Mastering Revision Total Hip Arthroplasty: A Comprehensive Intraoperative Guide to Well-Fixed Component Removal

Key Takeaway
Step into the OR for a masterclass on revision total hip arthroplasty. This guide covers meticulous preoperative planning, comprehensive surgical anatomy, and granular, real-time intraoperative execution for removing well-fixed components. We'll navigate complex dissections, extended trochanteric osteotomies, and acetabular explantation with precision, emphasizing bone preservation and complication avoidance. Prepare for an exhaustive breakdown of techniques, pearls, and postoperative management.
Comprehensive Introduction and Patho-Epidemiology
Welcome to the operating theater. Today, we are undertaking one of the most demanding, yet intellectually and technically rewarding procedures in our specialty: Revision Total Hip Arthroplasty (THA) requiring the removal of well-fixed components. This clinical scenario represents a unique paradox in adult reconstruction. We are faced with implants that have successfully achieved their primary biomechanical goal—rigid biologic osseointegration or cement mantle stability—but must nonetheless be extirpated due to extrinsic or intrinsic failures. The primary objective in these cases is not merely the removal of the hardware, but the meticulous preservation of the remaining host bone stock, the protection of encased neurovascular structures, and the optimization of the biologic bed for a durable, long-lasting subsequent reconstruction.
The epidemiology of revision THA is characterized by a steadily increasing burden, driven by the exponential rise in primary hip arthroplasties performed globally, particularly in younger, more active patient demographics. As primary THA survivorship extends into the second and third decades, the prevalence of late-term complications inevitably rises. The patho-epidemiology underlying the need to remove a well-fixed implant is multifaceted. The most insidious culprit remains particle-induced osteolysis. Particulate debris, historically generated by ultra-high-molecular-weight polyethylene (UHMWPE) wear, but also arising from modular junctions (trunnionosis) or metal-on-metal articulations, provokes a robust macrophage-mediated inflammatory cascade. This cellular activation leads to the release of osteolytic cytokines (such as TNF-alpha, IL-1, and IL-6), culminating in aggressive periprosthetic bone resorption.
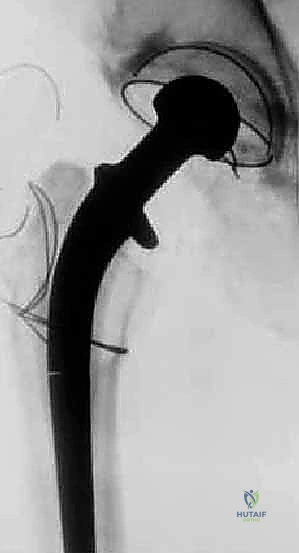
Crucially, profound osteolysis can occur even while the macro-structure of the implant remains rigidly fixed to the host bone. In such instances, the progressive loss of structural bone stock threatens impending catastrophic periprosthetic fracture or massive component subsidence. Furthermore, the advent of highly cross-linked polyethylene has significantly reduced wear rates, yet we now increasingly encounter adverse local tissue reactions (ALTR) and pseudotumor formation secondary to fretting and corrosion at modular taper junctions. These scenarios, alongside deep periprosthetic joint infection (PJI) and recurrent instability, mandate the removal of implants that stubbornly resist extraction. Mastering the techniques required to safely disengage these components without causing iatrogenic devastation is the hallmark of an advanced revision arthroplasty surgeon.
Detailed Surgical Anatomy and Biomechanics
Navigating the perilous landscape of a revision THA requires an encyclopedic knowledge of pelvic and proximal femoral anatomy, which is invariably distorted by prior surgical trauma, heterotopic ossification, and dense fibrotic scar tissue. The sciatic nerve remains the most critical and vulnerable structure during posterior and extensile approaches. It is paramount to recognize its three zones of heightened vulnerability. First, as it exits the greater sciatic notch deep to the piriformis, it can be compromised by aggressive posterior retractor placement or vigorous manipulation of the posterior acetabular column. Second, as it courses over the ischium, inferior and posterior to the acetabulum, it is at risk during the excision of inferior capsular scar or the placement of inferior retractors. Third, the nerve lies in perilous proximity to the femoral insertion of the gluteus maximus tendon; as we dissect distally to mobilize the proximal femur or perform an extended trochanteric osteotomy (ETO), meticulous identification and protection of the nerve in this distal zone are mandatory.
