Proximal Patellar Realignment: Medial Patellofemoral Ligament Reconstruction & Retinacular Plication Masterclass
Key Takeaway
This masterclass details proximal patellar realignment for chronic patellofemoral instability. We cover comprehensive surgical anatomy, meticulous preoperative planning, and step-by-step intraoperative execution of medial retinacular plication and MPFL reconstruction. Fellows will learn critical tensioning techniques, avoid common pitfalls, and understand postoperative rehabilitation and complication management for optimal patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
Welcome, fellows and colleagues, to the operating theater. Today, we are undertaking a masterclass on a challenging yet immensely rewarding pathology: patellofemoral instability. This condition, which predominantly presents as debilitating pain, apprehension, and mechanical dysfunction in children, adolescents, and young adults, can profoundly impact a patient's quality of life and athletic trajectory. It is of paramount importance to establish early in your training that mechanical instability—rather than isolated anterior knee pain—is the absolute primary indication for the proximal realignment procedures we will execute today. Performing a medial retinacular plication or a medial patellofemoral ligament (MPFL) reconstruction for isolated patellofemoral pain syndrome without objective instability is a historical error that reliably leads to dismal clinical outcomes and iatrogenic joint overload.
Patellofemoral instability manifests across a broad clinical spectrum, ranging from subtle, symptomatic subluxation to frank, traumatic lateral dislocation. The patho-epidemiology of this condition is multifactorial. While a subset of our patients will exhibit generalized ligamentous laxity (often quantified by a high Beighton score) predisposing them to atraumatic, recurrent subluxation, others—particularly high-demand athletes—experience instability driven by acute, high-energy traumatic events. Furthermore, underlying anatomical dysplasia plays a critical role in the natural history of the disease. A deficient lateral femoral slope of the trochlear groove, for instance, significantly reduces the inherent bony constraint of the joint. This morphological aberration drastically lowers the threshold of lateralizing force required to induce patellar translation or complete dislocation.
Injuries to the medial aspect of the patellofemoral joint during a dislocation event almost universally involve the disruption of the MPFL, frequently accompanied by plastic deformation, stretching, or complete macroscopic tearing of the medial retinaculum. This structural failure initiates a cascade leading to persistent apprehension and recurrent instability. The natural history of primary patellar dislocation suggests that patients with an atraumatic onset of instability, or those with significant baseline anatomic risk factors, possess a markedly higher likelihood of repeat episodes despite aggressive, protocol-driven non-operative measures. The literature demonstrates that following a second dislocation, the recurrence rate approaches 50%, underscoring the necessity for definitive surgical stabilization in the recurrent cohort.
The fundamental key to appropriate surgical intervention is the persistent, reproducible complaint of instability, corroborated by objective examination findings consistent with patellar tracking dysfunction, with or without concomitant pain. While an acute traumatic event may generate an osteochondral fracture or a symptomatic loose body necessitating urgent arthroscopic intervention, the primary, overarching goal of proximal realignment surgery is to restore native kinematics and stability. The debate surrounding acute, primary surgical intervention for first-time dislocators—especially young athletes without generalized ligamentous laxity—remains highly active in the literature. However, for those with recurrent instability who have exhausted diligent non-operative management, or those presenting with significant concomitant bony pathology such as large, displaced avulsion fractures, surgical stabilization is definitively indicated.
Detailed Surgical Anatomy and Biomechanics
Before we make our initial incision, we must meticulously review the complex, triplanar anatomy of the patellofemoral joint. A thorough, three-dimensional understanding of both the static and dynamic stabilizers is paramount to executing safe, anatomic, and biomechanically sound surgery.
Osteology and Articular Surfaces
The patellofemoral articulation is a highly complex joint comprising the patella—the largest sesamoid bone in the human body, embedded within the quadriceps tendon—and the trochlear groove of the distal femur. The posterior surface of the patella features distinct medial and lateral articular facets, separated by a central ridge, which articulate congruently with the femoral trochlea. The size, morphology, and congruence of these facets are critical variables for smooth, frictionless tracking during the flexion-extension arc. Variations in patellar morphology, such as Wiberg types I, II, and III, can subtly influence joint mechanics and contact pressures.
The femoral trochlea is a V-shaped cartilaginous groove that captures and guides the patella during early flexion. Its morphology is the primary static bony constraint to instability. A shallow trochlea, or one characterized by a hypoplastic lateral condyle or a supratrochlear spur, significantly predisposes the patient to lateral instability. This condition, known as trochlear dysplasia, is comprehensively categorized by the Dejour classification (Types A through D). The medial epicondyle serves as a crucial palpable landmark, acting as the broad femoral attachment site for the native MPFL. The adductor tubercle, located just superior to the medial epicondyle, is a common anatomical reference point, though precise radiographic identification (Schöttle's point) reveals the true isometric footprint is slightly more distal and anterior to the adductor tubercle.
