeurosis). This strong fascial expansion originates from the biceps tendon and sweeps medially to blend with the deep fascia of the forearm. Biomechanically, the lacertus fibrosus tightens significantly during active forearm pronation combined with elbow flexion, creating a dynamic compression mechanism. Distal to the lacertus, the nerve encounters the pronator teres. Classically, the median nerve passes between the superficial (humeral) head and the deep (ulnar) head of the pronator teres. However, anatomical variations are the rule rather than the exception. In up to 20% of individuals, the deep head is entirely absent or exists merely as a tenuous fibrous band. Less commonly, the nerve may pass deep to both heads or pierce the substance of the humeral head itself. The fascial bands within the pronator teres, particularly at the tendinous origin of the deep head, are frequent culprits in pronator syndrome.
Distal to the pronator teres, the median nerve and its newly arborized anterior interosseous branch dive beneath the fibrous arcade of the flexor digitorum superficialis (FDS). This aponeurotic arch spans the humeroulnar and radial origins of the FDS. The proximal edge of this arcade is often thickened and unyielding, acting as a guillotine against the nerve during forceful finger flexion and forearm pronation. The AIN itself branches from the radial, dorsal, or ulnar aspect of the median nerve (most commonly radial) typically just distal to the pronator teres. It travels distally along the interosseous membrane. The surgeon must also be aware of anomalous structures that can cause compression, such as Gantzer’s muscle (an accessory head of the flexor pollicis longus present in up to 45% of limbs), an accessory bicipital aponeurosis, or a persistent median artery. Furthermore, the Martin-Gruber anastomosis, a communicating motor branch from the median nerve (or AIN) to the ulnar nerve in the forearm, is present in roughly 15% of the population and must be recognized to avoid iatrogenic injury and to understand atypical electrodiagnostic findings.
Exhaustive Indications and Contraindications
Patient selection is the cornerstone of successful outcomes in proximal median nerve decompression. The diagnosis is primarily clinical, relying on a meticulous history and a highly specific physical examination. For pronator syndrome, indications for surgery include persistent symptoms refractory to a minimum of 3 to 6 months of non-operative management, which includes activity modification, splinting, and non-steroidal anti-inflammatory drugs. Clinical provocative tests are essential: pain reproduced by resisted forearm pronation with the elbow extended implicates the pronator teres; pain with resisted elbow flexion and supination implicates the lacertus fibrosus; and pain with resisted flexion of the proximal interphalangeal (PIP) joint of the middle finger implicates the FDS arcade. A positive Tinel's sign over the proximal forearm, rather than the carpal tunnel, further localizes the lesion.
For AIN syndrome, surgical indications are slightly more nuanced. Because spontaneous recovery can occur, particularly if the etiology is a subclinical neuritis rather than pure mechanical compression, a period of observation lasting 3 to 6 months is generally recommended. However, if there is no clinical or electrodiagnostic evidence of reinnervation after this period, or if the patient presents with a distinct, documented space-occupying lesion (such as a lipoma or ganglion cyst) causing the compression, early surgical intervention is strongly indicated. The pathognomonic clinical sign is the inability to form an "OK" sign, resulting in a flattened pinch posture due to FPL and index FDP weakness.
Electrodiagnostic studies (EMG/NCS) play a complex role. In pronator syndrome, NCS are notoriously unreliable and frequently normal, as the dynamic, activity-dependent nature of the compression is not captured in a static testing environment. However, EMG is invaluable in AIN syndrome, typically demonstrating fibrillation potentials and positive sharp waves in the FPL, FDP, and pronator quadratus, confirming the diagnosis and establishing a baseline for recovery. Furthermore, EMG/NCS are critical for ruling out concurrent carpal tunnel syndrome, cervical radiculopathy (C6-C7), or a double crush phenomenon. Contraindications to surgery are primarily related to misdiagnosis, particularly failing to recognize Parsonage-Turner syndrome, or severe medical comorbidities that preclude the safe administration of anesthesia.
