Arthroscopic Scapulothoracic Decompression: A Masterclass for Fellows
Key Takeaway
This masterclass guides fellows through arthroscopic scapulothoracic decompression for snapping scapula syndrome. We cover essential anatomy, meticulous patient positioning, and a granular, real-time surgical execution from initial portal placement to final debridement. Emphasizing critical pearls, pitfalls, and post-operative care, this session ensures a comprehensive understanding of preventing and managing complications for optimal patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
Scapulothoracic disorder, frequently referred to in the surgical vernacular as snapping scapula syndrome, represents a complex interplay of mechanical impingement, localized frictional forces, and chronic bursal inflammation. This condition is not merely a nuisance of audible joint noise; it is a profoundly debilitating pathology that disrupts the kinetic chain of the upper extremity, leading to severe functional limitation and chronic pain. As orthopedic surgeons, our mandate is to differentiate between benign physiological noise—which is present in up to one-third of the asymptomatic population—and pathological, symptomatic crepitus that warrants meticulous intervention. The evolution of our understanding of this disorder has transitioned from observational descriptions in the 19th century to highly sophisticated, minimally invasive arthroscopic interventions today.
The historical recognition of scapulothoracic crepitus is deeply rooted in classical orthopedic literature, dating back to Boinet’s initial descriptions in 1867. Boinet highlighted the clinical presence of this frictional phenomenon, but it was Mauclaire in 1904 who provided a nuanced classification of the acoustic profile of the disorder. Mauclaire brilliantly categorized the sounds into three distinct presentations: froissement (a subtle, soft tissue rustling), frottement (a more pronounced, grating friction sound), and craquement (a loud, distinct osseous snap or crack). This classification remains clinically relevant today, as it helps the examining surgeon differentiate between purely soft tissue bursitis (often producing froissement) and osseous impingement lesions, such as a prominent tubercle of Luschka or an osteochondroma (which typically manifest as craquement). Milch and Kuhn later refined these distinctions, emphasizing that surgical intervention is strictly reserved for painful crepitus, as painless snapping is considered a normal variant of scapulothoracic gliding.
Epidemiologically, symptomatic snapping scapula syndrome predominantly afflicts a younger, highly active demographic. The typical patient profile includes overhead athletes—such as baseball pitchers, swimmers, and volleyball players—as well as individuals engaged in heavy, repetitive manual labor. The pathophysiology is driven by sustained, repetitive arm elevation, which incites microtrauma and subsequent hypertrophic inflammation within the scapulothoracic bursae. Over time, this inflammatory cascade leads to fibrotic thickening of the bursal tissue, creating a self-perpetuating cycle of friction, localized edema, and mechanical impingement.
On physical examination, the hallmark pathognomonic finding is palpable or audible crepitus localized to the superomedial border or the inferior pole of the scapula. This is frequently accompanied by exquisite localized tenderness. Furthermore, clinicians must be astute in identifying "pseudowinging." Unlike true scapular winging caused by a long thoracic nerve palsy (serratus anterior paralysis) or spinal accessory nerve palsy (trapezius paralysis), pseudowinging is a non-neurologic, compensatory biomechanical alteration. It occurs when the patient subconsciously alters their periscapular muscle firing patterns to unload the painful, inflamed bursa, resulting in a clinically apparent, yet neurologically intact, prominence of the medial scapular border.
Detailed Surgical Anatomy and Biomechanics
Mastery of the scapulothoracic articulation is the absolute prerequisite for safe and effective arthroscopic decompression. Unlike the glenohumeral or tibiofemoral joints, the scapulothoracic interface is not a true diarthrodial synovial joint; rather, it is a complex, fascial gliding mechanism. The scapula articulates with the convex posterior thoracic cage, separated by a highly organized, stratified arrangement of muscular layers and lubricating bursae. The smooth translation of the scapula across the posterior chest wall is entirely dependent on the synchronized firing of periscapular musculature and the unimpeded function of these bursal sacs.