The tibial tuberosity represents the distal insertion point of the patellar tendon. Its spatial position relative to the deepest portion of the trochlear groove—quantified radiographically as the Tibial Tubercle-Trochlear Groove (TT-TG) distance—is a critical determinant of patellar tracking vectors. An excessive lateralization of the tibial tubercle (typically a TT-TG distance exceeding 20 mm) pathological increases the lateral vector pull on the patella during quadriceps contraction. In such cases, isolated soft-tissue procedures like MPFL reconstruction are destined to fail due to excessive sheer forces, mandating a concomitant bony realignment via a tibial tubercle osteotomy (TTO).
Ligamentous and Retinacular Structures
The primary medial soft-tissue restraints of the patellofemoral joint are the medial retinaculum and, most crucially, the medial patellofemoral ligament (MPFL). Biomechanical studies have definitively proven that the MPFL is the primary checkrein to lateral patellar translation, providing between 50% and 60% of the total restraining force at 0 to 30 degrees of knee flexion. The native MPFL is a distinct fascial band, approximately 15 to 20 mm wide, originating from the superior half of the medial aspect of the patella. It courses obliquely in layer two of the medial knee to insert into the medial epicondylar saddle of the femur, just proximal and posterior to the origin of the superficial medial collateral ligament (sMCL).
The medial retinaculum is a broader, more diffuse fascial layer that reinforces the medial joint capsule, intimately blending with the vastus medialis obliquus (VMO) and the MPFL. While it contributes less absolute tensile strength than the MPFL, it plays a vital role in overall medial capsular stability and proprioception. During a lateral dislocation event, the medial retinaculum often undergoes significant plastic deformation, necessitating plication or imbrication during surgical realignment to restore appropriate capsular tension and volume.
The Vastus Medialis Obliquus (VMO) represents the most distal and oblique muscle fibers of the vastus medialis, inserting directly into the superomedial aspect of the patella. It acts as the primary dynamic medial stabilizer of the joint. Its insertion is typically distal to the superior pole of the patella and anterior to the medial intermuscular septum. Conversely, the lateral retinaculum is composed of dense transverse and oblique fibers, which are often pathologically taut in chronic instability scenarios. This tightness contributes to abnormal lateral patellar tilt and excessive lateral translation, sometimes necessitating a carefully titrated lateral retinacular release or lengthening to balance the soft-tissue envelope.
Neurovascular Risks and Topography
During our medial surgical approach, we must maintain acute situational awareness of vital neurovascular structures to prevent devastating iatrogenic complications. The infrapatellar branch of the saphenous nerve is the most frequently injured structure during medial parapatellar incisions and subcutaneous dissection. This nerve typically courses obliquely from medial to lateral across the anterior aspect of the proximal tibia and medial knee capsule. Careful, layered dissection, avoiding excessive deep retraction, and the utilization of thick subcutaneous flaps are mandatory techniques to protect this nerve, thereby minimizing the risk of postoperative localized numbness, painful neuroma formation, or complex regional pain syndrome.
Deeper dissection, particularly when isolating the anatomical femoral attachment site for the MPFL reconstruction between the medial epicondyle and adductor tubercle, places the medial superior genicular artery at risk. This vessel courses in close proximity to the intermuscular septum and the distal femoral footprint. Inadvertent transection can lead to significant postoperative hematoma, increased pain, and compromised wound healing. Meticulous blunt dissection, precise identification of anatomical landmarks, and preemptive hemostasis using electrocautery are essential to safely navigate this vascular anatomy.
Furthermore, when drilling the femoral tunnel for MPFL graft fixation, the trajectory of the drill pin must be strictly monitored. A tunnel directed too anteriorly risks breaching the trochlear articular cartilage, while a tunnel directed too proximally or distally alters the isometry of the graft, leading to either graft laxity in flexion or catastrophic over-tensioning that can capture the patellofemoral joint and induce rapid-onset osteoarthritis.
Exhaustive Indications and Contraindications
The decision-making algorithm for patellofemoral instability is highly nuanced. Proximal soft-tissue realignment (MPFL reconstruction and/or retinacular plication) is highly successful when applied to the correct patient population, but catastrophic when applied outside of strict parameters. The primary indication is recurrent, symptomatic lateral patellofemoral instability that has failed a comprehensive, minimum 3-to-6-month course of targeted physical therapy.