| Category | Pronator Syndrome | Anterior Interosseous Nerve (AIN) Syndrome |
|---|---|---|
| Primary Indications | Refractory pain/paresthesia > 3-6 months; Positive provocative tests (resisted pronation, FDS testing); Failure of conservative care. | Persistent motor deficit > 3-6 months; Confirmed space-occupying lesion; Lack of EMG improvement over time. |
| Clinical Hallmarks | Volar forearm pain; Paresthesia in median digits; Positive proximal Tinel's; Minimal motor weakness. | Pure motor weakness (FPL, index FDP, PQ); Inability to make "OK" sign; No sensory deficits. |
| EMG/NCS Utility | Often equivocal or negative; Useful to rule out CTS or cervical radiculopathy. | Highly sensitive; Shows denervation in AIN-supplied musculature; Tracks reinnervation. |
| Absolute Contraindications | Active local infection; Unmanaged coagulopathy; Diagnosis of Parsonage-Turner Syndrome. | Active local infection; Unmanaged coagulopathy; Early phase (< 3 months) without mass lesion; Parsonage-Turner Syndrome. |
| Relative Contraindications | Severe medical comorbidities; Unrealistic patient expectations; Active workers' compensation litigation (proceed with caution). | Severe medical comorbidities; Concomitant severe cervical spine disease confounding recovery. |
Pre-Operative Planning, Templating, and Patient Positioning
Before a scalpel is ever handed to the surgeon, meticulous preoperative planning must be executed. This begins with a thorough review of all diagnostic imaging. While plain radiographs are generally low-yield for soft tissue entrapments, an anteroposterior and lateral view of the elbow is mandatory to rule out a supracondylar process of the humerus. If a supracondylar process is identified, the surgical approach must be planned to extend further proximally to ensure the ligament of Struthers can be identified and resected. Ultrasound and Magnetic Resonance Imaging (MRI) are highly sensitive for identifying space-occupying lesions, such as lipomas, ganglion cysts, or anomalous muscle bellies (like a hypertrophic Gantzer's muscle), which may alter the standard anatomical dissection.
In the operating theater, the patient is positioned supine with the affected upper extremity extended onto a standard radiolucent hand table or arm board. The arm must be entirely free to allow for full, unrestricted extension, flexion, pronation, and supination throughout the procedure, as dynamic testing of the fascial bands is a critical component of the decompression. A well-padded pneumatic tourniquet is applied high on the brachium. We typically inflate the tourniquet to 250 mmHg (or 100 mmHg above the patient's systolic blood pressure) after thorough exsanguination with an Esmarch bandage. A completely bloodless field is not just a convenience; it is an absolute necessity for the safe identification and preservation of the delicate vasa nervorum and the microscopic motor branches of the median nerve.
Draping must allow for an extensile exposure. The arm is prepped and draped in a standard sterile fashion, ensuring exposure from the mid-humerus down to the wrist flexion crease. Key anatomical landmarks are marked with a sterile surgical pen prior to exsanguination. These include the medial epicondyle, the biceps tendon, the brachial artery pulse, the elbow flexion crease, and the estimated course of the median nerve. The planned incision line—a modified Henry’s approach—is drawn, beginning proximally on the medial arm, crossing the antecubital fossa obliquely, and extending distally into the volar forearm. The surgeon must verbally confirm the tourniquet time and pressure with the anesthesia team, ensuring that ischemia time does not exceed 120 minutes without a reperfusion interval.
Step-by-Step Surgical Approach and Decompression Technique
Today, we will utilize an extensile exposure, a modification of Henry’s approach. While it can be associated with larger scars and potential cutaneous nerve injury, it offers the safest and most thorough exposure, allowing us to decompress all potential sites of compression, both proximally and distally, under direct visualization.
Superficial Dissection and Cutaneous Nerve Management
We begin by making a curvilinear skin incision. It originates on the medial aspect of the distal arm, approximately 5 cm proximal to the elbow flexion crease, nestled in the sulcus between the biceps and triceps. We then bring it gently across the elbow flexion crease, following the natural skin lines in a transverse or oblique fashion to prevent scar contracture, and extend it distally for approximately 10 to 12 cm into the proximal volar forearm.
