Comprehensive Management of Severe Intrinsic Contractures and Dupuytren Disease
Key Takeaway
Severe intrinsic contractures of the hand, often secondary to compartment syndrome, Volkmann ischemic contracture, or progressive Dupuytren disease, present profound reconstructive challenges. Successful management requires a meticulous understanding of hand biomechanics, precise surgical release of fibrotic muscle tissue, and advanced soft-tissue coverage techniques, such as cross-arm flaps for web space deepening. This guide details the indications, operative steps, and postoperative protocols for restoring optimal hand function.
INTRODUCTION TO INTRINSIC CONTRACTURES AND MUSCLE FIBROSIS
Severe intrinsic contractures of the hand represent a complex reconstructive challenge, typically arising from profound muscle fibrosis secondary to ischemic events, trauma, or progressive fibroproliferative disorders. The two most prominent etiologies encountered by the reconstructive hand surgeon are Volkmann ischemic contracture (a sequela of untreated or delayed treatment of compartment syndrome) and Dupuytren disease.
Both conditions lead to the replacement of normal, compliant myofascial structures with rigid, inelastic fibrous tissue. This results in severe biomechanical derangement, characterized by fixed flexion deformities, loss of the functional web spaces, and profound disability. The management of these conditions requires a highly structured, evidence-based approach, combining radical surgical release of the fibrotic tissues with sophisticated soft-tissue reconstruction to restore the functional cascade of the hand.
VOLKMANN ISCHEMIC CONTRACTURE AND COMPARTMENT SYNDROME
Volkmann ischemic contracture is the devastating end-stage result of an acute compartment syndrome that has been left untreated or inadequately decompressed. The ischemic cascade leads to myonecrosis, predominantly affecting the deep flexor compartment of the forearm (flexor digitorum profundus and flexor pollicis longus) and the intrinsic muscles of the hand.
Pathoanatomy and Biomechanics
The ischemic insult triggers a massive inflammatory response, leading to the phagocytosis of necrotic muscle fibers and subsequent replacement by dense, unyielding scar tissue. As this scar tissue matures, it contracts, drawing the wrist and digits into a characteristic posture:
* Wrist flexion
* Metacarpophalangeal (MCP) joint hyperextension
* Proximal interphalangeal (PIP) and distal interphalangeal (DIP) joint flexion
* Thumb adduction and flexion
Clinical Pearl: The severity of the contracture is directly proportional to the duration and magnitude of the ischemic insult. In severe cases, the entire flexor muscle mass is replaced by a fibrous cord, and the intrinsic muscles of the hand undergo severe fibrotic degeneration, leading to an "intrinsic-plus" or "intrinsic-minus" hand depending on the specific compartments involved.
Diagnostic Modalities
While acute compartment syndrome is a clinical diagnosis necessitating emergent fasciotomy, the evaluation of established Volkmann contracture or chronic exertional compartment syndrome relies on advanced diagnostic adjuncts:
* Intracompartmental Pressure Monitoring: Essential for diagnosing acute or chronic exertional compartment syndrome.
* Magnetic Resonance Imaging (MRI): Highly valuable in assessing the extent of muscle necrosis and fibrosis. MRI can precisely delineate viable muscle from fibrotic scar tissue, guiding the surgical approach for muscle slide procedures or free functional muscle transfers (e.g., free gracilis transfer).
SURGICAL RELEASE OF SEVERE INTRINSIC CONTRACTURES
The primary goal of surgery in severe intrinsic contractures is to restore a functional hand position, enabling grasp and pinch. This often requires a combination of muscle releases, tendon lengthenings, and complex web space reconstructions.
