Proximal Humerus Resection and Endoprosthetic Replacement: An Intraoperative Masterclass
Key Takeaway
Learn the intricate steps of proximal humerus resection and endoprosthetic replacement for musculoskeletal sarcoma. This masterclass covers preoperative planning, comprehensive surgical anatomy, meticulous intraoperative execution, and critical pearls for successful outcomes, guiding fellows through every crucial decision and technique in the operating theater.
Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues and fellows, to the operating theater. Today, we are undertaking one of the most challenging yet profoundly rewarding procedures in orthopedic oncology: a Proximal Humerus Resection with Endoprosthetic Replacement. Historically, high-grade sarcomas of the proximal upper extremity mandated morbid procedures such as forequarter amputations. However, with the advent of neoadjuvant chemoradiation protocols, advanced cross-sectional imaging, and sophisticated modular implant designs, limb-salvage surgery has become the gold standard. Our dual mandate today is uncompromising: achieve widely negative oncologic margins to ensure local disease control, and meticulously reconstruct the osseous and soft-tissue anatomy to restore maximal upper extremity function.
The proximal humerus is the third most common site for primary malignant bone tumors, following the distal femur and proximal tibia. We frequently encounter chondrosarcomas, osteosarcomas, and Ewing sarcomas in this anatomical region. Furthermore, the proximal humerus is a highly predilected site for solitary metastatic lesions, particularly from renal cell, thyroid, breast, lung, and prostate carcinomas. The biological behavior of these tumors dictates our surgical margins. For high-grade primary sarcomas, an extra-articular or intra-articular wide resection with a continuous cuff of normal tissue is imperative. For metastatic lesions or multiple myeloma, a marginal resection may suffice, prioritizing immediate skeletal stability and pain relief over wide oncologic clearance.
Executing this procedure demands a profound, almost intuitive grasp of the complex three-dimensional anatomy of the shoulder girdle. The proximity of the brachial plexus and the axillary vessels leaves zero margin for error. A successful outcome is not merely defined by the survival of the patient, but by the preservation of a functional, painless limb that allows for activities of daily living. This masterclass will dissect every phase of this operation, from the critical preoperative templating to the nuanced soft-tissue reconstruction that prevents the dreaded complication of postoperative instability.
Detailed Surgical Anatomy and Biomechanics
Mastery of the proximal humerus requires an intimate understanding of the internervous planes, the neurovascular bundles, and the dynamic stabilizers of the glenohumeral joint. The shoulder is inherently unstable, relying heavily on the rotator cuff and the deltoid for both static containment and dynamic motion. When we resect the proximal humerus, we inherently disrupt this delicate biomechanical balance.
The Deltopectoral Interval and Superficial Landmarks
Our primary surgical corridor is the deltopectoral interval, a true internervous plane. The anterior deltoid is innervated by the axillary nerve, while the pectoralis major is supplied by the medial and lateral pectoral nerves. The superficial landmark for this interval is the coracoid process superiorly and the deltoid tuberosity distally. The cephalic vein, running precisely within this groove, serves as our anatomical beacon. Venous drainage of the anterior shoulder relies heavily on this structure; therefore, preservation is preferred to mitigate postoperative limb edema, though it may be ligated if tumor extension dictates.
Critical Neurovascular Structures
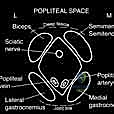
The neurovascular anatomy in this region is unforgiving. The axillary nerve (C5-C6) is the most vulnerable structure during this approach. Originating from the posterior cord of the brachial plexus, it traverses anterior to the subscapularis, dives through the quadrangular space (bounded by the teres minor superiorly, teres major inferiorly, long head of the triceps medially, and the surgical neck of the humerus laterally), and wraps around the posterior humeral neck approximately 5 to 7 centimeters distal to the lateral acromion. Iatrogenic injury to the axillary nerve results in catastrophic deltoid paralysis, rendering any functional reconstruction virtually useless.
Medial to the coracoid process and deep to the pectoralis minor lies the neurovascular bundle containing the brachial plexus and the axillary artery and vein. As the arm is manipulated during tumor resection, these structures are placed under tension. The musculocutaneous nerve, entering the coracobrachialis typically 3 to 8 centimeters distal to the coracoid, is also at risk during medial retraction of the conjoined tendon. Meticulous identification, mobilization, and protection of these structures using blunt retractors is a non-negotiable phase of the dissection.
