Masterclass: Revision Total Hip Arthroplasty with Proximal Femoral Replacement for Massive Bone Loss
Key Takeaway
This masterclass guides fellows through proximal femoral replacement for severe bone loss in revision total hip arthroplasty. We cover meticulous preoperative planning, critical surgical anatomy, precise intraoperative execution, and strategies to manage potential complications. Emphasizing modular components and soft tissue preservation, this procedure is a limb-sparing salvage for complex cases, ensuring optimal functional outcomes and stability.
Comprehensive Introduction and Patho-Epidemiology
The management of massive proximal femoral bone loss in the setting of revision total hip arthroplasty (THA) represents one of the most formidable challenges in adult reconstructive surgery. When patients present with catastrophic bone deficiency secondary to multiple previous arthroplasty failures, recalcitrant deep periprosthetic joint infections, massive osteolysis, or complex periprosthetic fractures (such as Vancouver B3 or C patterns), the reconstructive armamentarium is severely limited. In these dire clinical scenarios, proximal femoral replacement (PFR) utilizing a megaprosthesis serves as a critical, salvage limb-sparing procedure. The primary objective is no longer merely the optimization of high-demand kinematics, but rather the restoration of basic ambulatory function, the provision of a stable articulation, and the avoidance of a highly morbid major amputation or a functionally devastating resection arthroplasty (Girdlestone procedure).
Historically, the orthopedic community relied heavily on custom-manufactured, monolithic megaprostheses or allograft-prosthesis composites (APCs) to bridge these massive skeletal defects. While monolithic designs provided immediate structural continuity, they were plagued by a profound inability to intraoperatively fine-tune limb length, offset, and soft tissue tension, leading to unacceptably high rates of postoperative instability. Furthermore, while APCs theoretically offered the advantage of restoring host bone stock and providing anatomic sites for tendinous reattachment, their clinical reality was marred by catastrophic complication profiles. The literature consistently demonstrates that APCs carry prohibitive risks of deep infection, junctional nonunion at the host-allograft interface, recurrent dislocation, and eventual aseptic loosening due to allograft resorption and mechanical failure.
The advent and refinement of second-generation modular proximal femoral replacements have revolutionized our approach to these complex reconstructions. Modern modular systems afford the arthroplasty surgeon unprecedented intraoperative flexibility. By utilizing a combination of variable-length distal stems, intercalary segments, and proximal bodies with adjustable version and offset, the surgeon can meticulously dial in the biomechanical parameters required to optimize soft tissue tension and joint stability. These contemporary prostheses are also engineered with specialized porous coatings, hydroxyapatite collars, and dedicated suture holes or porous metal ingrowth surfaces designed specifically to facilitate the reattachment and integration of the remaining host soft tissue envelope.
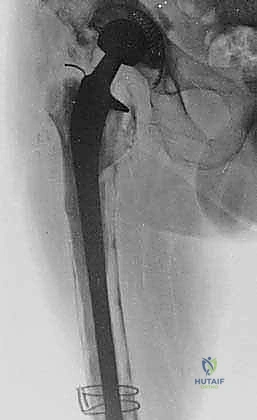
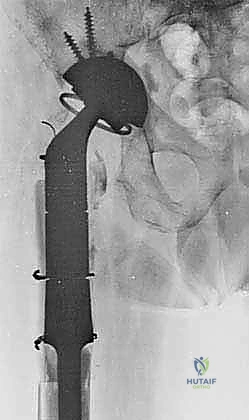
For the contemporary revision arthroplasty surgeon, modular proximal femoral replacement is widely considered a more reliable, reproducible, and less technically capricious solution compared to massive structural allografts. By bypassing the biological unpredictability of massive allograft incorporation, modular PFR provides immediate, rigid fixation and allows for expedited postoperative mobilization. However, the success of this procedure hinges entirely on a profound understanding of distorted regional anatomy, exhaustive preoperative templating, and meticulous surgical execution.
Detailed Surgical Anatomy and Biomechanics
Before a surgical incision is even contemplated, the reconstructive surgeon must possess an intimate, three-dimensional understanding of the regional anatomy. In the multiply revised hip, normal fascial planes are obliterated, replaced by dense, avascular scar tissue that tether critical neurovascular structures to the joint capsule and remnant bone. Navigating this distorted anatomical landscape requires both surgical precision and a deep appreciation for the altered biomechanical milieu.