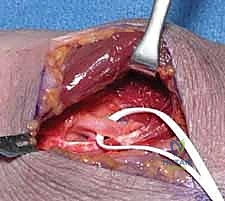
The initial incision must be carefully deepened through the subcutaneous fat using tenotomy scissors or a mosquito hemostat. Immediately identify and protect the superficial cutaneous nerves. The medial antebrachial cutaneous (MABC) nerve and its branches are highly variable and frequently cross the operative field. Similarly, branches of the lateral antebrachial cutaneous (LABC) nerve may be encountered radially. Use blunt dissection to spread tissues rather than cutting blindly. Damage to these nerves can lead to persistent, debilitating dysesthesias or painful neuromas that often eclipse the patient's original symptoms. Once identified, these nerves are atraumatically mobilized and protected with fine vessel loops.
Proximal Decompression: Struthers and Lacertus Fibrosus
Deepen the dissection through the superficial fascia to identify the median nerve proximal to the elbow flexion crease. It lies deep to the lacertus fibrosus and superficial to the brachialis muscle, immediately medial to the brachial artery.
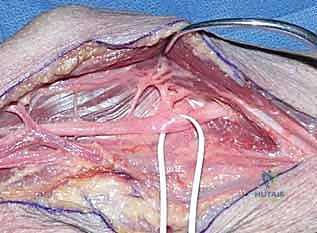
Once the median nerve is safely identified proximally, trace it distally. The first major point of compression encountered is the lacertus fibrosus. Identify its free proximal edge, which runs obliquely from the biceps tendon across the cubital fossa. Using fine, blunt-tipped scissors, carefully incise the lacertus fibrosus longitudinally, directly over the median nerve.
With the lacertus released, palpate the distal humerus proximally for a supracondylar process. If present, the ligament of Struthers will be seen extending from it to the medial epicondyle. Pass a right-angle clamp beneath the ligament to protect the nerve and artery, and resect a portion of the ligament to prevent reconstitution.
Pronator Teres Dissection and Release
Our next target is the pronator teres. Retract the proximal portion of the superficial pronator musculature gently. You will see the median nerve diving deep into the muscle belly. As we approach the pronator teres, be acutely aware of the motor branches supplying the flexor-pronator mass. These branches typically arise from the ulnar side of the median nerve. They are vital and must be protected at all costs; use a handheld nerve stimulator if there is any anatomical ambiguity.
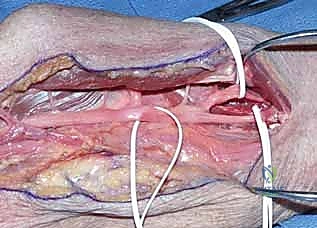
The median nerve typically passes between the superficial (humeral) and deep (ulnar) heads. In some cases, careful blunt dissection allows retraction of the entire pronator muscle mass in an ulnar direction, visualizing the nerve without formal release. However, if the tendinous bands of the pronator are tight, a release is mandatory. We carefully perform a step-cut (Z-lengthening) of the tendinous origin of the superficial head, or a fractional lengthening at its musculotendinous junction. If a distinct deep head is compressing the nerve, its tendinous insertion is also released.
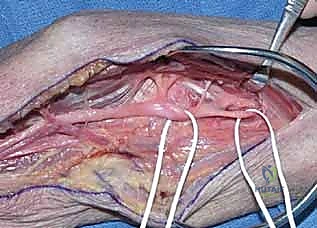
Releasing the pronator teres allows us to reflect the humeral head, significantly improving exposure. This reveals the median nerve, the AIN branching from its radial aspect, and the fibrous arcade of the flexor digitorum superficialis.
Decompressing the FDS Arcade and Final Inspection
Trace the median nerve distally to the fibrous arcade of the FDS. This strong aponeurotic band bridges the humeroulnar and radial heads of the FDS, often directly constricting the median nerve and the AIN. Identify the AIN branching from the radial side of the median nerve.
