Proximal Humerus Resection and Endoprosthetic Reconstruction: An Intraoperative Masterclass
Key Takeaway
This masterclass details proximal humerus resection and endoprosthetic reconstruction for shoulder girdle sarcomas. We cover critical anatomy, meticulous surgical steps, neurovascular protection, and advanced soft tissue techniques. Gain insights into preoperative planning, intraoperative execution, and comprehensive postoperative care, ensuring optimal outcomes and limb preservation for complex tumor cases.
Comprehensive Introduction and Patho-Epidemiology
The proximal humerus represents a frequent and highly challenging anatomical battleground in orthopedic oncology. It is the third most common site for primary malignant bone tumors, trailing only the distal femur and proximal tibia. Primary bone sarcomas such as osteosarcoma, chondrosarcoma, and Ewing sarcoma frequently present in this region, alongside aggressive benign lesions like giant cell tumors (GCT) and a myriad of metastatic carcinomas, most notably from renal cell, breast, thyroid, and lung primaries. Historically, the standard of care for high-grade sarcomas of the shoulder girdle was a forequarter amputation—a profoundly morbid and psychologically devastating procedure. However, the advent of sophisticated neoadjuvant chemotherapy protocols, advanced cross-sectional imaging, and modular endoprosthetic technology has revolutionized our approach. Today, limb-sparing surgery is achievable in approximately 95% of patients, representing a monumental triumph of modern multidisciplinary oncologic care.
The fundamental objective of a proximal humerus resection and endoprosthetic reconstruction is dual-fold: achieving a wide, oncologically sound margin to prevent local recurrence, and restoring a functional, stable, and pain-free upper extremity. Unlike the lower extremity, where weight-bearing and absolute stability are paramount, the upper extremity's primary purpose is to position the hand in space. Therefore, while active shoulder abduction and forward flexion are frequently compromised due to the necessary sacrifice of the deltoid and rotator cuff, preserving the intricate neurovascular supply to the elbow, wrist, and hand remains the absolute priority. The resulting "suspension shoulder" allows for excellent distal function, which vastly outperforms the functional capacity of any prosthetic limb following an amputation.
Understanding the patho-epidemiology of these tumors is critical for surgical planning. Osteosarcomas typically arise in the metaphysis of adolescents and young adults, frequently breaching the cortex to form massive extraosseous soft tissue components. These stage IIB lesions displace, rather than invade, the neurovascular bundle in their early stages, making meticulous dissection possible. Conversely, chondrosarcomas often present in older adults, frequently arising from the diaphysis or metaphysis, and may exhibit a more insidious, lobulated growth pattern that can subtly encase vital structures. Metastatic lesions, particularly renal cell carcinoma, are notoriously hypervascular and can mimic primary sarcomas in their aggressive local destruction, necessitating similar wide-excision principles.
The evolution of endoprosthetic reconstruction has provided surgeons with highly adaptable, modular systems capable of addressing massive segmental defects. These implants restore skeletal length and provide a stable foundation for soft tissue reattachment. Modern techniques utilize synthetic meshes (such as Trevira tubes) or allograft composites to facilitate the complex reattachment of the remaining shoulder girdle musculature to the prosthesis, enhancing stability and preventing the dreaded complication of inferior subluxation. This intraoperative masterclass will meticulously detail the anatomical, strategic, and technical nuances required to execute this formidable procedure successfully.
Detailed Surgical Anatomy and Biomechanics
Before a scalpel ever touches the skin, a profound, three-dimensional understanding of the shoulder girdle's surgical anatomy is mandatory. The proximal humerus is intimately surrounded by a complex network of musculature, a delicate joint capsule, and a highly unforgiving neurovascular bundle. Navigating this terrain requires absolute precision and a deep respect for the fascial planes that separate tumor from vital structures.