Neurovascular Structures in Revision Scenarios
The superior gluteal nerve and its accompanying vessels represent the most critical structures at risk during the proximal exposure. This neurovascular bundle is the sole motor supply to the gluteus medius, gluteus minimus, and tensor fasciae latae—the essential hip abductors. It exits the pelvis via the suprapiriform portion of the greater sciatic foramen. In a primary setting, the nerve typically courses approximately 4 to 5 cm proximal to the tip of the greater trochanter, deep to the gluteus medius. However, in cases of severe proximal femoral bone loss, proximal migration of the femur, or extensive heterotopic ossification, this safe zone is highly variable and often compromised. Iatrogenic injury to the superior gluteal nerve results in an irreversible abductor lurch (Trendelenburg gait), which significantly exacerbates the risk of postoperative dislocation in a megaprosthesis.
Posteriorly, the sciatic nerve demands paramount respect. As the largest nerve in the human body, it exits the pelvis through the infrapiriform foramen, coursing deep to the gluteus maximus and superficial to the short external rotators. In revision surgery, the sciatic nerve is frequently encased in dense perineural fibrosis or drawn anteriorly toward the posterior column of the acetabulum. It is at extreme risk during a posterolateral approach, aggressive posterior retractor placement, or during the extraction of extruded cement mantles. Furthermore, the sciatic nerve (specifically its peroneal division) is highly susceptible to traction neurapraxia during the restoration of limb length. Any anticipated lengthening greater than 3 to 4 cm necessitates extreme caution and warrants the use of intraoperative neuromonitoring.
Anteriorly, the femoral nerve and vessels reside within the femoral triangle. While generally protected during standard lateral or posterolateral exposures, they become vulnerable during extensile anterior dissections, the release of severe flexion contractures, or the management of anteriorly migrated hardware. The perforating vessels, arising from the profunda femoris artery, are of particular surgical consequence during the distal exposure of the femur. As the vastus lateralis is elevated or mobilized via a slide osteotomy, these perforators must be systematically identified and meticulously ligated to prevent massive postoperative hematoma formation. Preservation of the main pedicle to the vastus lateralis, crossing anteriorly along the rectus femoris fascia, is absolutely critical if a vastus lateralis muscle flap is planned for soft tissue coverage of the metallic prosthesis.
Musculotendinous Intervals and the Abductor Mechanism
The integrity of the abductor mechanism is the single most important determinant of postoperative stability and functional outcome following a proximal femoral replacement. The gluteus medius and minimus normally insert onto the greater trochanter. In the catastrophic revision setting, the greater trochanter is frequently fractured, osteolyzed, or entirely absent. The remaining abductor musculature is often atrophic, scarred, or frankly avulsed. The surgical strategy must prioritize the meticulous preservation of any functional abductor tissue. During the exposure, a thick sleeve of tissue should be maintained to allow for robust reattachment to the prosthesis or to the tensor fasciae latae and vastus lateralis via a direct repair or synthetic mesh augmentation.
The deep external rotators (piriformis, quadratus femoris, obturator internus, and the gemelli) are typically sacrificed or found to be absent in these multiply revised hips. Their absence significantly contributes to the inherent posterior instability of the reconstruction. The adductor group (brevis, longus, gracilis, and magnus), located medially, are rarely in the direct surgical field but can cause a severe adduction contracture that may require percutaneous or open tenotomy to achieve appropriate soft tissue balancing and reduce the risk of lateral dislocation.
The vastus lateralis is the workhorse of the femoral exposure and closure. Arising from the greater trochanter and the linea aspera, it forms the entire lateral compartment of the thigh. For extensive distal exposure, the vastus lateralis is typically reflected anteriorly from the linea aspera in a subperiosteal fashion. Following the implantation of the megaprosthesis, the vastus lateralis must be carefully mobilized and wrapped around the proximal body of the implant. This crucial step obliterates dead space, provides a biological envelope to reduce infection risk, and serves as an anchoring point for the reattachment of the proximal abductor mechanism.
Osteology and Biomechanical Considerations
The fundamental osteological challenge in proximal femoral replacement is identifying a segment of host bone capable of supporting the massive mechanical loads transferred by the megaprosthesis. The normal proximal landmarks—the trochanters, the calcar, the femoral neck—are entirely absent. The surgeon must meticulously debride all necrotic, infected, or compromised bone until a bleeding, circumferentially intact cortical diaphyseal bed is encountered. This dictates the level of the transverse osteotomy.