Patients presenting with an acute, first-time dislocation are generally managed non-operatively, unless specific complicating factors are present. These factors include the presence of a large, displaced osteochondral fracture (often originating from the medial patellar facet or lateral femoral condyle) that requires urgent excision or internal fixation. Additionally, acute surgery may be considered in high-level elite athletes with massive, undeniable structural avulsions of the MPFL off the femoral attachment, though this remains a topic of spirited debate.
Contraindications must be rigorously respected. The most critical contraindication is performing an MPFL reconstruction for isolated anterior knee pain without objective clinical or radiographic evidence of instability. Furthermore, isolated soft-tissue proximal realignment is contraindicated in the presence of severe, uncorrected bony malalignment. If a patient possesses a TT-TG distance greater than 20 mm, severe patella alta (Caton-Deschamps index > 1.2), or high-grade Dejour C or D trochlear dysplasia, an isolated MPFL reconstruction will fail biomechanically. These patients require concomitant bony procedures, such as a tibial tubercle osteotomy or a trochleoplasty, to normalize the osseous architecture before soft-tissue restraints can be expected to function.
| Parameter | Indications for Proximal Realignment (MPFL/Plication) | Absolute & Relative Contraindications |
|---|---|---|
| Clinical Presentation | Recurrent lateral instability; Failed conservative management (PT > 3-6 months). | Isolated patellofemoral pain without objective instability (Absolute). |
| Acute Trauma | First-time dislocation with large, displaced osteochondral fracture requiring fixation. | First-time dislocation without loose bodies or massive avulsions (Relative). |
| Bony Alignment (TT-TG) | Normal to mildly elevated TT-TG distance (< 15-17 mm). | TT-TG distance > 20 mm (Requires concomitant Tibial Tubercle Osteotomy). |
| Patellar Height | Normal patellar height (Caton-Deschamps index 0.8 - 1.2). | Severe Patella Alta (Caton-Deschamps > 1.2) (Requires distalization TTO). |
| Trochlear Morphology | Normal trochlea or mild dysplasia (Dejour Type A). | Severe Trochlear Dysplasia (Dejour Type C/D) (May require Trochleoplasty). |
Pre-Operative Planning, Templating, and Patient Positioning
Effective preoperative planning is the absolute cornerstone of a successful surgical outcome. We must meticulously synthesize every piece of clinical and radiographic information to tailor our surgical approach, ensuring we address all underlying pathoanatomical variables contributing to the patient's instability.
Patient History and Physical Findings
A comprehensive history will elucidate the precise mechanism of injury—differentiating between a high-energy traumatic blow and a low-energy, atraumatic twisting event. We must quantify the frequency of instability episodes, the ease with which the patella subluxates, and the profound impact this has on the patient's activities of daily living and athletic participation. It is critical to screen for generalized hyperlaxity using the Beighton criteria, as hyperlax patients possess a higher baseline risk of recurrent instability and may require more robust graft choices or concomitant procedures.
The physical examination is the most critical diagnostic tool. We must assess for joint-line tenderness, painful popping, and meniscal pathology to rule out concomitant intra-articular derangements. The presence and magnitude of a joint effusion provide clues to the chronicity and severity of the recent instability episode. Patellar stability testing is performed with the knee relaxed in 20 to 30 degrees of flexion to disengage the patella from the trochlear constraint. We assess medial and lateral translation, quantifying it by quadrants of patellar width. Translation of the patella greater than 2 quadrants (50% of its width) laterally, accompanied by a firm lack of an endpoint and severe patient apprehension, confirms gross incompetency of the MPFL and medial retinaculum.
We must also evaluate the dynamic tracking of the patella. We observe for the classic "J sign" during active terminal knee extension. This is a visible, abrupt lateral shift of the patella as it exits the trochlear groove, strongly indicating patellar maltracking and potential trochlear dysplasia or patella alta. Furthermore, assessing the tightness of the lateral retinaculum is crucial; if the patella cannot be translated medially by at least 1 quadrant (approximately 10-12 mm) or cannot be tilted past neutral to the horizontal plane, a concomitant lateral retinacular lengthening or release may be required to prevent excessive lateral tethering post-reconstruction.