Musculature and Joint Involvement
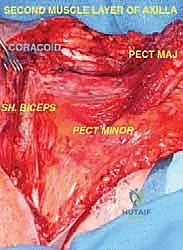
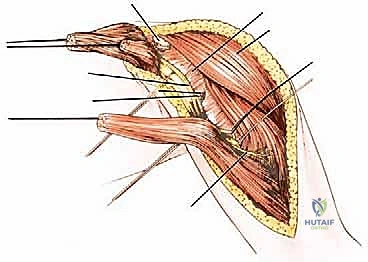
Malignant tumors of the proximal humerus frequently present with substantial extraosseous soft tissue components, extending deep to the deltoid and anteriorly toward the pectoral fascia. These masses commonly displace the subscapularis and coracobrachialis muscles medially. The rotator cuff—comprising the supraspinatus, infraspinatus, teres minor, and subscapularis—is intimately attached to the greater and lesser tuberosities. Early involvement of these pericapsular tissues is common. In most wide resections for high-grade sarcomas, the insertions of the rotator cuff must be sacrificed to achieve adequate oncologic margins, profoundly altering the biomechanics of the reconstructed shoulder.
The glenohumeral joint itself is highly vulnerable to tumor invasion. Tumor spread into or around the joint can occur via four primary mechanisms, which must be systematically evaluated on preoperative MRI. First, direct capsular extension occurs when the tumor breaches the thin joint capsule. Second, the long head of the biceps tendon, which originates from the supraglenoid tubercle and courses through the bicipital groove, acts as a direct intra-articular conduit for tumor extension. Third, a pathologic fracture can create a massive fracture hematoma, seeding tumor cells throughout the joint space and surrounding fascial compartments. Finally, an improperly placed biopsy tract can contaminate the joint or vital neurovascular structures, converting a limb-sparing candidate into an amputation case.
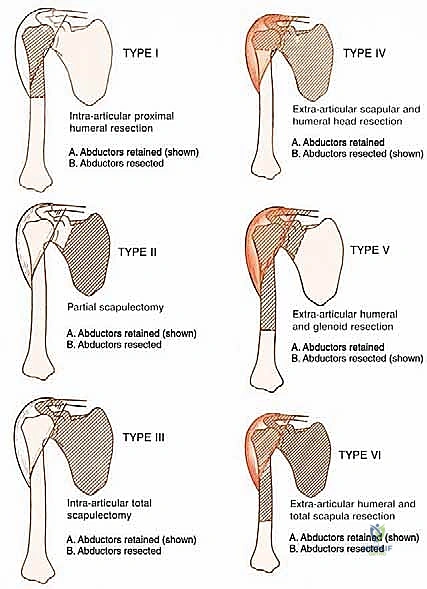
Because of these risks, for high-grade tumors, we generally do not recommend a Type I (intra-articular) resection. The risk of microscopic joint contamination and subsequent local recurrence is simply too high. Instead, extra-articular resections (Type V), which involve resecting the proximal humerus en bloc with the entire glenohumeral joint capsule and often the glenoid articular surface, are the preferred oncologic standard for aggressive lesions.
Neurovascular Structures: Our Guiding Lights
The neurovascular bundle is the absolute limiting factor in limb-sparing surgery of the upper extremity. The surgeon must flawlessly identify, isolate, and protect these structures, often dissecting them directly off the pseudocapsule of the tumor mass.
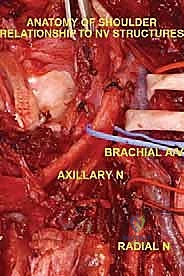
The Axillary Nerve (C5, C6) is of paramount concern. Arising from the posterior cord of the brachial plexus, it courses inferior to the subscapularis, passing through the quadrangular space accompanied by the posterior circumflex humeral artery. It then winds posteriorly around the surgical neck of the humerus, directly deep to the deltoid muscle. Because tumors of the proximal humerus invariably expand into this region, the axillary nerve is at extreme risk. In radical extra-articular resections, the axillary nerve and the deltoid muscle it innervates are frequently sacrificed to ensure a wide margin.
The Musculocutaneous Nerve (C5, C6, C7) originates from the lateral cord and is the first nerve to pierce the coracobrachialis muscle, typically 5 to 8 centimeters distal to the coracoid process. It then descends between the biceps brachii and brachialis muscles. Its anatomical path is highly variable and can be distorted by medial tumor expansion. Early identification of the musculocutaneous nerve is critical; inadvertent transection results in catastrophic loss of elbow flexion and forearm supination, severely compromising the functional outcome of the limb salvage.