A rigid biomechanical prerequisite for a successful proximal femoral replacement is the presence of adequate distal femoral bone stock. The surgeon must secure a minimum of 10 to 12 cm of diaphyseal bone distal to the resection level to achieve stable fixation of the stem. If this minimum length is not attainable, the lever arm of the megaprosthesis will inevitably lead to early mechanical failure, loosening, or periprosthetic fracture, and the surgeon must immediately pivot to a total femoral replacement.
The biomechanics of a proximal femoral replacement are characterized by severe stress shielding and altered joint reaction forces. Because the proximal femur is replaced by a rigid metallic construct, the physiological load transfer to the remaining native distal femur is drastically altered. The modulus of elasticity of the cobalt-chromium or titanium stem is vastly greater than that of cortical bone, leading to proximal stress shielding and potential distal cortical hypertrophy. The modular junctions of the prosthesis are also subject to immense bending and torsional moments, necessitating precise intraoperative assembly (often with Morse tapers and locking screws) to prevent catastrophic fretting, corrosion, or mechanical dissociation.
Exhaustive Indications and Contraindications
The decision to proceed with a proximal femoral replacement must be made judiciously, as it represents a major, life-altering intervention with a high complication profile. It is reserved for clinical scenarios where traditional revision techniques (such as long stems with impaction grafting) are biomechanically impossible or have already failed.
The most common non-neoplastic indication for a proximal femoral replacement is massive aseptic osteolysis with severe bone loss (Paprosky Type IIIb or IV femoral defects) where the remaining proximal bone is a mere cortical shell incapable of supporting any standard or modular revision stem. In these cases, the bone is essentially functionally dead, and attempting to bypass it with a long stem will inevitably lead to early subsidence and failure.
Complex periprosthetic fractures represent another major indication, specifically Vancouver Type B3 fractures (fracture around a loose stem with severely compromised proximal bone stock) or highly comminuted Vancouver Type C fractures extending into the proximal diaphysis in an elderly, low-demand patient. In these acute settings, PFR allows for immediate weight-bearing and avoids the prolonged immobilization and high nonunion rates associated with complex osteosynthesis in osteoporotic bone.
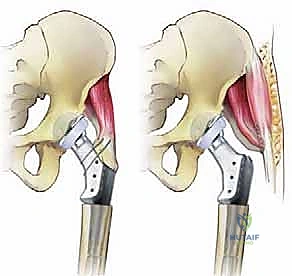
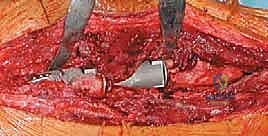
Recalcitrant periprosthetic joint infections (PJI) often necessitate PFR. Following a thorough first-stage debridement and placement of an antibiotic spacer, the subsequent bone loss can be catastrophic. During the second-stage reimplantation, if the proximal femur is deemed non-viable or structurally inadequate following serial debridements, a PFR is often the only viable reconstructive option to restore limb continuity.
| Indication Category | Specific Clinical Scenarios | Rationale for Proximal Femoral Replacement |
|---|---|---|
| Aseptic Loosening | Paprosky Type IIIb or IV defects; failed APCs. | Insufficient proximal bone to support a revision stem; need for immediate structural stability. |
| Periprosthetic Fracture | Vancouver Type B3; highly comminuted Type C in elderly. | Bypasses non-constructible bone; allows immediate full weight-bearing in frail patients. |
| Infection (Stage 2) | Massive bone loss following explantation and spacer placement. | Eradicates dead space; provides stable reconstruction when host bone is completely compromised. |
| Neoplastic Disease | Primary bone sarcomas; extensive metastatic destruction. | En bloc resection of the tumor and immediate functional restoration of the limb. |
Contraindications must be strictly observed to prevent catastrophic outcomes. An absolute contraindication is the presence of an active, uncontrolled deep periprosthetic joint infection in a single-stage setting. Implanting a massive, modular metallic device into an infected bed guarantees failure. Inadequate distal bone stock (less than 10 cm of diaphyseal bone) is a strict biomechanical contraindication, as it precludes stable stem fixation and necessitates a total femoral replacement.