The bursal anatomy of the scapulothoracic joint is traditionally divided into major (anatomically consistent) and minor (adventitial) bursae. The two major bursae are the primary targets of our arthroscopic intervention. The infraserratus bursa is the most clinically significant. It is situated deep to the serratus anterior muscle, directly interfacing with the periosteum of the ribs and the intercostal musculature. Because this bursa endures the most direct compressive and frictional forces during scapular protraction and rotation, it is the most frequent site of pathological inflammation. The supraserratus bursa, while less commonly the primary pain generator, lies in the fascial plane between the deep surface of the subscapularis muscle and the superficial surface of the serratus anterior. Inflammation here can also significantly restrict internal rotation and overhead elevation.
In addition to the major bursae, up to four minor, adventitial bursae can develop in response to abnormal biomechanical stresses. These are not present at birth but form as a reactive physiological response to chronic friction. They are typically located at the superomedial angle of the scapula, the base of the scapular spine, the inferior scapular angle, and occasionally at the trapezoid insertion. The superomedial angle is of particular surgical interest, as it serves as the conjoined insertion site for the levator scapulae and the rhomboid minor. Chronic traction and friction at this junction frequently lead to profound fibrotic thickening, necessitating targeted radiofrequency ablation and, often, bony resection of the underlying scapular angle.
Navigating this anatomy arthroscopically requires a profound respect for the regional neurovascular structures, the injury of which constitutes a catastrophic complication. The dorsal scapular nerve (C5) and its accompanying artery are the most critical structures at risk. This neurovascular bundle descends deep to the levator scapulae and rhomboid muscles, coursing parallel to, and approximately 2 to 3 centimeters medial to, the medial border of the scapula. It provides essential motor innervation to the rhomboids and levator scapulae. Portals placed too far medially or superiorly place this bundle in imminent jeopardy. Furthermore, the dorsal scapular nerve arborizes extensively at its terminal branches, meaning that inferiorly placed portals are generally safer. The spinal accessory nerve (CN XI), innervating the trapezius, and the transverse cervical artery are located more superiorly in the posterior triangle of the neck; while less directly in the arthroscopic field, aggressive superior dissection or misguided trocar insertion can compromise them.
Exhaustive Indications and Contraindications
The decision to proceed with arthroscopic scapulothoracic decompression must be predicated on a rigorous, evidence-based evaluation of the patient's pathology and their response to non-operative modalities. Surgery is never the first-line treatment for snapping scapula syndrome. A comprehensive trial of conservative management, typically lasting a minimum of three to six months, is mandatory. This regimen should include targeted periscapular physiotherapy focusing on serratus anterior and lower trapezius strengthening, postural correction, nonsteroidal anti-inflammatory medications, and diagnostic/therapeutic corticosteroid injections into the affected bursa.
Surgical intervention is indicated only when a patient presents with persistent, debilitating, painful crepitus that has definitively failed this exhaustive non-operative protocol. The presence of a confirmed osseous lesion—such as an osteochondroma, a malunited rib or scapular fracture, or a prominent, pathologically hooked superomedial angle (the tubercle of Luschka)—also serves as a strong indication for arthroscopic or open resection, as these mechanical blocks will not resolve with physical therapy alone. Furthermore, the patient must experience significant, albeit temporary, relief from a fluoroscopically or ultrasound-guided local anesthetic injection into the scapulothoracic bursa, as this confirms the bursa as the primary pain generator.
Contraindications must be meticulously respected to avoid catastrophic outcomes and surgical failures. Absolute contraindications include the presence of active local or systemic infection, and painless crepitus. Operating on painless physiological snapping is a severe clinical error that exposes the patient to unnecessary surgical risk without any potential for functional benefit. True neurological scapular winging—resulting from long thoracic nerve or spinal accessory nerve palsies—is an absolute contraindication for isolated bursectomy, as the underlying pathology is paralytic, not frictional.