Management of the Adducted Thumb
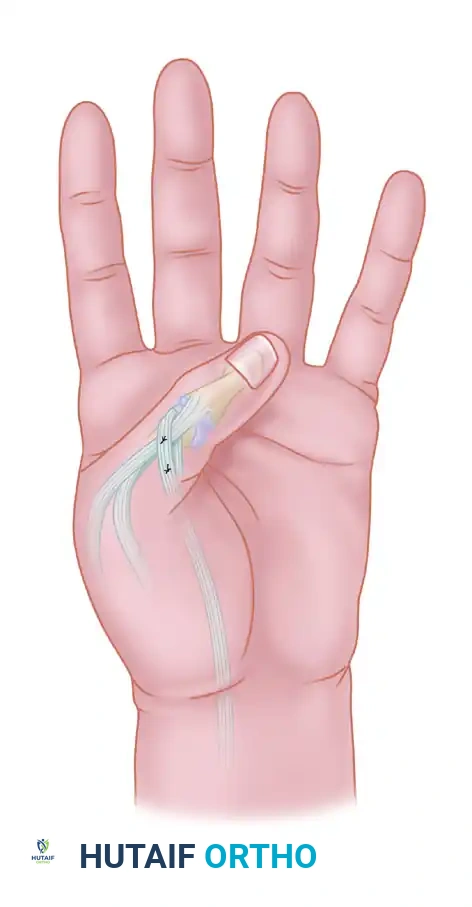
One of the most debilitating components of severe intrinsic fibrosis is the adduction contracture of the thumb. Fibrosis of the adductor pollicis and the first dorsal interosseous muscle obliterates the first web space, destroying the patient's ability to perform a functional pinch or grasp large objects.
When the contracture is severe, simple soft-tissue release is insufficient. The fibrotic muscle must be radically recessed or excised, and the resulting massive soft-tissue defect must be covered with vascularized tissue to prevent recurrence.
Surgical Technique: Web Space Deepening with Cross-Arm Flap
In cases of profound thumb web space contracture, a staged cross-arm flap provides excellent, durable coverage after radical release.
Step 1: Release and Defect Creation
1. An incision is made along the contracted first web space.
2. The fibrotic fascia, adductor pollicis insertion, and first dorsal interosseous are systematically released or recessed.
3. In extreme cases, the index metacarpal may be excised to artificially widen the web space and provide local filleted skin for coverage.
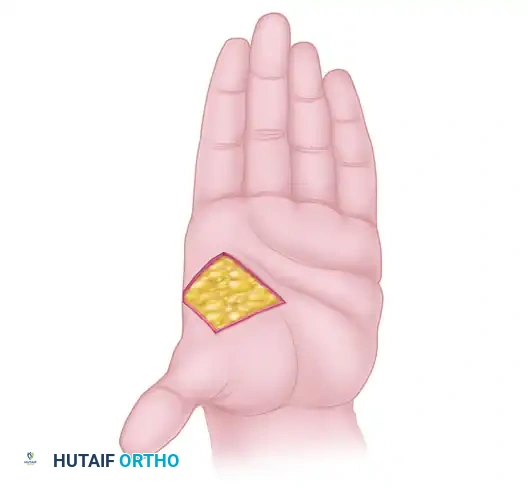
4. Following the release, the thumb is abducted and extended, revealing a large, diamond-shaped soft-tissue defect.

Figure 1: Web space deepening after skin division and muscle recession, demonstrating the raw, diamond-shaped palmar and dorsal defect.
Step 2: First-Stage Flap Coverage
1. A cross-arm flap is designed on the contralateral arm. The flap is typically designed with two triangular components: a distal triangular flap (b) and a proximal triangular flap (a).
2. The distal triangular flap (b) is elevated and sutured directly into the dorsal aspect of the thumb web defect.
3. The arm is immobilized in a secure, comfortable position against the contralateral hand to prevent tension on the pedicle.
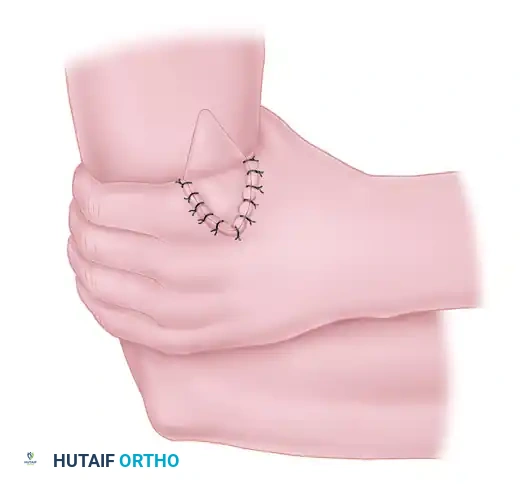
Figure 2: Position of the hand with the triangular distal flap (b) sutured into the dorsal thumb web defect. The outline of the proximal triangular flap (a), which will be used for palmar web coverage during the second stage, is visible.