Biomechanical Considerations in Reconstruction
The native proximal humerus serves as the fulcrum for the rotator cuff and the deltoid. In a tumor resection, we frequently sacrifice portions of the rotator cuff footprint (the greater and lesser tuberosities). If the rotator cuff is resected, an anatomic endoprosthesis will inevitably migrate superiorly, leading to impingement and poor forward elevation. In contemporary practice, if the axillary nerve and deltoid are preserved but the cuff is sacrificed, we often utilize a reverse total shoulder arthroplasty (RTSA) tumor prosthesis. This medializes the center of rotation and distalizes the humerus, tensioning the deltoid to act as the primary elevator of the arm, compensating for the absent rotator cuff.
Exhaustive Indications and Contraindications
The decision to proceed with limb-salvage surgery via proximal humerus resection and endoprosthetic reconstruction is complex and must be made in a multidisciplinary tumor board setting. The primary objective is always patient survival, followed by limb preservation and functional optimization.
Patient selection is paramount. The ideal candidate has a primary bone sarcoma or an isolated metastatic lesion confined to the proximal humerus without neurovascular encasement. The patient must be medically optimized to withstand a prolonged surgical procedure and possess the cognitive and physical capacity to participate in a rigorous, months-long postoperative rehabilitation protocol.
Conversely, limb salvage is absolutely contraindicated if achieving negative oncologic margins necessitates the resection of the major neurovascular bundle (the brachial plexus and axillary vessels), which would result in a flail, insensate, and ischemic limb. In such scenarios, a forequarter amputation remains the only viable oncologic and functional option. Intractable periprosthetic joint infections or massive soft tissue defects that cannot be covered by local or free flaps also present severe relative contraindications.
| Category | Indications | Contraindications |
|---|---|---|
| Absolute | Primary high-grade bone sarcomas (Osteosarcoma, Ewing) localized to proximal humerus. | Encasement of the brachial plexus or axillary artery/vein by the tumor. |
| Absolute | Symptomatic, destructive solitary metastasis with impending or actual pathologic fracture. | Inability to achieve negative oncologic margins without sacrificing the limb's viability. |
| Absolute | Aggressive benign bone tumors (e.g., Giant Cell Tumor) with massive bone destruction. | Active, uncontrolled systemic or local infection (e.g., severe osteomyelitis). |
| Relative | Failed previous osteosynthesis or arthroplasty with massive proximal bone loss. | Severe patient comorbidities precluding prolonged anesthesia or rehabilitation. |
| Relative | Multiple myeloma lesions requiring immediate structural stability for pain control. | Extensive soft tissue contamination from a poorly placed prior biopsy tract. |
Pre-Operative Planning, Templating, and Patient Positioning
Before the scalpel touches the skin, the battle is won or lost in the preoperative planning phase. A meticulous, multi-modal approach to imaging and templating is the cornerstone of a successful oncologic resection and biomechanical reconstruction.
Advanced Imaging and Oncologic Templating
We rely on a triad of imaging modalities. Plain radiographs provide a baseline understanding of bone destruction and deformity. The MRI, however, is our most critical tool. Coronal, sagittal, and axial T1-weighted images without contrast are essential for determining the intramedullary extent of the tumor and defining the "skip" lesions. T2-weighted and STIR images exquisitely delineate the extraosseous soft-tissue mass and its proximity to the neurovascular bundle. We mandate a minimum of a 2 to 3-centimeter macroscopic marrow margin beyond the MRI-defined tumor extent for high-grade sarcomas.
A fine-cut CT scan of the entire humerus is utilized for precise bone templating. We overlay digital templates of our modular endoprosthetic system onto the CT images. This allows us to determine the exact level of our osteotomy, the required length of the diaphyseal replacing segment, and the diameter of the intramedullary stem. We must also account for the biopsy tract, which must be excised en bloc with the tumor specimen. The biopsy must have been performed meticulously, directly in line with our planned deltopectoral incision.