The Radial Nerve (C5-T1), a continuation of the posterior cord, courses anterior to the latissimus dorsi and teres major before entering the posterior compartment of the arm via the triangular interval. It runs in the spiral groove of the posterior humerus. While proximal tumors may not directly involve the radial nerve, it becomes highly vulnerable during the distal humeral osteotomy. The nerve must be definitively isolated and retracted prior to applying the oscillating saw to the humeral shaft.
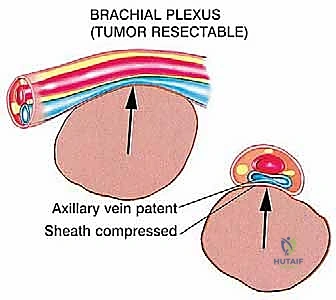
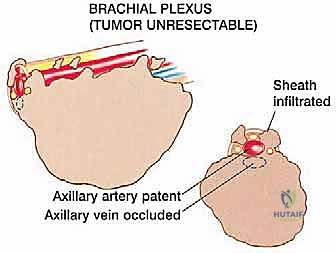
The Axillary and Brachial Arteries and Veins form the central axis of our dissection. The axillary artery, surrounded by the cords of the brachial plexus, is often compressed and displaced anteriorly or medially by large tumors. A critical operative maneuver is the early identification and ligation of the anterior and posterior circumflex humeral vessels. These vessels act as a tether, binding the main neurovascular bundle to the proximal humerus. Once ligated and divided, the entire axillary artery, vein, and brachial plexus can be mobilized and allowed to fall away from the tumor mass, dramatically opening the surgical field and facilitating a safer, more aggressive resection.
Exhaustive Indications and Contraindications
The decision to proceed with a proximal humerus resection and endoprosthetic reconstruction is complex and must be made within the context of a multidisciplinary tumor board. The primary indication is a high-grade primary bone sarcoma (e.g., osteosarcoma, Ewing sarcoma, high-grade chondrosarcoma) localized to the proximal humerus without neurovascular encasement. Additionally, aggressive benign tumors with massive bone destruction, such as Campanacci Grade III giant cell tumors where joint preservation is impossible, are excellent indications. Finally, patients with solitary, destructive metastatic lesions (e.g., renal cell carcinoma) who have a reasonable life expectancy and intractable pain or impending pathologic fracture are also prime candidates for this durable reconstruction.
However, careful patient selection is paramount. We must rigorously distinguish between tumors that merely displace the neurovascular bundle and those that frankly invade it. Induction chemotherapy is often utilized for osteosarcoma and Ewing sarcoma; a poor histologic response or tumor progression during neoadjuvant therapy may force a re-evaluation of limb salvage feasibility. Pathologic fractures were historically considered an absolute contraindication to limb salvage due to hematoma contamination. Today, if the fracture occurs in a patient highly responsive to chemotherapy and the hematoma is contained, limb salvage remains an option, though it requires a wider, more radical resection of the contaminated soft tissue compartments.
Absolute contraindications are non-negotiable and must be identified preoperatively to prevent disastrous intraoperative complications or guaranteed local recurrence. Encasement of the major neurovascular bundle (axillary artery, vein, or brachial plexus cords) by the tumor mandates an amputation. Similarly, massive, uncontrolled invasion into the adjacent chest wall that precludes a negative margin resection is an absolute contraindication.
Relative contraindications require nuanced clinical judgment. Borderline chest wall extension may be manageable with concurrent rib resections by a thoracic surgeon, but increases morbidity. Tumor contamination from a poorly placed, transverse biopsy incision may necessitate the excision of massive amounts of skin and muscle, potentially compromising soft tissue coverage of the prosthesis. Active or recent deep infection in the surgical field is a strong relative contraindication, as placing a massive metallic endoprosthesis into an infected bed guarantees catastrophic failure.