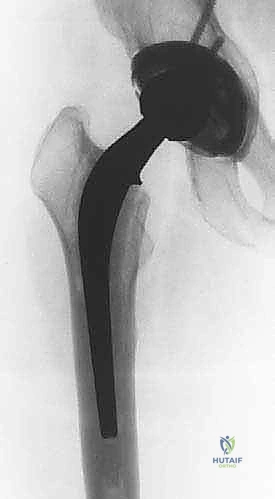
The superior gluteal nerve and the anterior neurovascular bundle demand equal vigilance. The superior gluteal nerve, which innervates the critical hip abductors (gluteus medius and minimus) and the tensor fasciae latae, courses anteriorly along the outer table of the ilium, approximately 4 to 5 centimeters superior to the tip of the greater trochanter. Aggressive superior dissection, errant retractor placement, or proximal migration of the acetabular component can easily compromise this nerve, resulting in a devastating and often irreversible Trendelenburg gait. Anteriorly, the femoral nerve, artery, and vein are generally shielded by the robust iliopsoas muscle belly during posterior approaches. However, in the setting of severe anterior acetabular defects, medial protrusio, or during anterior supine approaches, these structures become highly vulnerable. Penetration of the anterior acetabular wall with drills, screws, or extraction instruments can lead to catastrophic vascular injury.
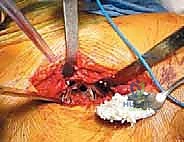
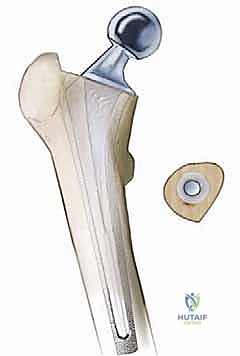
Understanding the osteology and the altered biomechanics of the revised hip is fundamental to component extraction and subsequent fixation. The proximal femur in a revision setting often exhibits stress shielding, where the rigid metallic implant has bypassed physiological load transfer to the proximal host bone, leading to cortical thinning and profound osteopenia. The vastus ridge and the linea aspera serve as critical landmarks for muscular attachments and for planning the ETO. The linea aspera, the thick posterior diaphyseal ridge, acts as the posterior boundary for the ETO cut, ensuring a robust cortical hinge. On the acetabular side, the anterior and posterior columns provide the structural foundation. A well-fixed porous-coated acetabular shell distributes hoop stresses evenly across the peripheral rim. Attempting to pry or lever such a shell without completely disrupting the bone-implant interface will inevitably result in a blowout fracture of the supporting columns, converting a contained defect into a massive uncontained structural failure.
Exhaustive Indications and Contraindications
The decision to remove a well-fixed total hip arthroplasty component is never taken lightly and must be supported by rigorous clinical and radiographic indications. The primary and most absolute indication is chronic Periprosthetic Joint Infection (PJI). Sepsis surrounding a THA, frequently driven by biofilm-forming organisms such as Staphylococcus aureus or coagulase-negative staphylococci, progresses relentlessly along the implant interfaces. Irrigation and debridement with modular component exchange are notoriously ineffective for chronic infections. The gold standard remains a two-stage exchange arthroplasty, which necessitates the complete, meticulous extraction of all foreign materials, including well-fixed stems, acetabular shells, and all cement and restrictive plugs, followed by thorough debridement and placement of an antibiotic-eluting spacer.

Beyond infection, mechanical and biologic failures dictate the need for extraction. Recurrent instability that is refractory to conservative management and modular liner exchange often requires the removal of a well-fixed acetabular or femoral component to correct profound malversion or offset deficiencies. Severe, progressive osteolysis represents another critical indication. As particulate debris generates a massive macrophage response, the resulting bone resorption can critically undermine the structural integrity of the pelvis or femur. Even if the implant remains clinically stable, the progressive loss of bone stock threatens future reconstructive options and mandates intervention before catastrophic failure occurs. Furthermore, the failure of modular locking mechanisms, severe functionally limiting limb-length discrepancies, and the need to address adverse local tissue reactions (ALTR) from metal-on-metal or taper corrosion are definitive indications for the removal of otherwise stable hardware.