Clinical Decision Matrix
| Parameter | Indications for Arthroscopic Decompression | Absolute/Relative Contraindications |
|---|---|---|
| Symptomatology | Severe, localized pain with audible/palpable crepitus | Painless snapping/crepitus (Physiologic) |
| Conservative Therapy | Failure of > 3-6 months of targeted physical therapy | Incomplete or absent trial of conservative care |
| Diagnostic Injection | Significant temporary relief from local anesthetic | No relief from targeted bursal injection |
| Osseous Anatomy | Symptomatic osteochondroma, prominent Luschka tubercle | Normal anatomy with uncorrected cervical radiculopathy |
| Neurological Status | Intact periscapular motor function (Pseudowinging only) | True neurogenic winging (Long Thoracic/Spinal Accessory palsy) |
| Systemic Health | Medically optimized for general anesthesia | Active local/systemic infection, severe medical comorbidities |
Pre-Operative Planning, Templating, and Patient Positioning
Pre-operative planning is the foundation upon which surgical success is built. It begins with a meticulous review of the patient's advanced imaging. While standard anteroposterior and true lateral radiographs of the scapula are standard, tangential scapular views (often referred to as a Y-view or a specifically angled scapular outlet view) are critical for profiling the superomedial angle and identifying an excessively hooked tubercle of Luschka. If any osseous anomaly is suspected, a Computed Tomography (CT) scan with 3-dimensional surface-rendered reconstructions is mandatory. This allows the surgeon to accurately template the dimensions of the bony resection, ensuring that enough bone is removed to eliminate impingement without destabilizing the muscular insertions. Magnetic Resonance Imaging (MRI) is highly sensitive for identifying bursal fluid, fibrotic thickening, and ruling out soft-tissue neoplasms such as elastofibroma dorsi, a benign but space-occupying lesion common in the subscapular region.
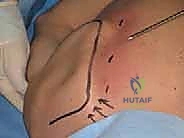
Patient positioning is arguably the most critical non-surgical step of the procedure, as it directly dictates the safety and accessibility of the scapulothoracic interval. The procedure is performed under general endotracheal anesthesia to ensure complete muscle relaxation, which is vital for expanding the working space. The patient is placed in the prone position on a specialized operating table equipped with a chest roll or a Jackson frame to allow free abdominal excursion and prevent elevated intrathoracic pressure. All bony prominences, particularly the face, knees, and ankles, must be meticulously padded to prevent pressure necrosis and neuropraxia.
The defining maneuver for this procedure is the "Chicken Wing" position. The affected arm is carefully brought behind the patient's back, placing the shoulder in a position of extension and maximal internal rotation.
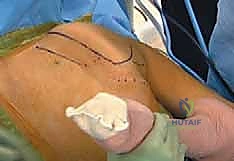
This specific biomechanical positioning forces the scapula into protraction, effectively lifting the medial border and the superomedial angle away from the posterior chest wall. This maneuver expands the potential space of the infraserratus bursa from a virtual cleft into a definable cavity, drastically reducing the risk of iatrogenic pleural puncture during portal placement. The arm is securely draped and held in this position using a sterile padded arm holder or by an assistant. If a bony resection is templated, a C-arm fluoroscope must be positioned to allow unobstructed anteroposterior and tangential views of the scapula, ensuring the sterile field is not compromised during imaging.
Step-by-Step Surgical Approach and Fixation Technique
The arthroscopic masterclass begins with the precise establishment of portals. The margin for error here is minimal; misplaced portals can lead to devastating neurovascular injuries or pneumothorax.