Relative contraindications include severe medical comorbidities that preclude a lengthy, blood-intensive surgical procedure. Patients with profound peripheral vascular disease, severe cardiopulmonary compromise, or those who are non-ambulatory at baseline may be better served by a resection arthroplasty or chronic suppressive antibiotic therapy. Furthermore, a complete lack of an abductor mechanism, while not an absolute contraindication, drastically increases the risk of dislocation and may require the concurrent use of a constrained acetabular liner or a dual-mobility articulation.
Pre-Operative Planning, Templating, and Patient Positioning
The success of a proximal femoral replacement is predicated on exhaustive preoperative planning. These are not procedures that can be improvised intraoperatively. The surgeon must meticulously evaluate the patient's clinical status, rule out occult infection, and utilize advanced imaging to map the surgical strategy.
Clinical Evaluation and Infection Surveillance
A comprehensive clinical assessment begins with a detailed history and physical examination. The nature of the patient's pain is highly informative: "start-up" thigh pain is pathognomonic for a loose femoral stem, whereas deep groin pain often suggests acetabular component loosening or intra-articular pathology. A meticulous gait analysis is mandatory; an antalgic gait indicates mechanical pain, while a profound Trendelenburg lurch signals catastrophic abductor deficiency, which will dictate the complexity of the soft tissue reconstruction and the choice of acetabular articulation.
Infection must be considered present until definitively proven otherwise. A standard serological workup, including Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP), is mandatory. Regardless of the serological results, a preoperative fluoroscopically guided hip aspiration is strongly recommended. Synovial fluid should be analyzed for cell count, differential, and extended aerobic, anaerobic, and fungal cultures. If the diagnosis remains equivocal, advanced biomarkers such as synovial alpha-defensin or Next-Generation Sequencing (NGS) should be employed. Intraoperatively, the surgical team must be prepared to send multiple independent tissue samples for frozen section analysis to quantify polymorphonuclear leukocytes (PMNs) per high-power field before committing to reimplantation.
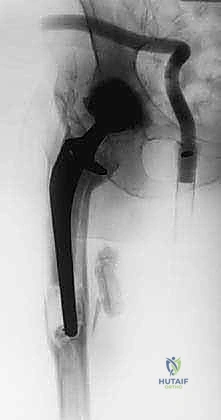
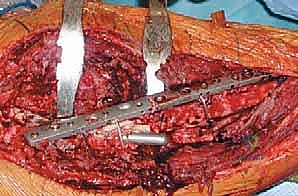
Advanced Imaging Modalities
Standard orthogonal plain radiographs (AP pelvis, true lateral of the hip, and full-length femur) are the foundation of the imaging workup, providing a macroscopic view of bone destruction, hardware failure, and overall limb alignment. However, plain films drastically underestimate the extent of bone loss.
A high-resolution Computed Tomography (CT) scan with Metal Artifact Reduction Sequence (MARS) is absolutely essential. The CT scan allows for precise, three-dimensional mapping of the remaining cortical bone. It is critical for identifying subtle, non-displaced periprosthetic fractures, evaluating the integrity of the acetabular columns, and most importantly, determining the exact level of viable diaphyseal bone where the transverse osteotomy will be performed. Magnetic Resonance Imaging (MRI) is occasionally utilized to assess the viability of the abductor musculature or to delineate the extent of soft tissue masses or fluid collections.
In cases where the patient has undergone multiple previous surgeries, particularly those involving intrapelvic hardware migration or previous vascular bypass procedures, a preoperative consultation with a vascular surgeon and formal CT angiography are highly recommended. This maps the distorted vascular anatomy, particularly the relationship of the external iliac and femoral vessels to the anterior capsule, mitigating the risk of catastrophic iatrogenic hemorrhage during the exposure.
Digital Templating and Component Selection
Digital templating is a non-negotiable step in the preoperative workflow. Using specialized software and the patient's calibrated radiographs, the surgeon must construct a precise architectural blueprint for the reconstruction.
The first step in templating is establishing the resection level. The surgeon must identify the most proximal region of the diaphysis that exhibits a circumferentially intact, healthy cortical tube. From this resection line, the surgeon must measure distally to ensure the presence of at least 10 to 12 cm of diaphyseal bone to accommodate the distal stem.
Templating must meticulously account for the restoration of limb length and femoral offset. The contralateral, unaffected hip (if present) serves as the primary reference. The surgeon must calculate the exact combination of modular components—the distal stem length and diameter, the length of the intercalary segment, the height of the proximal body, and the neck length—required to recreate the native center of rotation. Templating for both cemented and cementless distal fixation options is mandatory, as intraoperative bone quality may dictate a change in strategy.