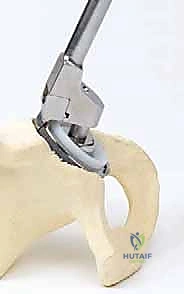
Contraindications to the removal of well-fixed components are primarily relative and depend heavily on patient physiology and the specific clinical scenario. Absolute contraindications include active, untreated distant systemic infections (e.g., endocarditis, active pneumonia) that must be optimized prior to elective revision. Severe medical comorbidities that render the patient unfit for a prolonged, physiologically demanding surgical procedure with significant anticipated blood loss must be carefully weighed against the benefits of revision. A relative contraindication exists in the setting of massive, un-reconstructable bone loss where the current implant, though perhaps suboptimally positioned, provides the only remaining stability, and removal would lead to a flail joint or require a total femoral replacement in a low-demand patient.
| Category | Indications for Removal of Well-Fixed Components | Contraindications (Absolute & Relative) |
|---|---|---|
| Infectious | Chronic Periprosthetic Joint Infection (PJI) requiring 2-stage exchange. | Active, untreated systemic bacteremia or distant infection (Absolute). |
| Mechanical | Recurrent dislocation due to severe component malposition; Locking mechanism failure; Component fracture. | Patient medically unfit for prolonged anesthesia and massive blood loss (Absolute). |
| Biologic | Progressive, massive osteolysis threatening structural integrity; Adverse Local Tissue Reaction (ALTR) / Metallosis. | Asymptomatic, non-progressive osteolysis in an elderly, low-demand patient (Relative). |
| Functional | Severe, functionally debilitating Limb-Length Discrepancy (LLD) refractory to shoe lifts. | Massive, un-reconstructable pelvic discontinuity where current implant provides only stability (Relative). |
Pre-Operative Planning, Templating, and Patient Positioning
Preoperative planning in revision THA is the blueprint for success; it is an exhaustive process that begins long before the patient enters the operating room. Diagnostic imaging is the cornerstone of this phase. We require, at a minimum, high-quality, biplanar plain radiographs of the pelvis and the entire femur down to the knee. These images must be scrutinized for signs of subtle loosening, radiolucent lines, endosteal scalloping, and the extent of any cement mantle. Oblique Judet views are absolutely essential, as they provide an orthogonal assessment of the anterior and posterior acetabular columns, often revealing occult osteolysis or column discontinuities that are completely masked on standard anteroposterior (AP) projections.
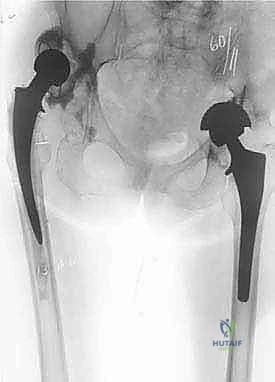
Advanced cross-sectional imaging has revolutionized our preoperative assessment. Computed Tomography (CT) scanning, particularly utilizing Metal Artifact Reduction Sequence (MARS) protocols, is indispensable for evaluating the true volumetric extent of pelvic and femoral osteolysis. Plain radiographs notoriously underestimate bone loss by up to 50%. CT imaging allows for precise three-dimensional mapping of defects, guiding the necessity for structural allografts, trabecular metal augments, or custom triflange components. In addition to imaging, a rigorous laboratory workup is mandatory. Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP) must be drawn; if elevated, a preoperative hip aspiration is performed for synovial fluid cell count, differential, and extended cultures to definitively rule out an indolent PJI before embarking on a presumed aseptic revision.
Digital templating is a dynamic, multi-step process in revision surgery. We must template not only the anticipated reconstructive implants but also the extraction strategy. When templating the femur, we assess the required length of an Extended Trochanteric Osteotomy (ETO) to bypass the well-fixed portion of the stem. The reconstructive stem must achieve a minimum of 4 to 6 centimeters of diaphyseal scratch fit distal to the most distal aspect of the ETO or any cortical defect. On the acetabular side, we estimate the size of the true acetabulum and plan for the center of rotation, anticipating the need for jumbo cups or augmentations based on the Paprosky classification of bone loss. Patient positioning is typically in the lateral decubitus position, utilizing a rigid pelvic fixation device (peg board or rigid bean bag) to ensure the pelvis remains perfectly orthogonal to the floor. The entire limb must be prepped and draped free to allow for dynamic assessment of stability, leg length, and extensive distal dissection if a long ETO is required.