Initial Portal Placement and Bursal Distention
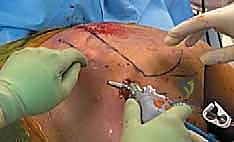
First, the surgeon must palpate and demarcate the medial border of the scapula and the scapular spine with a sterile marking pen. The primary viewing portal is located approximately 2 to 3 centimeters medial to the medial scapular edge, perfectly aligned with the horizontal axis of the scapular spine.
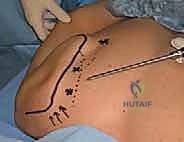
Before any incision is made, the scapulothoracic space must be hydro-distended. An 18-gauge spinal needle is introduced at the marked portal site. Crucially, the trajectory of the needle must be strictly parallel to the contour of the ribs. Directing the needle perpendicularly invites catastrophic penetration of the parietal pleura and lung parenchyma. The needle is advanced until a distinct tactile "pop" or loss of resistance is felt, indicating entry into the infraserratus bursa. Approximately 150 to 200 mL of sterile physiological saline, admixed with epinephrine to induce local vasoconstriction, is injected. This massive distention creates a safe, fluid-filled cavern, pushing the delicate neurovascular structures and the pleura safely away from the surgical instruments.
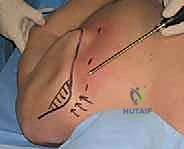
A standard 1-cm stab incision is made, and a 6.0-mm arthroscopic cannula with a blunt obturator is introduced, again maintaining a trajectory strictly parallel to the thorax. A 4.0-mm, 30-degree arthroscope is inserted. The initial view often reveals a cobweb-like network of inflamed, hypertrophic bursal tissue.
Establishing the Working Portal and Bursectomy
With visualization established, a second working portal is created under direct intra-articular visualization. This inferior portal is typically placed 3 to 4 centimeters distal to the primary viewing portal, remaining 2 to 3 centimeters medial to the scapular border to protect the dorsal scapular nerve.
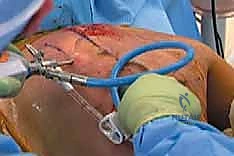
A spinal needle is used to localize the ideal trajectory before incision. Once the second cannula is established, a radiofrequency ablation wand is introduced.
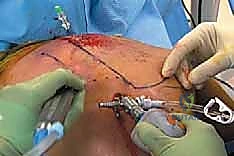
The surgeon begins a systematic, sweeping bursectomy. The goal is to clear the fibrotic debris and inflammatory tissue from the infraserratus space, exposing the glistening fascia of the serratus anterior laterally and the periosteum of the ribs medially.
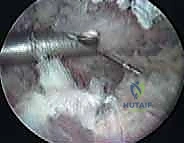
Meticulous hemostasis is maintained throughout using the radiofrequency device. The dissection is carried superiorly toward the superomedial angle, the most common site of impingement.
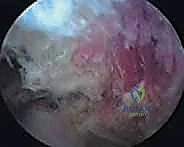
Superomedial Angle Resection (Osteoplasty)
If pre-operative templating or intraoperative findings dictate an osseous resection, the arthroscope is directed toward the superomedial angle. The overlying fibrotic tissue, often a confluence of the inflamed bursa and the degenerated insertions of the levator scapulae, is carefully ablated to expose the bare bone of the scapula.
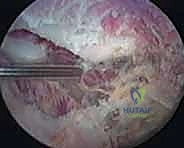
An arthroscopic motorized burr is introduced through the working portal.
The surgeon performs a precise, controlled osteoplasty, resecting the prominent tubercle of Luschka. The resection typically encompasses the superior 2 to 3 centimeters of the medial border and extends laterally along the superior border, effectively blunting the angle.

It is imperative to avoid excessive resection, which could detach the levator scapulae and compromise scapular suspension. Intraoperative fluoroscopy can be utilized at this stage to confirm the adequacy of the resection against the pre-operative template.
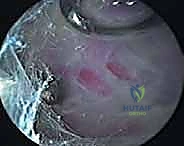
Once the resection is complete, the area is thoroughly irrigated to remove bone debris. The instruments are withdrawn, and the portal sites are closed with simple interrupted non-absorbable sutures.