Step 3: Second-Stage Division and Inset
1. Approximately 3 weeks later, after neovascularization from the recipient bed has occurred, the flap pedicle is divided.
2. The proximal triangular flap (a) is elevated from the donor arm, transferred, and inset into the palmar aspect of the web space defect.
3. Meticulous closure is performed to ensure a deep, wide, and supple first web space.
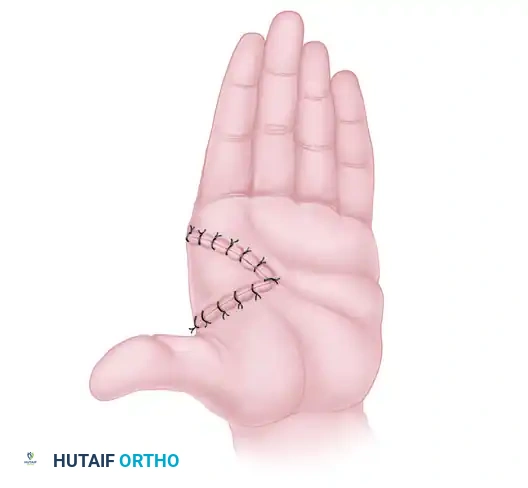
Figure 3: Final web space reconstruction after the transfer and inset of the palmar flap at the second-stage operation, 3 weeks later.
Surgical Warning: When performing radical intrinsic releases, the surgeon must meticulously identify and protect the digital neurovascular bundles, which are often displaced or encased within the dense fibrotic tissue.
DUPUYTREN DISEASE: PATHOGENESIS AND CLINICAL PRESENTATION
Dupuytren disease is a progressive, proliferative fibroplasia of the subcutaneous palmar tissue. It manifests as palpable nodules and longitudinal cords that ultimately result in secondary, irreversible flexion contractures of the finger joints (MCP and PIP joints).
Pathophysiology and Secondary Changes
The fundamental pathological unit of Dupuytren disease is the myofibroblast. Driven by complex genetic and environmental factors, normal fascial bands hypertrophy and contract, forming pathological cords.
Secondary changes associated with advanced disease include:
* Thinning of the overlying subcutaneous fat.
* Dense adhesion of the fibrotic cords to the overlying dermis.
* Pitting or dimpling of the palmar skin due to contracture of the vertical septa of Legueu and Juvara.
The rate of progression is highly variable. While some patients experience rapid, severe flexion deformities over a few months, the majority develop deformities gradually over several years. Exacerbations and remissions are common, but spontaneous regression is exceedingly rare.
The Dupuytren Diathesis
Ectopic deposits of Dupuytren-like fibroplasia can occur in other anatomical regions. Patients exhibiting these ectopic lesions are considered to have a "Dupuytren diathesis," indicating a highly aggressive, genetically driven form of the disease that is prone to rapid progression and high recurrence rates following surgery.
Associated conditions include:
* Ledderhose Disease: Plantar fibromatosis occurring in the medial plantar fascia (seen in approximately 5% of Dupuytren patients).
* Peyronie Disease: Plastic induration and curvature of the penis (seen in approximately 3% of patients).
* Garrod Nodules ("Knuckle Pads"): Fibrotic deposits on the dorsum of the proximal interphalangeal joints.
Demographics and Etiology
Dupuytren contracture is predominantly a disease of middle-aged to elderly adults, most commonly presenting in the 40s to 60s.
* Gender: It occurs 10 times more frequently in men than in women. According to McFarlane's extensive epidemiological studies, the disease manifests significantly earlier in men (average age 33 to 63 years) compared to women (46 to 70 years).
* Genetics: It is most prevalent in individuals of Scandinavian and Celtic descent (often referred to as the "Viking disease"). It is occasionally reported in populations of African descent and is rare in Asian populations.