| Category | Specific Clinical Scenarios | Management / Impact on Surgery |
|---|---|---|
| Primary Indications | High-grade osteosarcoma, Ewing sarcoma, dedifferentiated chondrosarcoma. | Standard en bloc extra-articular or wide intra-articular resection with endoprosthesis. |
| Secondary Indications | Aggressive Giant Cell Tumor (Stage III), solitary destructive metastasis (RCC, Thyroid). | Resection to negative margins; endoprosthesis provides immediate stability and pain relief. |
| Absolute Contraindications | Encasement of the axillary artery/vein or brachial plexus cords. | Limb salvage impossible; proceed to forequarter amputation. |
| Absolute Contraindications | Massive, un-resectable chest wall invasion. | Palliative care or amputation if necessary for pain/fungation control. |
| Relative Contraindications | Pathologic fracture with massive, uncontained hematoma. | Requires radical extra-articular resection; high risk of local recurrence. |
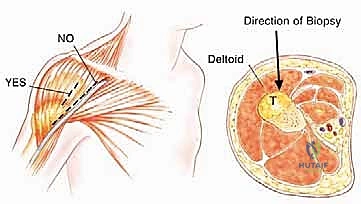
| Relative Contraindications | Poorly placed prior biopsy tract (e.g., posterior approach, transverse incision). | Tract must be excised en bloc; may require complex flap coverage (e.g., latissimus dorsi flap) if closure is compromised. |
Pre-Operative Planning, Templating, and Patient Positioning
Flawless execution in the operating theater is entirely dependent on meticulous preoperative planning. This begins with a comprehensive suite of advanced imaging modalities to define the tumor's intraosseous and extraosseous extent, map its relationship to vital structures, and precisely plan the level of the humeral osteotomy.
Advanced Imaging Modalities
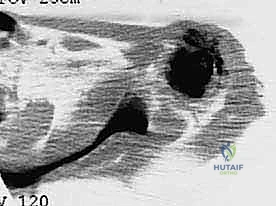
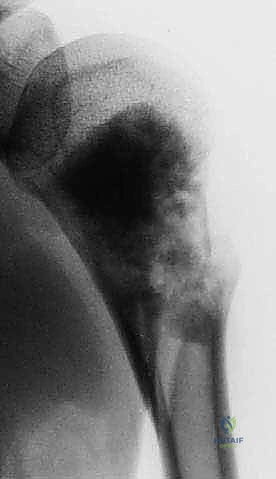
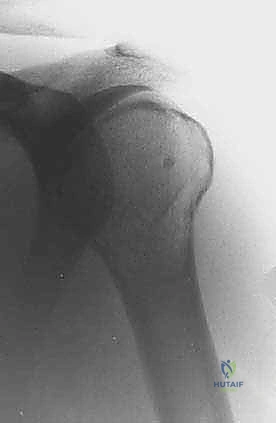
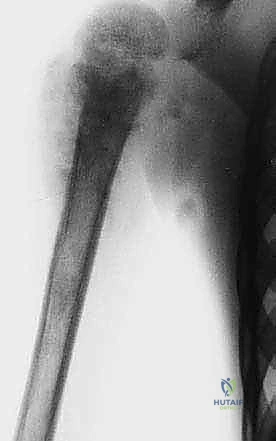
Plain radiography remains the initial step, providing a macroscopic view of bone destruction, periosteal reaction, and the overall mechanical integrity of the humerus. However, Magnetic Resonance Imaging (MRI) of the entire humerus is the gold standard for oncologic staging. T1-weighted coronal and sagittal images are critical for identifying the exact intramedullary extent of the tumor (skip lesions) and determining the level of the osteotomy, which must be planned at least 3 to 5 centimeters distal to the furthest microscopic extent of the disease. T2-weighted axial images are indispensable for evaluating the extraosseous soft tissue mass, specifically its proximity to the neurovascular bundle, joint capsule, and deltoid muscle.
Computed Tomography (CT) scans of the chest are mandatory to rule out pulmonary metastases. Additionally, a high-resolution CT of the shoulder girdle provides superior detail of cortical breakthrough and can help assess borderline chest wall or glenoid involvement. In cases where the tumor is massive or heavily displaces the neurovascular bundle, preoperative CT angiography or conventional angiography is highly recommended. This confirms the patency of the axillary vessels and maps out any aberrant anatomy or major feeding vessels that will require early ligation.