Complications, Incidence Rates, and Salvage Management
While arthroscopic scapulothoracic decompression is a minimally invasive procedure, the proximity to the thoracic cavity and major neurovascular bundles demands absolute vigilance. Complications, though relatively rare in the hands of an experienced arthroscopist, can be life-threatening or severely functionally compromising.
The most feared complication is iatrogenic pneumothorax. This occurs when the spinal needle, trocar, or arthroscopic instruments penetrate the parietal pleura and enter the thoracic cavity. The incidence is reported to be less than 1% in high-volume centers, but the risk is significantly elevated if the surgeon fails to maintain a trajectory parallel to the rib cage during portal establishment. If a pneumothorax is suspected intraoperatively (evidenced by a sudden drop in oxygen saturation, increased airway pressures, or direct visualization of pleural violation), the procedure must be immediately aborted. A stat intraoperative chest radiograph is obtained, and a thoracostomy tube (chest tube) must be placed by the surgical team or a consulting thoracic surgeon.
Neurovascular injury is another severe complication. The dorsal scapular nerve and artery are at the highest risk. Injury to this nerve results in denervation of the rhomboids and levator scapulae, leading to profound, true scapular winging and severe functional deficit. This is almost entirely preventable by adhering to the "safe zone" for portal placement—strictly maintaining a distance of at least 2 centimeters medial to the medial border of the scapula. If nerve transection occurs, salvage management requires complex microsurgical nerve grafting or regional muscle transfer procedures, which have variable success rates.
Post-operative hematoma can occur due to the highly vascular nature of the periscapular musculature. Meticulous intraoperative hemostasis with radiofrequency ablation and the use of epinephrine in the distention fluid mitigate this risk. Large, symptomatic hematomas may require surgical evacuation, while smaller ones can be managed expectantly. Finally, recurrence of symptoms or incomplete relief is often due to inadequate bony resection of the superomedial angle or failure to address underlying glenohumeral pathology. Salvage management in these cases involves a thorough re-evaluation, advanced imaging, and potentially a revision arthroscopic or open decompression.
Complication Matrix and Management Strategies
| Complication | Estimated Incidence | Mechanism of Injury | Salvage / Management Strategy |
|---|---|---|---|
| Pneumothorax | < 1.0% | Perpendicular trocar/needle trajectory puncturing pleura | Immediate chest tube placement (Thoracostomy), abort procedure |
| Dorsal Scapular Nerve Injury | < 0.5% | Portals placed too close to the medial scapular border | Microsurgical repair, Rhomboid/Pectoralis muscle transfer |
| Post-Operative Hematoma | 2.0 - 5.0% | Inadequate RF ablation, rebound vasodilation | Observation for small bleeds; Surgical evacuation if expanding/compressive |
| Incomplete Resection / Recurrence | 5.0 - 10.0% | Insufficient burring of Luschka tubercle, missed adventitial bursa | Revision arthroscopy, extended bony resection, aggressive post-op PT |
| Infection (Superficial/Deep) | < 1.0% | Standard surgical site contamination | Oral/IV Antibiotics, Arthroscopic irrigation and debridement |
Phased Post-Operative Rehabilitation Protocols
The surgical intervention is merely the first step in the restorative process; a meticulously structured, phased rehabilitation protocol is paramount to achieving an optimal functional outcome. The primary goals of post-operative physiotherapy are to prevent periscapular adhesion formation, restore normal scapulohumeral rhythm, and rebuild the strength of the dynamic scapular stabilizers.