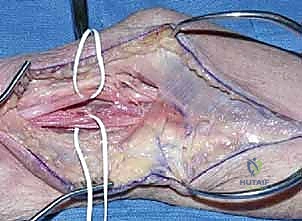
Carefully identify the proximal edge of the fibrous arcade. Using a scalpel or Stevens tenotomy scissors, incise the fibrous portion of the FDS arcade longitudinally. Ensure a complete release, extending the cut distally until the muscle bellies of the FDS are soft and yielding. The anterior interosseous nerve branches to the FPL and index FDP are extremely vulnerable here; meticulous, atraumatic technique under direct visualization with surgical loupes is non-negotiable.

Once all potential compression sites have been thoroughly released, perform a final, comprehensive inspection. Pass a finger along the course of the nerve to ensure no hidden fascial bands remain. Deflate the tourniquet, achieve meticulous hemostasis with bipolar electrocautery to prevent hematoma, and irrigate the wound copiously. The pronator teres is loosely approximated if a Z-lengthening was performed, and the skin is closed in a layered fashion.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, complications following median nerve decompression in the proximal forearm can and do occur. The most devastating complication is iatrogenic nerve injury. Injury to the main trunk of the median nerve is exceptionally rare but catastrophic, leading to profound sensory and motor deficits in the hand. More common is injury to the delicate motor branches supplying the flexor-pronator mass or the AIN itself during dissection of the pronator teres or FDS arcade. Incidence of transient neuropraxia due to excessive traction is estimated at 2% to 5%, usually resolving spontaneously within weeks to months. Permanent motor branch injury requires complex salvage procedures, such as nerve grafting or tendon transfers (e.g., brachioradialis to FPL transfer for permanent AIN palsy).
Cutaneous nerve injury is perhaps the most frequent source of postoperative morbidity. The MABC and LABC nerves are highly susceptible to transection or traction injury during the initial superficial exposure. Incidence of cutaneous nerve injury ranges from 5% to 10% in extensile approaches. This can result in localized numbness, which is generally well-tolerated, or the formation of a painful neuroma, which is highly debilitating. Salvage management for a symptomatic neuroma involves surgical re-exploration, excision of the neuroma, and capping the proximal nerve stump or burying it deep within the adjacent muscle belly (e.g., brachialis) to prevent mechanical irritation.
Postoperative hematoma is a significant risk due to the extensive muscular dissection required. A large hematoma can cause secondary compression of the median nerve, necessitating emergent surgical evacuation. Meticulous bipolar hemostasis after tourniquet deflation is the primary preventative measure. Infection rates are low (<1%) but require standard protocols of irrigation, debridement, and targeted antibiotic therapy if they occur. Finally, incomplete release or failure to identify all anatomical variants (such as an unrecognized Gantzer's muscle) can lead to persistent or recurrent symptoms, necessitating revision decompression.
| Complication | Estimated Incidence | Prevention Strategy | Salvage / Management |
|---|---|---|---|
| Cutaneous Nerve Injury (MABC/LABC) | 5% - 10% | Meticulous superficial dissection; use of loupe magnification; vessel loops for retraction. | Neuroma excision; burying proximal stump in muscle; targeted nerve blocks. |
| Transient Neuropraxia | 2% - 5% | Avoid excessive traction on the median nerve; limit tourniquet time < 120 mins. | Observation; supportive splinting; physical therapy; usually resolves in 3-6 months. |
| Motor Branch Transection (AIN or PT/FCR branches) | < 1% | Identify branches on the ulnar side of median nerve; direct visualization before cutting FDS arcade. | Primary microsurgical repair; nerve grafting; late tendon transfers (e.g., BR to FPL). |
| Postoperative Hematoma | 1% - 3% | Complete tourniquet deflation and meticulous bipolar hemostasis prior to closure; use of a drain if oozing persists. | Emergent surgical evacuation if causing secondary nerve compression; otherwise, observation. |
| Incomplete Decompression / Recurrence | 2% - 5% | Extensile approach; systematic release of all 5 potential choke points; dynamic intraoperative testing. | Revision surgery with broader exposure; neurolysis; potential wrapping with nerve conduits/vein grafts. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation protocol following proximal median nerve decompression is designed to balance the need for tissue healing with the imperative to prevent perineural adhesions and restore functional range of motion. Rehabilitation is generally divided into three distinct phases, tailored to the extent of the surgical release and the patient's preoperative baseline.