Digital templating is performed using the MRI and plain radiographs. The surgeon must calculate the exact length of the bone to be resected and select the appropriate modular endoprosthetic components. Because sarcomas of the proximal humerus typically involve one-third to one-half of the bone's length, modular systems with varying intercalary segment lengths are essential. The diameter of the distal humeral canal must also be templated to ensure the correct stem size is available, keeping in mind that the distal humerus has a distinct anterior bow that must be accommodated by the prosthetic stem.
Patient Positioning and Operating Room Setup
Proper patient positioning is critical for optimal exposure and ease of intraoperative fluoroscopy. The patient is placed in a modified beach-chair position, with the backrest elevated to approximately 30 to 45 degrees. A beanbag or specialized shoulder positioner is used to stabilize the torso. The head is securely taped and turned slightly to the contralateral side to avoid brachial plexus traction.
The entire forequarter, from the base of the neck to the sternal notch medially, and down to the fingertips, must be prepped and draped free. This allows for full, unencumbered manipulation of the arm, which is essential for dislocating the shoulder, performing the osteotomy, and assessing the final soft tissue tension of the reconstruction. A sterile tourniquet is not used. The C-arm fluoroscopy unit should be positioned on the contralateral side of the table, draped, and ready to swing in to confirm the osteotomy level and verify the final seating of the prosthetic stem.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution of a proximal humerus resection is a masterclass in anatomical dissection, oncologic discipline, and biomechanical reconstruction. The procedure demands a seamless transition from aggressive tumor extirpation to meticulous orthopedic reconstruction.

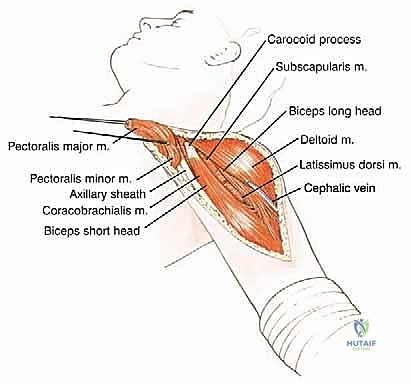
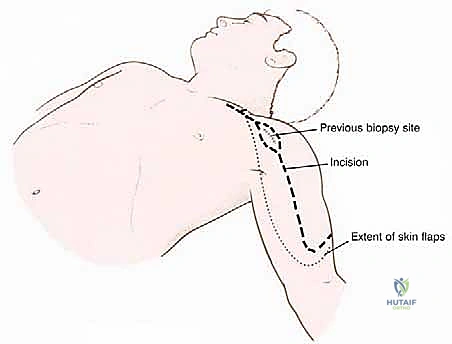
Incision and Superficial Dissection
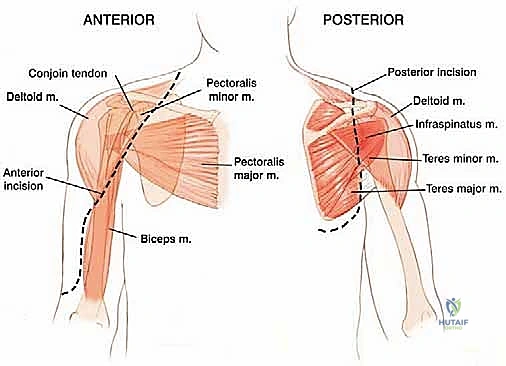
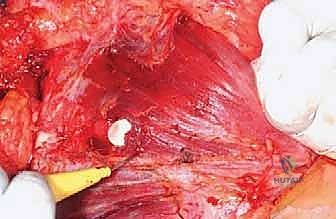
The procedure utilizes an extended deltopectoral approach. The incision begins at the lateral third of the clavicle, passes over the coracoid process, and extends distally along the deltopectoral groove, following the anterolateral border of the humerus as far distal as necessary to achieve the planned osteotomy level. If a previous biopsy was performed, the entire biopsy tract, including the skin ellipse and underlying soft tissue, must be excised en bloc with the main tumor specimen to prevent seeding.