Patient Positioning and Fluoroscopy Setup
For proximal humerus resections, the modified beach chair position is universally preferred. This position offers unparalleled exposure to the anterior shoulder, facilitates gravity-assisted venous drainage, and most importantly, allows for dynamic, unobstructed manipulation of the arm during the trial reduction phase to assess stability and range of motion.
The patient is positioned with the head secured in a specialized headrest, ensuring the cervical spine is neutral and the eyes and ears are rigorously protected. A bump is placed beneath the medial border of the ipsilateral scapula to protract the shoulder girdle, bringing the glenohumeral joint forward. The operative arm is draped completely free, utilizing a sterile extremity drape. We employ a pneumatic arm positioner or a dedicated surgical assistant to manage limb traction and rotation.
The fluoroscopy unit (C-arm) must be positioned at the head of the bed, coming in from the contralateral side. It is imperative to confirm, prior to prepping and draping, that the C-arm can easily obtain true anteroposterior (AP) and axillary lateral views of the shoulder and the entire humeral shaft down to the elbow without compromising the sterile field.
Step-by-Step Surgical Approach and Fixation Technique
The execution of this procedure requires a seamless transition between aggressive oncologic resection and delicate reconstructive microsurgery-like tissue handling.
Incision and Superficial Dissection
We initiate an extended deltopectoral incision. The skin incision begins just lateral to the tip of the coracoid process and extends distally along the deltopectoral groove for 15 to 20 centimeters, tracking toward the deltoid insertion. If a previous biopsy tract is present, we design an elliptical incision to encompass the tract with a 1-2 cm margin of healthy skin, ensuring it remains attached to the underlying tumor specimen.
We dissect through the subcutaneous fat to identify the deltopectoral fascia. The cephalic vein is identified. While it can be retracted laterally with the deltoid, retracting it medially with the pectoralis major often protects it better from the vigorous lateral retraction required during the procedure. We incise the fascia medial to the vein and bluntly develop the interval between the anterior deltoid and the pectoralis major. The clavipectoral fascia, visible deep in the interval, is longitudinally incised lateral to the conjoined tendon.
Deep Dissection and Neurovascular Protection
Exposure of the proximal humerus requires meticulous deep dissection. We identify the insertion of the pectoralis major on the lateral lip of the bicipital groove. Depending on the distal extent of the tumor, we may need to release the upper half or the entirety of the pectoralis major tendon, tagging it with heavy non-absorbable sutures for later repair.
The most critical maneuver is the identification and protection of the axillary nerve. We palpate the inferior border of the subscapularis and trace it laterally to the humerus. Using blunt finger dissection along the anterior surface of the subscapularis, sweeping inferiorly, we identify the axillary nerve as it dives into the quadrangular space. We gently pass a vessel loop around it. Concurrently, we place a broad, blunt retractor (such as a Richardson or Kolbel) medially to retract the conjoined tendon, shielding the brachial plexus and axillary vessels.
Rotator Cuff Release and Capsulotomy
To mobilize the proximal humerus, we must detach the rotator cuff. For an intra-articular resection (where the joint is not contaminated by tumor), we sequentially release the subscapularis from the lesser tuberosity, and the supraspinatus, infraspinatus, and teres minor from the greater tuberosity. These tendons are sharply dissected off the bone, leaving a small cuff of tissue on the specimen if margins allow, and tagged with heavy FiberWire sutures. The long head of the biceps is typically tenodesed to the pectoralis major or coracoid, and its intra-articular portion is resected. A circumferential capsulotomy is then performed, dislocating the humeral head anteriorly.
Oncologic Resection and Osteotomy
With the proximal humerus fully mobilized and the neurovascular structures protected, we proceed to the osteotomy. Using a sterile caliper and a ruler, we measure the exact distance from the articular surface of the humeral head to our pre-planned osteotomy site, cross-referencing our MRI and CT templates.
We mark the bone with electrocautery. Malleable retractors are placed circumferentially around the humeral shaft at the level of the cut to protect the radial nerve posteriorly and the neurovascular bundle medially. Using a high-speed oscillating saw with continuous cold saline irrigation to prevent thermal necrosis of the remaining bone, we perform a transverse osteotomy. The entire proximal humerus and tumor specimen is delivered en bloc from the surgical field. The marrow from the remaining humeral shaft is immediately sampled and sent for frozen section analysis to definitively confirm a negative osseous margin.
