Surgical Pelvic Resection: Gluteus Maximus Muscle & Nerve Preservation
Key Takeaway
Discover the latest medical recommendations for Surgical Pelvic Resection: Gluteus Maximus Muscle & Nerve Preservation. The gluteus maximus muscle is essential for closure in most pelvic resections. It is supplied by the inferior and superior gluteal nerves and arteries, which exit the pelvis at the greater sciatic notch. Protecting these accompanying nerves during surgery is vital to preserve the gluteus maximus muscle and abductor function.
Comprehensive Introduction and Patho-Epidemiology
The pelvic ring represents a formidable anatomic location for the surgical management of primary and metastatic musculoskeletal neoplasms. Surgical resection in the pelvis is inherently more challenging than appendicular skeleton resections due to the complex, three-dimensional osseous anatomy and the intimate proximity of the tumor to vital abdominal viscera, the lumbosacral plexus, and major vascular axes. The dense ligamentous structures and the unique load-bearing requirements of the pelvic girdle further complicate both the extirpation of the malignancy and the subsequent biomechanical reconstruction. Oncologic decision-making regarding the resectability of a pelvic tumor mandates an exhaustive assessment of potential osseous, neurovascular, and visceral involvement.
Osseous resection and subsequent reconstruction are frequently executed adjacent to major peripheral nerves, deep to the iliac vessels, or in direct continuity with the genitourinary or gastrointestinal tracts. Consequently, tumor surgery in the pelvic girdle carries one of the highest rates of perioperative complications, deep surgical site infections, and mechanical reconstruction failures of any anatomic site. The intricate vascular arborization of the pelvis means that achieving wide surgical margins often necessitates the sacrifice of major arterial feeders, rendering the remaining soft tissue envelope highly susceptible to ischemia and subsequent necrosis.
Primary bone sarcomas of the pelvis account for approximately 10% to 15% of all primary malignant bone tumors. Chondrosarcoma is the most prevalent primary pelvic malignancy in adults, frequently presenting as a low-grade, slow-growing mass that can reach massive proportions before clinical detection. Because conventional chondrosarcomas are largely resistant to both chemotherapy and radiation, wide en bloc surgical resection remains the sole curative modality. Osteosarcoma and Ewing sarcoma are more commonly encountered in the pediatric and adolescent populations, demanding aggressive neoadjuvant chemotherapeutic protocols prior to surgical intervention. Furthermore, the pelvis is a highly frequent site for metastatic bone disease, particularly from carcinomas of the breast, prostate, lung, kidney, and thyroid, as well as multiple myeloma.
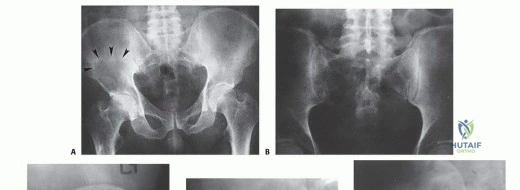
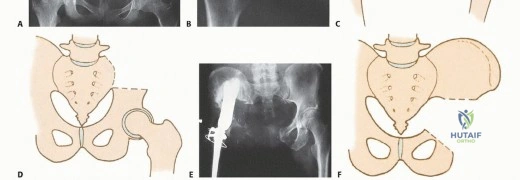
The historical standard of care for malignant pelvic tumors was the hindquarter amputation (hemipelvectomy), a profoundly morbid procedure that leaves the patient with severe functional deficits and profound psychological trauma. However, advancements in advanced cross-sectional imaging, neoadjuvant therapies, and sophisticated reconstructive endoprosthetics have facilitated a paradigm shift toward limb-sparing internal hemipelvectomy. The Enneking and Dunham classification remains the universal vernacular for describing these resections: Type I (ilium), Type II (periacetabular), Type III (pubis and ischium), and Type IV (sacrum). Combinations of these resections (e.g., Type I/II or Type II/III) are frequently required depending on the tumor's epicenter and its intraosseous and extraosseous extension.
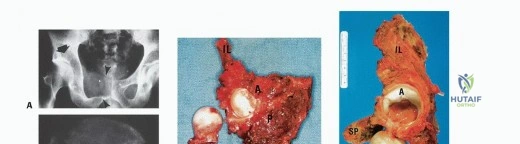
Successful limb salvage is predicated not only on achieving negative oncologic margins (R0) but also on the meticulous preservation of the gluteus maximus muscle and its neurovascular pedicle. The gluteus maximus is the linchpin of pelvic reconstruction, providing critical soft-tissue coverage over massive endoprostheses to mitigate infection and serving as the primary extensor for functional ambulation. The epidemiological distribution of pelvic tumors necessitates a multidisciplinary approach. While benign aggressive lesions such as giant cell tumors of bone or aneurysmal bone cysts may occasionally require extensive resection, the primary indication for internal hemipelvectomy remains high-grade sarcomas. The survival rates for these patients have improved significantly with modern systemic therapies, shifting the surgical focus heavily toward maximizing long-term functional outcomes and minimizing the morbidity associated with massive composite resections.