Advanced Imaging and Diagnostic Studies
Standard radiographic evaluation is mandatory and includes weight-bearing anteroposterior (AP), true lateral, and Merchant (sunrise) axial views. The true lateral radiograph is critical for assessing patellar height using the Caton-Deschamps or Insall-Salvati indices, and for evaluating trochlear morphology, looking for the "crossing sign," "double contour sign," or a supratrochlear spur indicative of dysplasia. The Merchant view allows for the assessment of patellar tilt, subluxation, and the congruence angle.
Magnetic Resonance Imaging (MRI) is the gold standard for evaluating the soft-tissue envelope. It provides exquisite detail regarding the location and severity of MPFL injury—whether it is an avulsion from the femoral origin, a mid-substance tear, or a patellar insertion failure. MRI is also highly sensitive for detecting classic bone bruising patterns (typically on the anterolateral aspect of the lateral femoral condyle and the inferomedial aspect of the patella) and identifying occult osteochondral lesions or loose bodies that require concurrent arthroscopic management.
Computed Tomography (CT) scans are increasingly utilized for precise, three-dimensional assessment of axial alignment. The CT scan is the most accurate modality for measuring the Tibial Tubercle-Trochlear Groove (TT-TG) distance. By superimposing axial cuts of the deepest portion of the trochlea and the center of the tibial tubercle, we can quantify the lateralization vector. As previously stated, a TT-TG distance exceeding 20 mm is a strong indication for a medializing tibial tubercle osteotomy to normalize the extensor mechanism vector prior to, or in conjunction with, soft-tissue realignment.
Preoperative Setup and Patient Positioning
Prior to formal positioning, a comprehensive Examination Under Anesthesia (EUA) is performed. Once the patient is fully relaxed, we can accurately assess patellar translation without the confounding variable of quadriceps guarding. We assess ligamentous stability across all planes and compare the lateral translation of the patella at 0, 30, and 45 degrees of knee flexion to the contralateral, unaffected knee. This confirms our preoperative diagnosis and dictates the necessary degree of surgical tensioning.
The patient is positioned supine on a standard radiolucent operating table. The operative leg must be kept entirely free, allowing for a full, unencumbered range of motion from hyperextension to maximum flexion throughout the procedure. A well-padded tourniquet is applied to the proximal thigh to ensure a bloodless surgical field during the critical dissection phases. The foot of the bed may be flexed slightly to optimize visualization, though the leg must be able to rest in full extension for isometry testing.
A lateral thigh post is positioned just proximal to the knee joint. This allows the surgeon to apply a valgus moment to the knee, effectively opening the medial compartment and facilitating exposure. The contralateral leg is carefully padded and secured, either flat on the table or in a well-padded stirrup, depending on surgeon preference and the need for fluoroscopic access. The C-arm fluoroscopy unit is positioned on the contralateral side of the table, draped sterilely, and must be capable of obtaining perfect true lateral and AP views of the knee to ensure anatomic placement of the femoral MPFL tunnel.
Step-by-Step Surgical Approach and Fixation Technique
Alright, fellows, the patient is prepped, draped, and the EUA confirms our plan. Scalpel in hand. Let’s execute this masterclass in proximal realignment.
Initial Incision and Dissection
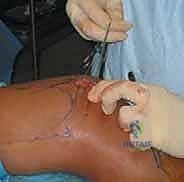
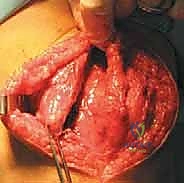
We initiate the procedure with a generous, curvilinear medial parapatellar incision. I center this incision over the widest portion of the patella, extending it proximally approximately 3 to 4 cm above the superior pole and distally 3 to 4 cm below the inferior pole. This specific trajectory provides ample, extensile exposure for both the medial retinacular plication and the patellar fixation phase of the MPFL reconstruction, while avoiding the direct anterior midline where scar tissue can be problematic for kneeling.
Using sharp dissection and electrocautery, we meticulously elevate full-thickness subcutaneous flaps both anteriorly and posteriorly. This is a critical step. By developing broad flaps, we achieve excellent mobility of the prepatellar skin, which minimizes the required length of our external incision and reduces skin tension during closure. More importantly, maintaining a thick, full-thickness flap protects the delicate infrapatellar branch of the saphenous nerve, which arborizes superficially in this region. We keep our dissection strictly superficial to the retinacular fascia until we are ready for the deep arthrotomy.
Once the flaps are adequately developed, we expose the underlying extensor mechanism. You can clearly visualize the quadriceps tendon, the muscular belly of the VMO, the medial retinaculum, and the medial border of the patella. We take a moment to identify the precise insertion of the VMO fibers into the superomedial aspect of the patella, as this dynamic stabilizer must be respected and re-tensioned during our closure.