Reconstruction and Endoprosthetic Implantation
Once the pathologist confirms negative margins, we shift to reconstruction. The medullary canal of the distal humerus is sequentially reamed using flexible or rigid reamers under fluoroscopic guidance to accommodate the prosthetic stem. We aim for a line-to-line fit if using a cementless, porous-coated stem, or a 2 mm oversized ream if utilizing polymethylmethacrylate (PMMA) bone cement.

A trial reduction is paramount. We assemble the trial stem, the intercalary replacing segment (matching our resected bone length), and the trial humeral head. We reduce the prosthesis and rigorously assess soft tissue tension, deltoid contour, and limb length. The arm is taken through a full range of motion to ensure there is no anterior or posterior instability.
For definitive implantation, if cementing, the canal is thoroughly irrigated with pulsatile lavage and dried with epinephrine-soaked sponges. A cement restrictor is placed distally. PMMA is introduced in a retrograde fashion using a cement gun, and the definitive stem is inserted with precise rotational alignment (typically 20 to 30 degrees of retroversion relative to the epicondylar axis).
Soft Tissue Reconstruction and Closure
The longevity of the reconstruction depends entirely on the soft tissue envelope. We frequently utilize an attachment tube (e.g., a Trevira tube) or a synthetic mesh sutured over the prosthesis. The tagged rotator cuff tendons are meticulously sutured to this mesh or to the specific suture holes on the prosthesis using heavy non-absorbable sutures in a grasping mattress fashion. This provides dynamic stability.
If a reverse total shoulder tumor prosthesis is used, the subscapularis is repaired if possible, but the primary stabilizer is the tensioned deltoid. We ensure meticulous hemostasis, place two large-bore closed suction drains (one deep near the neurovascular bundle, one superficial to the prosthesis), and perform a layered closure. The deltopectoral interval is loosely approximated, the subcutaneous tissues are closed with absorbable sutures, and the skin is closed with surgical staples or a subcuticular stitch.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, endoprosthetic reconstruction of the proximal humerus carries a significant complication profile. The Henderson classification is widely used to categorize failure modes in limb salvage surgery: Type I (Soft Tissue Failure), Type II (Aseptic Loosening), Type III (Structural Failure), Type IV (Infection), and Type V (Tumor Progression).
Soft tissue failure, specifically glenohumeral instability and dislocation, is the most common mechanical complication, particularly when the rotator cuff is heavily resected. Aseptic loosening occurs due to stress shielding and the massive biomechanical forces exerted on the relatively short intramedullary stem. Infection is a devastating complication, amplified by the immunocompromised state of oncology patients undergoing neoadjuvant or adjuvant chemotherapy.
| Complication Type (Henderson) | Estimated Incidence | Pathophysiology & Risk Factors | Salvage & Management Strategy |
|---|---|---|---|
| Type I: Instability / Dislocation | 10% - 25% | Loss of rotator cuff, inadequate soft tissue tensioning, axillary nerve palsy. | Closed reduction and immobilization. If recurrent, revision surgery to increase soft tissue tension, use of constrained liners, or conversion to a Reverse TSA tumor prosthesis. |
| Type II: Aseptic Loosening | 5% - 15% | Stress shielding, micromotion at the bone-cement interface, inadequate initial fixation. | Revision arthroplasty with a longer, thicker stem. Use of uncemented, porous-coated stems with extra-cortical bone bridging techniques. |
| Type III: Structural Failure | 2% - 5% | Fatigue fracture of the prosthetic stem or the intercalary segment due to repetitive loading. | Extraction of broken components. Revision with a larger diameter stem or a custom-designed mega-prosthesis. |
| Type IV: Deep Periprosthetic Infection | 5% - 12% | Immunosuppression (chemotherapy/radiation), prolonged operative time, extensive dead space. | Aggressive surgical debridement, implant retention (DAIR) if acute. Two-stage revision with antibiotic cement spacer for chronic infections. Suppressive antibiotics. |
| Type V: Local Tumor Recurrence | 3% - 10% | Inadequate surgical margins, highly aggressive tumor biology, tumor spillage intraoperatively. | Restaging. Often requires radical resection, potentially forequarter amputation, and adjuvant oncologic therapies. |
Phased Post-Operative Rehabilitation Protocols
Rehabilitation following a proximal humerus tumor resection is a delicate balancing act. We must protect the precarious soft-tissue repairs and the bone-implant interface while preventing devastating adhesive capsulitis. The protocol is highly individualized, dictated by the extent of the rotator cuff and deltoid resection.