Step-by-Step Surgical Approach and Fixation Technique
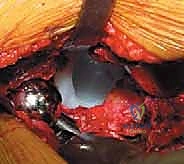
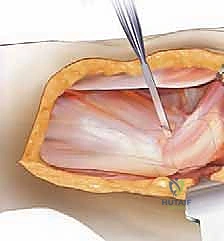
The surgical approach for removing well-fixed components must be extensile, versatile, and meticulously executed. The posterior approach is the workhorse of revision THA, offering excellent expansile capabilities to both the ilium and the femoral diaphysis. Upon incising the fascia lata and splitting the gluteus maximus, the surgeon is immediately confronted with distorted anatomy and dense scar tissue. The sciatic nerve must be identified and carefully protected, though formal neurolysis is only performed if the nerve is encased in scar or if massive lengthening is anticipated. The pseudocapsule is excised to mobilize the proximal femur. Dislocation of the hip can be exceedingly difficult; if the joint is stiff, in situ dissociation of the modular head or sectioning of the femoral neck may be required to prevent iatrogenic femoral or acetabular fractures during forceful dislocation maneuvers.

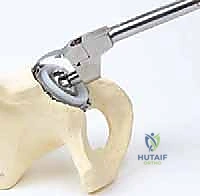
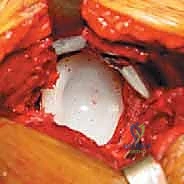
Extraction of a well-fixed uncemented acetabular component demands extreme patience and specialized instrumentation. The fundamental principle is to disrupt the bone-implant interface completely before applying any extractive force. We utilize specialized explant systems consisting of a centering head that locks into the modular shell, combined with sweeping, hemispherical blades that match the outer diameter of the cup. These blades are carefully driven along the interface, maintaining strict adherence to the metal shell to avoid diving into and destroying the precious remaining subchondral host bone. If an explant system is unavailable or incompatible, multiple curved, flexible osteotomes (e.g., Gouges) are meticulously advanced circumferentially around the shell. Any retaining screws must be identified and removed; stripped screw heads may require high-speed metal-cutting burrs. Only when the shell spins freely within the cavity should extraction be attempted.
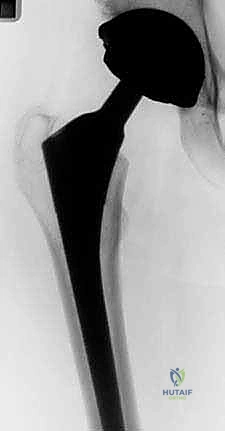
Addressing the well-fixed femoral stem frequently necessitates an Extended Trochanteric Osteotomy (ETO). The ETO is a controlled, longitudinal osteotomy of the lateral femur that includes the greater trochanter and a portion of the lateral diaphyseal cortex. It provides unparalleled direct visualization of the stem-bone or stem-cement interface, allows for the safe extraction of the implant and cement mantle, and facilitates the anatomic correction of varus remodeling. The length of the ETO is dictated by preoperative templating, typically extending just distal to the porous coating of an uncemented stem or distal to the cement mantle.
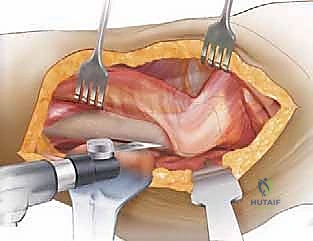
To perform the ETO, the vastus lateralis is elevated anteriorly, exposing the lateral cortex. A posterior longitudinal cut is made just anterior to the linea aspera using an oscillating saw, ensuring a robust posterior cortical hinge. An anterior longitudinal cut is made, followed by a transverse distal cut connecting the two, carefully avoiding penetration of the prosthetic stem, which can dull the saw blade and create stress risers.