Medial Retinacular Plication Technique
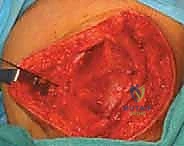
We now proceed to the retinacular plication. I make a longitudinal incision into the medial retinaculum and joint capsule, starting 3 to 4 cm proximal to the superior pole of the patella and extending distally to the level of the inferior pole. Critically, I am making this incision precisely 2 to 3 mm medial to the patellar tendon and the patellar border itself. Leaving this small, robust cuff of tendon and retinaculum attached to the patella is essential; it provides a strong, fibrous anchor point for our imbrication sutures later in the procedure.
We incise through the full depth of the retinaculum and capsule, entering the joint space. We evacuate any hemarthrosis and perform a brief visual inspection of the medial articular facet and trochlea to ensure no loose osteochondral fragments were missed on imaging. Once the joint is cleared, we prepare for the plication. The medial retinacular tissue is often stretched and attenuated. We will use heavy, non-absorbable sutures (e.g., #2 FiberWire or similar high-tensile suture) to imbricate the medial tissue. We pass the sutures through the robust medial edge of the VMO and retinaculum, and then through the 2 mm cuff we left on the patellar side in a pants-over-vest fashion. We leave these sutures untied for now, as they will be tensioned only after the MPFL graft is secured.
MPFL Reconstruction Graft Preparation and Patellar Fixation
Simultaneously, our assistant has prepared the graft on the back table. For a primary MPFL reconstruction, a gracilis or semitendinosus autograft is an excellent choice, providing more than adequate tensile strength (the native MPFL has a yield load of approximately 208 N, which is easily exceeded by a doubled gracilis graft). Alternatively, a high-quality allograft can be utilized to minimize donor-site morbidity. The graft is whipstitched at both ends with high-strength suture and sized appropriately.

We return to the patella for graft fixation. We expose the proximal half of the medial patellar border. Using fluoroscopy to ensure we do not breach the anterior cortex or the articular surface, we drill two parallel transverse tunnels (typically 3.2 mm or 4.5 mm, depending on graft size) into the medial edge of the patella, spaced approximately 10 to 15 mm apart in the proximal half. It is paramount to avoid converging these tunnels or breaching the anterior cortex, as this creates a significant stress riser and risks a catastrophic iatrogenic patellar fracture.
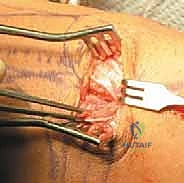
The free ends of the graft are passed into these patellar tunnels and secured. Fixation can be achieved via suspensory buttons on the lateral cortex, radiolucent interference screws within the tunnels, or robust suture anchors if a tunnel-less technique is preferred. Once the patellar fixation is rock-solid, we route the looped end of the graft deep to the medial retinaculum and superficial to the joint capsule, directing it towards the medial epicondyle. This creates an extra-articular, anatomic trajectory for the reconstructed ligament.
Femoral Tunnel Preparation and Graft Tensioning
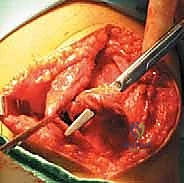
Identifying the anatomic femoral footprint—Schöttle's point—is arguably the most critical step of the entire procedure. We make a small 2 cm incision over the medial epicondyle. Using blunt dissection, we identify the saddle between the adductor tubercle and the medial epicondyle. We place a radiopaque marker (a Beath pin or a specialized guide) at this anticipated point and bring in the C-arm for a perfect, true lateral radiograph.
On the true lateral image (ensuring the posterior condyles are perfectly superimposed), Schöttle's point is located 1 mm anterior to the posterior cortical extension line, 2.5 mm distal to the posterior articular border of the medial femoral condyle, and proximal to the level of the posterior point of Blumensaat's line. Once confirmed radiographically, we drill a guide pin across the femur, directed slightly proximal and anterior to avoid the intercondylar notch. We then ream a socket over the pin to the appropriate depth and diameter for our graft.
We pass the looped end of the graft into the femoral socket using a passing suture. Now, we must establish isometry and tension. We cycle the knee through a full range of motion while holding tension on the graft. The graft should be relatively taut in full extension and early flexion (0 to 30 degrees) and should become slightly lax as the knee flexes past 60 degrees. If the graft tightens excessively in deep flexion, the femoral tunnel is non-anatomic (typically too proximal), and must be repositioned to prevent joint capture and severe stiffness.
Once isometry is confirmed, we fix the graft in the femoral tunnel using a bioabsorbable or PEEK interference screw. **Crucially, the graft is tensioned and fixed with the knee in