Phase I encompasses the immediate postoperative period, from days 0 to 14. Upon leaving the operating room, the patient is placed in a bulky, soft compressive dressing with a posterior plaster splint holding the elbow in 90 degrees of flexion and the forearm in neutral rotation. This minimizes tension on the healing tissues, particularly if a Z-lengthening of the pronator teres was performed. During this phase, strict elevation is mandated to control edema. Active range of motion of the shoulder and digits is encouraged immediately to prevent stiffness and promote venous return. At the 10 to 14-day mark, the splint and sutures are removed.
Phase II spans weeks 2 through 6. The primary goal during this phase is the initiation of neural gliding exercises and the restoration of elbow and forearm range of motion. Patients are instructed in specific median nerve gliding exercises, which involve sequential movements of the neck, shoulder, elbow, wrist, and fingers to mobilize the nerve through its anatomical bed without placing it under excessive longitudinal strain. Active and active-assisted range of motion for elbow flexion/extension and forearm pronation/supination is initiated. Gentle scar massage and desensitization techniques are also introduced once the incision is fully healed to prevent restrictive scar adherence to the underlying fascia.
Phase III begins at week 6 and extends until maximum medical improvement is achieved, typically around 3 to 6 months postoperatively. This phase focuses on progressive strengthening and a return to full functional activities. Isotonic and isokinetic strengthening exercises for the flexor-pronator mass are gradually introduced. For patients recovering from AIN syndrome, specific attention is paid to re-educating the FPL and index FDP muscles as reinnervation occurs, often utilizing biofeedback and fine motor dexterity tasks. Heavy lifting, forceful gripping, and repetitive pronation activities are slowly integrated into the patient's routine, with close monitoring for any recurrence of symptoms.
Summary of Landmark Literature and Clinical Guidelines
The evolution of our understanding and surgical management of proximal median nerve compression is deeply rooted in several landmark anatomical and clinical studies. The foundational description of Anterior Interosseous Nerve syndrome is credited to Kiloh and Nevin in their seminal 1952 paper, which clearly delineated the pure motor presentation of this neuropathy and differentiated it from generalized median nerve lesions. Their work remains the bedrock upon which our clinical diagnosis of AIN syndrome is built.
Further anatomical clarity was provided by Spinner in the 1970s. Spinner’s exhaustive cadaveric studies meticulously detailed the anatomical variations of the pronator teres, the FDS arcade, and the branching patterns of the AIN. He was instrumental in identifying the multiple potential sites of compression, emphasizing that a localized, limited incision is often inadequate for a complete decompression. His work established the rationale for the extensile approach we utilize today. Eversmann's contributions further refined the surgical indications, particularly highlighting the dynamic nature of pronator syndrome and the necessity of intraoperative provocative testing to confirm the exact site of fascial impingement.
Modern clinical guidelines, supported by the American Society for Surgery of the Hand (ASSH), emphasize a systematic approach to diagnosis, relying heavily on specific clinical examination findings over purely electrodiagnostic criteria, particularly for pronator syndrome. Current literature strongly supports a trial of conservative management for 3 to 6 months for both syndromes, reserving surgical intervention for refractory cases or those with progressive motor deficits. Recent outcome studies demonstrate that with accurate diagnosis and comprehensive, multi-site surgical decompression using the techniques described herein, patients can expect a high rate of symptom relief and functional return, cementing this procedure as a critical tool in the peripheral nerve surgeon's armamentarium.