Multiple broad osteotomes are then sequentially impacted along the osteotomy from posterior to anterior, gently levering the lateral bone fragment open like a book. The gluteus medius and minimus remain attached to the proximal aspect of this fragment, preserving their blood supply and functional tension.

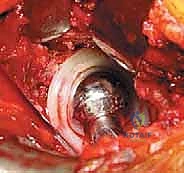
Once the ETO is open, the well-fixed stem is exposed. For uncemented stems, high-speed pencil burrs, flexible osteotomes, and ultrasonic devices are used to disrupt the porous ingrowth at the interface. For cemented stems, the cement mantle is carefully sectioned and removed using specialized cement splitters and ultrasonic tools.
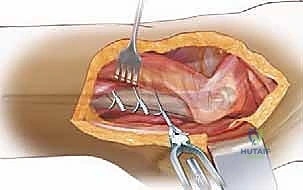
Following extraction and subsequent preparation of the canal, the new revision stem is inserted. The ETO fragment is then reduced anatomically around the new stem and secured using multiple heavy-gauge cerclage wires or cables. The rigid fixation of the ETO to the diaphyseal bypass stem is critical for achieving union, which typically occurs reliably due to the preserved soft tissue pedicle.
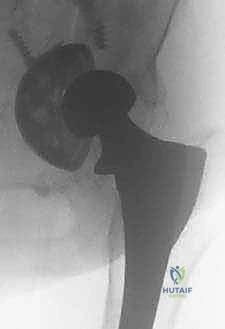
Complications, Incidence Rates, and Salvage Management
Despite meticulous planning and execution, the extraction of well-fixed components carries a significantly elevated risk of intraoperative and postoperative complications compared to primary THA. Intraoperative periprosthetic fractures are perhaps the most immediate threat. On the acetabular side, aggressive levering of a partially fixed cup can result in a transverse or column fracture, instantly converting a contained Paprosky Type II defect into an uncontained Type III or a catastrophic pelvic discontinuity. The incidence of intraoperative acetabular fracture during revision is estimated at 2-5%. Salvage requires immediate recognition, rigid internal fixation using column plates if possible, and spanning the defect with a highly porous jumbo cup, trabecular metal augments, or a custom triflange construct.

Femoral fractures are equally treacherous, occurring in up to 10% of cases involving well-fixed stem extraction without an ETO. The use of an ETO significantly reduces the risk of uncontrolled diaphyseal splintering but introduces the risk of fracturing the osteotomy fragment itself, particularly at the thin base of the greater trochanter. If the diaphyseal femur fractures distally during stem extraction or reaming, the salvage strategy mandates bypassing the most distal extent of the fracture by a minimum of two cortical diameters with a long, diaphyseal-engaging splined, tapered stem, supplemented by cerclage cabling or strut allografts.
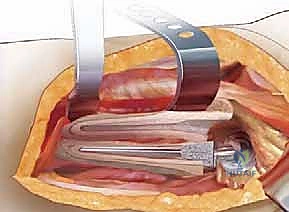
Neurovascular complications, particularly sciatic nerve palsy, occur at a higher rate in revision surgery (up to 3-5%) due to altered anatomy, scar tethering, and the necessity for limb lengthening. Intraoperative monitoring (SSEP and MEP) can be a valuable adjunct during complex revisions. If a postoperative palsy is noted, immediate evaluation for a compressive hematoma is required; otherwise, management is supportive with an ankle-foot orthosis (AFO). Infection and instability are the most common causes for re-revision. The dead space created by massive bone loss and the prolonged operative times predispose these patients to PJI. Instability, occurring in up to 10-15% of revisions, is mitigated by restoring offset, utilizing dual-mobility articulations, or, in extreme cases of abductor deficiency, utilizing constrained liners.