The cephalic vein is identified within the deltopectoral interval. Depending on the tumor's location, the vein is either retracted medially with the pectoralis major or laterally with the deltoid; often, it is ligated to facilitate wider exposure. The deltoid is retracted laterally, and the pectoralis major is retracted medially. If the tumor closely approximates the deltoid, a cuff of the anterior deltoid muscle must be left attached to the tumor specimen to ensure a wide margin.
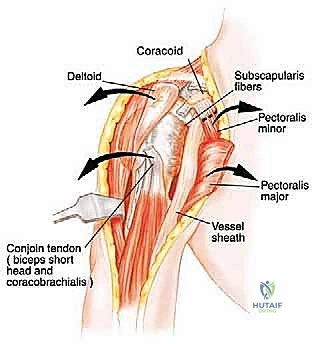
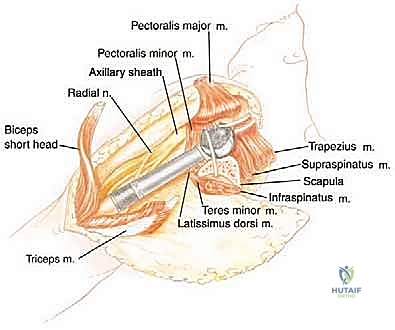
Neurovascular Isolation and Protection
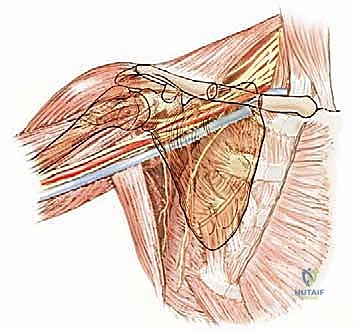
The conjoined tendon (coracobrachialis and short head of the biceps) is identified. To maximize exposure of the neurovascular bundle, the coracoid process can be osteotomized and reflected distally along with the conjoined tendon. The clavipectoral fascia is incised, revealing the axillary artery, vein, and the cords of the brachial plexus.
At this juncture, the most critical maneuver of the dissection is performed: the identification and ligation of the anterior and posterior circumflex humeral arteries and veins. These vessels tether the main neurovascular bundle to the surgical neck of the humerus. Once ligated and divided, the entire neurovascular bundle can be mobilized medially and posteriorly, completely freeing it from the tumor mass. The musculocutaneous nerve must be visually identified entering the coracobrachialis and protected throughout this retraction.
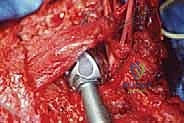
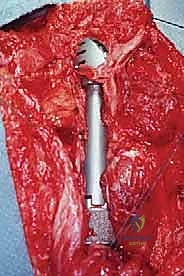
Osteotomy and Tumor Resection
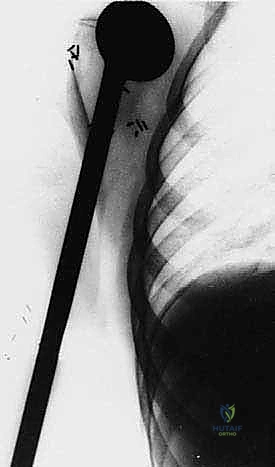
Attention is then turned to the distal aspect of the dissection. The radial nerve is identified in the spiral groove, mobilized, and protected with a vessel loop. Using preoperative MRI measurements, the exact level of the humeral osteotomy is marked on the bone. A margin of at least 3 cm (preferably 5 cm) from the most distal extent of the marrow abnormality is required. Intraoperative fluoroscopy confirms the level.
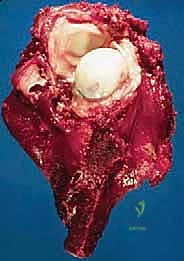
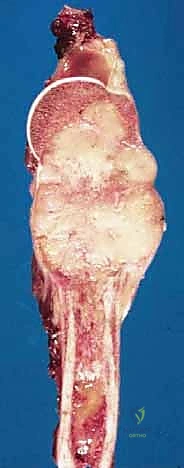
The humerus is transected using an oscillating saw. A sample of the distal marrow is immediately sent for frozen section analysis to definitively confirm a negative bone margin. Once the humerus is divided, the distal end of the tumor specimen is grasped, and the bone is elevated.