Phase I: Immediate Post-Operative Protection (Weeks 0-2)
During the initial two weeks, the focus is on pain control, wound healing, and preventing severe stiffness. The patient is placed in a standard shoulder sling for comfort, but strict immobilization is discouraged. Patients are instructed to remove the sling multiple times a day to perform gentle, passive range of motion (ROM) exercises, including pendulum exercises and passive forward elevation as tolerated. Active internal rotation and aggressive reaching across the body are restricted to prevent excessive traction on the healing soft tissues at the medial scapular border. Cryotherapy is utilized aggressively to manage localized edema.
Phase II: Active-Assisted and Early Active Motion (Weeks 2-6)
Once the portal incisions have healed and acute pain has subsided, the sling is definitively discontinued. The focus shifts to restoring full, active range of motion. Therapists initiate active-assisted ROM exercises, utilizing pulleys and wand exercises. Crucially, periscapular isometric exercises are introduced to awaken the serratus anterior, rhomboids, and lower trapezius without placing undue sheer stress on the scapulothoracic interface. Postural re-education is heavily emphasized, correcting any compensatory thoracic kyphosis or forward head posture that the patient may have developed pre-operatively.
Phase III: Strengthening and Dynamic Stabilization (Weeks 6-12)
At the six-week mark, assuming full, painless active ROM has been achieved, the protocol transitions into aggressive strengthening. Isotonic strengthening of the entire periscapular musculature is initiated. Exercises targeting the serratus anterior (e.g., "scapular push-ups" or dynamic hugs) and the lower trapezius (e.g., prone Y-raises) are foundational. Closed kinetic chain exercises are particularly beneficial for enhancing proprioception and dynamic stability. The therapist must closely monitor the patient for any return of scapular dyskinesis or pseudowinging, adjusting the load and volume accordingly.
Phase IV: Return to High-Level Function (Weeks 12+)
The final phase is tailored to the patient's specific occupational or athletic demands. This involves plyometric training, sport-specific kinetic chain integration, and endurance conditioning. Overhead athletes will begin a structured, progressive throwing or serving program. Return to full, unrestricted activity is typically permitted between 3 to 5 months post-operatively, contingent upon the patient demonstrating symmetrical periscapular strength, absence of painful crepitus, and flawless scapulohumeral mechanics.
Summary of Landmark Literature and Clinical Guidelines
The evolution of scapulothoracic decompression has been thoroughly documented in the orthopedic literature, marking a distinct paradigm shift from highly morbid open procedures to elegant arthroscopic techniques. Understanding this literature is essential for the academic surgeon.
Early literature, dominated by open procedures, highlighted the significant morbidity associated with massive muscle takedowns. Seminal papers by Harper et al. and Manske et al. detailed the open resection of the superomedial scapular angle. While these procedures were effective in eliminating the mechanical impingement, they were frequently complicated by severe post-operative pain, prolonged rehabilitation times, and permanent weakness of the rhomboids and levator scapulae due to the necessity of detaching and reattaching these critical muscular stabilizers.
The transition to arthroscopic management was pioneered in the late 1990s and early 2000s. A landmark study by Pearse et al. provided a critical comparative analysis between open and arthroscopic techniques. Their findings unequivocally demonstrated that arthroscopic bursectomy and osseous resection yielded equivalent, if not superior, pain relief and functional outcomes compared to open surgery, but with a drastically reduced complication profile, minimal blood loss, and significantly accelerated rehabilitation timelines. This paper was instrumental in establishing arthroscopy as the gold standard for this pathology.
Furthermore, the contemporary understanding of scapular mechanics and the importance of conservative management is heavily indebted to the extensive work of Ben Kibler. Kibler’s classification of scapular dyskinesis and his foundational research on the kinetic chain have deeply influenced clinical guidelines. Current consensus guidelines dictate that surgical intervention must be preceded by a rigorous, Kibler-inspired physical therapy regimen focusing on core stability, postural correction, and targeted serratus/lower trapezius rehabilitation. Surgery is universally recognized in the modern literature not as a primary treatment, but as a definitive salvage procedure for mechanically recalcitrant cases of painful snapping scapula syndrome.