Phase I: Maximum Protection (Weeks 0 to 6)
Immediately postoperatively, the arm is placed in a specialized shoulder immobilizer or an abduction sling, depending on the tension of the soft tissue repair. The primary goal in this phase is wound healing and protection of the reconstruction.
* Strict immobilization is maintained for the first 4 weeks, except for hygiene.
* Passive range of motion (PROM) for the elbow, wrist, and hand is initiated immediately to prevent distal contractures.
* Gentle pendulum exercises may begin at week 3 or 4, strictly under the supervision of a specialized physical therapist, provided the soft tissue repair is deemed stable by the operating surgeon. Active shoulder motion is strictly prohibited.
Phase II: Active-Assisted Motion (Weeks 6 to 12)
As the soft tissues begin to heal to the prosthesis or mesh, we gradually introduce motion.
* The sling is progressively weaned during the day.
* Active-assisted range of motion (AAROM) is initiated. This includes supine wand exercises, pulley systems, and wall walks.
* Submaximal, pain-free isometric exercises for the deltoid and remaining rotator cuff are introduced.
* We must manage patient expectations during this phase; forward elevation is often limited, and the focus is on achieving functional motion for eating and hygiene (typically 60 to 90 degrees of abduction and flexion).
Phase III: Strengthening and Functional Integration (Weeks 12 and Beyond)
Once radiographic evidence of stable fixation is confirmed and soft tissue healing is robust, we transition to strengthening.
* Active range of motion (AROM) is aggressively pursued.
* Progressive resistive exercises utilizing elastic bands and light weights are incorporated, focusing on the deltoid and periscapular stabilizers (trapezius, serratus anterior, rhomboids).
* Functional training mimics activities of daily living. Patients with anatomic endoprostheses and resected cuffs will rely heavily on compensatory scapulothoracic motion. Patients with reverse tumor prostheses will focus intensely on deltoid recruitment.
* Heavy lifting (greater than 10-15 lbs) and high-impact activities are permanently restricted to prevent premature aseptic loosening and structural failure.
Summary of Landmark Literature and Clinical Guidelines
The evolution of proximal humerus limb salvage is heavily documented in orthopedic oncology literature. A foundational understanding of these landmark studies is essential for any reconstructive surgeon.
The Musculoskeletal Tumor Society (MSTS) scoring system, developed by Enneking et al., remains the gold standard for evaluating functional outcomes following limb salvage. Studies utilizing the MSTS score consistently demonstrate that while proximal humerus replacements yield lower scores than distal femur replacements (primarily due to the loss of active forward elevation), the overall patient satisfaction and pain relief are exceptionally high, far surpassing amputation cohorts.
Henderson et al. (2011) provided the definitive classification system for failure in limb-salvage surgery, which we discussed in the complications section. This classification shifted the paradigm from merely reporting "failure" to standardizing the etiology, thereby driving innovations in implant design (such as HA-coated collars for extra-cortical bone ingrowth to combat Type II failures).
More recently, the shift towards Reverse Total Shoulder Arthroplasty (RTSA) principles in tumor prostheses has revolutionized outcomes for patients requiring massive rotator cuff resection. Studies by Ruggieri et al. and De Wilde et al. have demonstrated that utilizing a reverse construct in the oncologic setting significantly improves active forward elevation and drastically reduces the incidence of Type I (instability) failures compared to traditional anatomic hemiarthroplasty mega-prostheses. Current clinical guidelines strongly advocate for the use of reverse tumor prostheses whenever the axillary nerve is intact but the rotator cuff footprint is sacrificed to achieve oncologic margins.