* Comorbidities: The lesion is more frequent and often more severe in individuals with diabetes mellitus and epilepsy (up to 42% prevalence in some epileptic cohorts). The relationship with alcoholism remains heavily debated in the literature, with conflicting reports regarding direct causation.
* Mortality: Mikkelsen et al. noted that mortality rates may be statistically increased in men who develop aggressive Dupuytren disease before the age of 60, likely due to associated systemic comorbidities.
Clinical Pearl: While the exact etiology remains elusive, hand trauma and repetitive manual labor are considered potential contributing factors. The histological presence of hemosiderin within the nodules suggests micro-hemorrhage from fascial tears. However, because the nondominant hand is affected just as frequently as the dominant hand, mechanical trauma alone cannot be the sole causative agent. In genetically susceptible individuals, a single traumatic event may precipitate the onset of the disease.
SURGICAL MANAGEMENT OF DUPUYTREN DISEASE
The indication for surgical intervention in Dupuytren disease is typically an MCP joint contracture exceeding 30 degrees, or any degree of PIP joint contracture that impairs hand function.
Surgical Approaches
- Regional Fasciectomy: The gold standard treatment. It involves the excision of the diseased fascial cords while preserving the normal palmar fascia. Incisions are typically made using Brunner zig-zag lines or longitudinal incisions modified with Z-plasties to prevent scar contracture.
- Dermofasciectomy: Indicated for recurrent disease or severe skin involvement. The diseased fascia and the overlying involved skin are excised en bloc, and the defect is covered with a full-thickness skin graft. This significantly reduces the local recurrence rate.
- Percutaneous Needle Aponeurotomy (PNA): A minimally invasive option for isolated, well-defined cords, primarily at the MCP joint. It offers rapid recovery but carries a higher recurrence rate compared to open fasciectomy.
Biomechanics of Cord Excision
Understanding the specific anatomical cords is critical for safe excision:
* Pretendinous Cord: Primarily responsible for MCP joint contracture.
* Central Cord: An extension of the pretendinous cord into the digit, causing PIP joint flexion.
* Spiral Cord: The most dangerous structure surgically. It spirals around the neurovascular bundle, displacing it centrally, superficially, and proximally. The surgeon must identify the bundle proximally in normal tissue before tracing it distally through the spiral cord to avoid iatrogenic transection.
POSTOPERATIVE PROTOCOLS AND REHABILITATION
Whether treating Volkmann ischemic contracture or Dupuytren disease, the surgical release is only the first step. Rigorous postoperative rehabilitation is mandatory to maintain the surgical gains and prevent recurrent stiffness.
Splinting and Orthotics
- Immediate Postoperative Phase: The hand is immobilized in a bulky, non-compressive dressing with a volar orthosis. For Dupuytren releases, the MCP and PIP joints are splinted in maximum safe extension. For intrinsic releases, the hand is splinted in an intrinsic-minus position (MCP joints extended, IP joints flexed) to stretch the healing intrinsic muscles.
- Subacute Phase (1-3 weeks): Custom thermoplastic extension splints are fabricated. Night splinting is strictly enforced for 3 to 6 months to counteract the physiological forces of scar contracture.
Hand Therapy
Supervised occupational therapy begins within the first week postoperatively.
* Active and active-assisted range of motion (ROM) exercises are initiated to promote tendon gliding and prevent peritendinous adhesions.
* Scar massage and silicone gel sheeting are utilized once the incisions have fully epithelialized to soften the surgical scar and reduce dermal tethering.
* Strengthening exercises are gradually introduced at 6 to 8 weeks, focusing on restoring grip and pinch strength.
CONCLUSION
The management of severe intrinsic contractures and Dupuytren disease demands a profound understanding of hand anatomy, biomechanics, and the pathophysiology of fibroplasia. From the radical excision of ischemic muscle in Volkmann contracture to the meticulous dissection of the spiral cord in Dupuytren disease, the surgeon must employ a precise, evidence-based approach. By combining advanced surgical techniques—such as the cross-arm flap for web space deepening—with rigorous postoperative rehabilitation, the reconstructive surgeon can successfully restore form and function to the severely compromised hand.
You Might Also Like