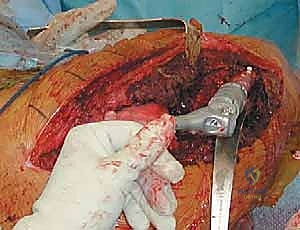
Patient Positioning and Intraoperative Setup
The patient is typically positioned in the lateral decubitus position, which provides the most versatile and extensile access to both the acetabulum and the entire length of the femur. Meticulous padding is critical; an axillary roll must be placed to protect the dependent brachial plexus, and all bony prominences (especially the dependent fibular head) must be heavily padded to prevent compression neuropathies during these prolonged procedures.
The pelvis must be rigidly secured with anterior and posterior bolsters to ensure it remains strictly perpendicular to the floor. Any unrecognized pelvic tilt will lead to severe miscalculations in component version and leg length. Prior to draping, a formal clinical assessment of limb length is performed and recorded for intraoperative comparison.
Given the high probability of significant limb lengthening (often exceeding 4 cm to restore soft tissue tension), the utilization of intraoperative neuromonitoring—specifically Somatosensory Evoked Potentials (SSEPs) and spontaneous Electromyography (sEMG) of the sciatic and femoral nerves—is strongly recommended. This provides the surgical team with real-time, objective feedback regarding nerve strain during the trial reduction phase.
The draping protocol must be expansive. Following a meticulous 10-minute surgical scrub and the application of an iodine-based adhesive drape, the entire lower extremity must be prepped into the sterile field. Crucially, the knee must be completely exposed and freely mobile within the operative field. This allows the surgeon to accurately assess leg length, evaluate femoral version by referencing the transmalleolar axis or the posterior condyles, and provides immediate access should a fracture propagate distally, necessitating an extensile approach to the distal femur.
Step-by-Step Surgical Approach and Fixation Technique
The execution of a proximal femoral replacement is a masterclass in extensile exposure, meticulous debridement, and precise biomechanical reconstruction. The surgical team must be prepared for a highly dynamic intraoperative environment where the templated plan meets the reality of compromised host tissues.
Incision and Deep Exposure
An extended posterolateral approach or a direct lateral approach is typically utilized, often incorporating and extending previous surgical incisions. The skin incision must be generous, often extending from proximal to the greater trochanter down the lateral aspect of the thigh to the level of the knee, depending on the required length of the distal stem.
Upon incising the fascia lata, the surgeon encounters the "pseudo-capsule"—a dense, hypertrophic layer of scar tissue that has formed around the failed implant. This tissue must be carefully excised to expose the underlying joint space and the remnant proximal femur. The sciatic nerve must be systematically identified and protected throughout the posterior exposure, taking immense care not to place aggressive retractors that could cause traction neurapraxia.
The vastus lateralis is identified and mobilized. In cases of massive bone loss, the vastus lateralis is elevated subperiosteally from the linea aspera, sweeping it anteriorly. The perforating vessels from the profunda femoris must be meticulously identified, clamped, and ligated (preferably with suture ligatures rather than electrocautery) to prevent postoperative hematoma. The integrity of the vastus lateralis must be preserved, as it will be utilized as a biological wrap around the final prosthesis.
Component Extraction and Debridement
The extraction of failed components and the debridement of compromised tissue is often the most time-consuming phase of the procedure. Loose stems can typically be extracted with standard slap hammers. However, well-fixed distal cement mantles or broken cementless stems require specialized extraction tools, flexible osteotomes, and occasionally, an extended trochanteric osteotomy (ETO) or a distal cortical window.
Once the hardware is removed, exhaustive debridement is mandatory. All devitalized bone, metallosis-stained tissue, and inflammatory pseudotumors must be radically excised. The proximal femur is sequentially resected using an oscillating saw until healthy, bleeding, circumferentially intact cortical bone is reached. This transverse osteotomy marks the foundation of the reconstruction.

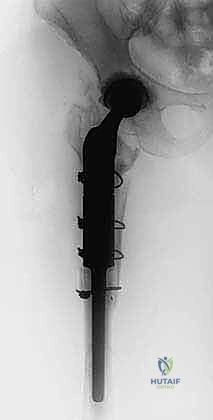
Femoral Preparation and Osteotomy
With the osteotomy complete, attention turns to the preparation of the distal femoral canal. The surgeon must prepare a minimum of 10 to 12 cm of diaphyseal bone to accept the distal stem.