| Complication | Estimated Incidence in rTHA | Intraoperative / Postoperative Salvage Management |
|---|---|---|
| Intraoperative Femoral Fracture | 4% - 10% | Bypass fracture by >2 cortical diameters with long splined stem; cerclage cables; strut allograft. |
| Intraoperative Acetabular Fracture | 2% - 5% | Column plating; use of highly porous jumbo cups, augments, or cup-cage constructs. |
| Sciatic Nerve Palsy | 1% - 5% | Rule out compressive hematoma; AFO bracing; physical therapy; consider nerve exploration if transection suspected. |
| Postoperative Instability / Dislocation | 5% - 15% | Closed reduction; brace; if recurrent, re-revision for dual-mobility, constrained liner, or offset correction. |
| ETO Nonunion / Migration | 1% - 3% | Observation if asymptomatic; re-cabling and bone grafting if migrating and causing abductor weakness. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following a complex revision THA with component extraction is fundamentally distinct from primary THA and must be highly individualized based on the quality of bone stock, the security of the initial fixation, and the presence of an ETO or structural allografts. The immediate postoperative phase (Phase 1: Weeks 0-2) is focused on wound healing, deep vein thrombosis (DVT) prophylaxis, and the protection of the surgical reconstruction. If an ETO was performed, or if significant uncontained bone defects were grafted, weight-bearing is strictly limited. Patients are typically restricted to toe-touch weight-bearing (TTWB) or a maximum of 20 pounds of flat-foot weight-bearing on the operative extremity to prevent subsidence of the diaphyseal stem and to protect the ETO construct.
Phase 2 (Weeks 2-6) involves the gradual progression of passive and active-assisted range of motion. However, active abduction is strictly prohibited if an ETO was utilized. The gluteus medius and minimus are attached to the osteotomized fragment; premature active contraction can cause superior migration or catastrophic failure of the cerclage cables, leading to nonunion and a permanent Trendelenburg gait. Hip precautions (avoiding extreme flexion, adduction, and internal rotation for posterior approaches) are strictly enforced, as the soft tissue envelope is inherently compromised in revision settings.
Phase 3 (Weeks 6-12) marks the transition to progressive weight-bearing and active muscle strengthening. At the 6-week mark, new radiographs are obtained to assess the incorporation of the ETO and the stability of the implants. If bridging callus is visible and the ETO appears stable, patients are transitioned to partial, and eventually full, weight-bearing using an assistive device. Active abductor strengthening is initiated cautiously, beginning with isometric exercises and progressing to gravity-resisted movements. Phase 4 (Beyond 12 weeks) focuses on normalizing gait mechanics, core stabilization, and returning to low-impact activities of daily living. Patients must be counseled that maximal medical improvement following a massive revision THA may take up to 12 to 18 months, and high-impact activities are permanently restricted to ensure the longevity of the complex reconstruction.
Summary of Landmark Literature and Clinical Guidelines
The evolution of techniques for removing well-fixed components is heavily grounded in decades of rigorous clinical research and biomechanical studies. The American Academy of Orthopaedic Surgeons (AAOS) clinical practice guidelines emphasize the necessity of exhaustive preoperative workups, particularly the utilization of the Musculoskeletal Infection Society (MSIS) criteria to definitively rule out PJI prior to presumed aseptic revisions. The literature consistently demonstrates that failure to identify an indolent infection prior to retaining or replacing components leads to unacceptably high failure rates.
The popularization and refinement of the Extended Trochanteric Osteotomy (ETO) represent a watershed moment in revision arthroplasty. Landmark papers by Younger et al. and Paprosky et al. have definitively shown that the ETO provides superior, safe access to the femoral diaphysis, drastically reducing the incidence of uncontrolled intraoperative femoral fractures compared to traditional extraction methods. Their long-term follow-up studies confirm that when executed correctly and secured with modern cerclage systems, the ETO heals reliably in over 95% of cases, without significantly compromising ultimate abductor strength.
Furthermore, the management of acetabular defects following the extraction of well-fixed cups has been revolutionized by the introduction of highly porous trabecular metal components. Studies by Sporer and Paprosky have validated the use of these biomaterials in bridging massive uncontained defects, demonstrating superior biologic ingrowth and long-term survivorship compared to historical techniques utilizing massive structural allografts. As we continue to advance our understanding of periprosthetic osteolysis and implant biomechanics, the literature underscores a paradigm shift toward earlier intervention in the presence of progressive bone loss, rather than waiting for catastrophic clinical failure, thereby preserving the vital host bone necessary for a successful, enduring revision reconstruction.