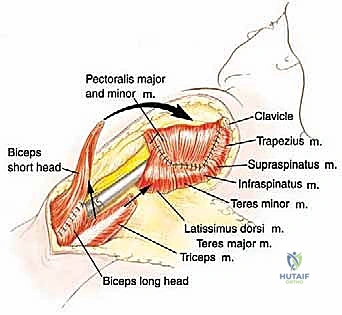
For a standard extra-articular resection (Type V), the dissection proceeds proximally outside the joint capsule. The insertions of the latissimus dorsi, teres major, and pectoralis major are transected, leaving an adequate cuff of tissue on the tumor. The rotator cuff muscles (supraspinatus, infraspinatus, teres minor, subscapularis) are transected at their musculotendinous junctions, far from the capsule. The entire proximal humerus, enveloped in its capsule, tumor pseudocapsule, and surrounding muscle cuffs, is removed en bloc.
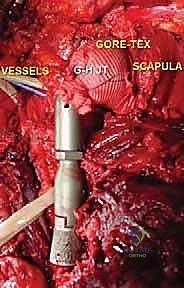

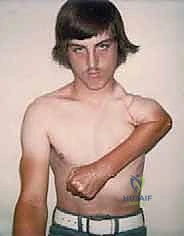
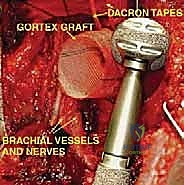
Endoprosthetic Reconstruction and Soft Tissue Balancing
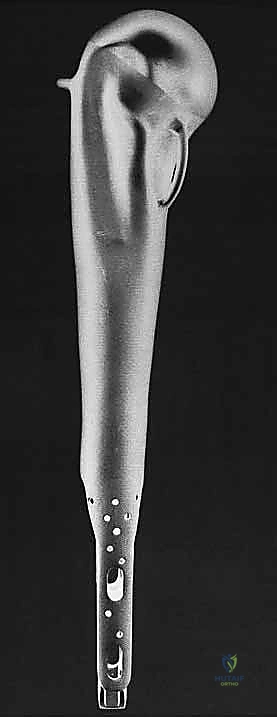
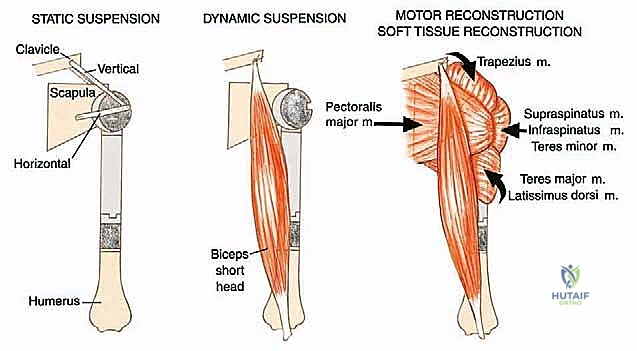
With the tumor removed, the defect is measured to select the appropriate modular components. The distal humeral canal is sequentially reamed and broached. A trial reduction is performed. It is crucial to restore the correct length to maintain resting tension in the remaining myofascial sleeves, preventing inferior subluxation. The prosthetic humeral head must be set in approximately 20 to 30 degrees of retroversion relative to the epicondylar axis of the distal humerus to ensure stability.
Once trialed successfully, the canal is thoroughly lavaged and dried. A cement restrictor is placed, and polymethylmethacrylate (PMMA) bone cement is injected in a retrograde fashion using a cement gun. The definitive stem is inserted in the precise predetermined version.
The final, and arguably most challenging, phase is the soft tissue reconstruction. A bare metallic implant provides no substrate for muscle attachment. Therefore, the prosthesis is typically wrapped in a synthetic mesh (e.g., Trevira tube) or a structural allograft. The remaining tendons of the rotator cuff, pectoralis major, and latissimus dorsi are heavily sutured into this mesh using non-absorbable sutures. This static suspension is vital. If the coracoid was osteotomized, it is repaired with a screw. The wound is closed over closed-suction drains in multiple layers, ensuring absolute hemostasis to prevent hemat