The choice between a cemented and a cementless distal stem depends entirely on the quality of the diaphyseal bone. In younger patients with robust, thick cortical bone, a cementless, fully porous-coated or fluted tapered splined stem is preferred to achieve biologic fixation and long-term durability. The canal is sequentially reamed and broached to achieve a tight, diaphyseal scratch fit.
In elderly patients with severe osteopenia, capacious canals, or irradiated bone, a cemented stem is the gold standard. Cemented fixation provides immediate, rigid stability and bypasses the requirement for biologic ingrowth. The canal must be meticulously prepared with pulsatile lavage, a distal cement restrictor must be placed, and the bone cement must be delivered in a retrograde fashion using third-generation cementing techniques to ensure a uniform, pressurized mantle.
Trialing, Soft Tissue Tensioning, and Abductor Reattachment
Following the preparation of the distal canal, the modular trial components are assembled. The surgeon selects the appropriate distal stem, intercalary segment, and proximal body based on the preoperative template and intraoperative measurements.
The trial reduction is the most critical phase for ensuring postoperative stability. The surgeon must meticulously assess three parameters: limb length, femoral offset, and soft tissue tension. Limb length is verified by comparing the intraoperative position of the knees and utilizing preoperative reference pins placed in the ilium. The "shuck test" is performed; a well-tensioned megaprosthesis should allow no more than a few millimeters of distraction.
If the abductor mechanism is severely deficient, the surgeon must strongly consider revising the acetabular component to a dual-mobility articulation or a constrained liner to mitigate the profound risk of dislocation.
Once the final components are implanted and torqued to the manufacturer's specifications, the soft tissue reconstruction begins. The vastus lateralis is mobilized and wrapped securely around the metallic proximal body, sutured to the remnant gluteus maximus fascia posteriorly and the tensor fasciae latae anteriorly. The remaining abductor tendons (gluteus medius and minimus) are aggressively mobilized and attached to the specialized suture holes or porous ingrowth surfaces of the proximal prosthesis using heavy, non-absorbable sutures or specialized claw plates. In severe cases, synthetic mesh (e.g., Trevira tube) or allograft augmentation may be utilized to bridge the gap and provide a scaffold for fibrous tissue ingrowth.
Complications, Incidence Rates, and Salvage Management
Proximal femoral replacement is inherently associated with a high complication profile, reflecting the catastrophic nature of the underlying pathology and the magnitude of the surgical insult. The surgical team must be intimately familiar with these risks and possess the technical acumen to manage them.
Instability and dislocation represent the most frequent and frustrating complications, with historical rates ranging from 10% to over 30%. This is primarily driven by the profound loss of the abductor mechanism, the excision of the native joint capsule, and the inability to perfectly restore native soft tissue tension.
Deep periprosthetic joint infection is the second most common major complication, particularly in patients undergoing PFR for a prior infection or in the setting of massive oncologic resections. The massive metallic surface area, combined with a compromised, scarred soft tissue envelope, creates an ideal environment for bacterial colonization.
| Complication | Estimated Incidence | Etiology and Risk Factors | Salvage Management Strategies |
|---|---|---|---|
| Dislocation / Instability | 10% - 30% | Abductor deficiency; inadequate offset; component malposition; soft tissue laxity. | Closed reduction with prolonged bracing; revision to dual-mobility or constrained liner; soft tissue augmentation (mesh/allograft). |
| Deep Infection (PJI) | 5% - 15% | Massive foreign body; poor soft tissue envelope; prolonged operative time; prior PJI. | Aggressive DAIR (Debridement, Antibiotics, Implant Retention) if acute; Two-stage revision with custom articulating spacer if chronic. |
| Aseptic Loosening | 5% - 10% | Inadequate distal fixation (<10cm bone); stress shielding; poor bone quality. | Revision to a longer, thicker stem; transition to total femoral replacement if distal bone is completely exhausted. |
| Periprosthetic Fracture | 3% - 8% | Stress risers at the stem tip; severe osteoporosis; intraoperative cortical perforation. | Open reduction and internal fixation with cerclage cables/strut grafts; revision to a longer bypassing stem. |
| Nerve Palsy (Sciatic/Femoral) | 2% - 5% | Excessive limb lengthening (>4cm); direct retractor trauma; post-op hematoma. | Observation and AFO for foot drop; surgical decompression |