Detailed Surgical Anatomy and Biomechanics
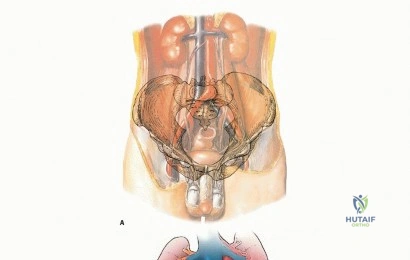
A profound understanding of pelvic anatomy is the cornerstone of safe and oncologically sound resections. The surgeon must navigate multiple anatomic compartments while preserving crucial neurovascular structures and the gluteus maximus muscle, which is paramount for both functional ambulation and robust soft-tissue coverage. The surgical anatomy of the pelvis is unforgiving; a millimeter of deviation during deep dissection can result in catastrophic hemorrhage or irreversible neurological deficit.

Neurovascular Topography and the Lumbosacral Plexus
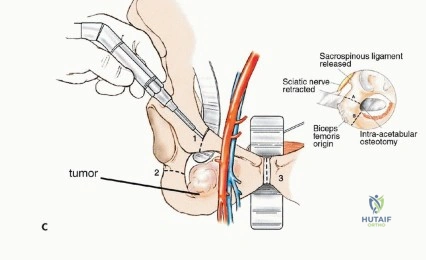
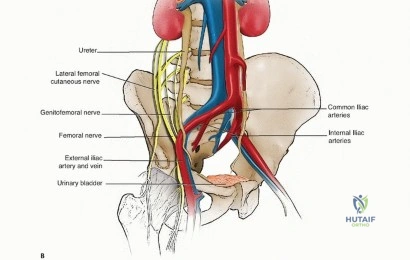
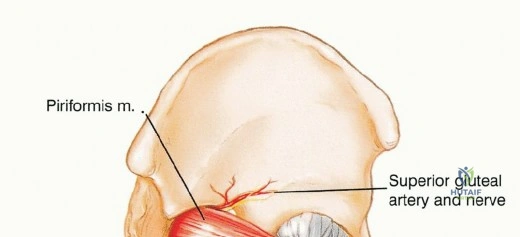
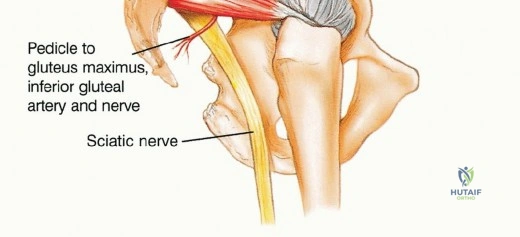
The lumbosacral plexus is intimately associated with the posterior pelvic ring. The sciatic nerve, arising from the ventral rami of L4, L5, S1, S2, and S3, exits the pelvis via the greater sciatic notch, typically coursing inferior to the piriformis muscle before entering the posterior thigh lateral to the ischial tuberosity. In approximately 10% of the population, the nerve or its divisions penetrate the piriformis muscle belly, a critical anatomical variant that must be recognized during the posterior approach to avoid iatrogenic transection. The nerve is accompanied by the inferior gluteal artery, which forms the primary vascular pedicle for the gluteus maximus.
Early identification and protection of the sciatic nerve are imperative. Intrapelvically, it should be isolated distally at the greater sciatic notch and proximally inferior to the psoas muscle. As the nerve traverses the notch, meticulous care must be taken to avoid traction injuries during tumor mobilization. Continuous intraoperative neuromonitoring (somatosensory evoked potentials and spontaneous electromyography) is highly recommended. The superior gluteal nerve (L4, L5, S1) exits the greater sciatic notch superior to the piriformis and supplies the gluteus medius, gluteus minimus, and tensor fasciae latae. In Type I and Type II resections, the superior gluteal nerve is frequently sacrificed to achieve oncologic margins, rendering the hip abductors non-functional and mandating specific reconstructive strategies to prevent profound Trendelenburg gait.
Arterial Supply and Venous Drainage Systems
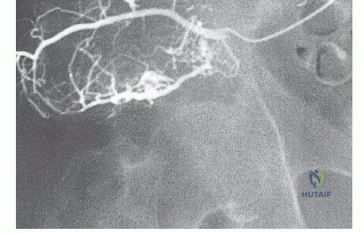
The vascular anatomy of the pelvis is dominated by the common iliac artery and its bifurcation into the external and internal iliac arteries. The internal iliac artery provides the primary blood supply to the pelvic viscera and the gluteal musculature. The superior gluteal artery, the largest branch of the internal iliac, exits the greater sciatic notch superior to the piriformis. The inferior gluteal artery exits inferior to the piriformis, running in close proximity to the sciatic nerve. Venous drainage typically mirrors the arterial supply, but the presacral venous plexus and the internal iliac veins are highly variable, thin-walled, and prone to massive hemorrhage if avulsed.
In the context of gluteus maximus preservation, the inferior gluteal artery is the critical lifeline. When a Type I (iliac) resection is performed, the superior gluteal artery is almost universally sacrificed as it courses over the ilium. Consequently, the inferior gluteal artery becomes the sole vascular supply to the gluteus maximus flap. Iatrogenic injury to this vessel during the posterior approach or sciatic nerve neurolysis will result in catastrophic flap necrosis, exposing the underlying endoprosthesis and virtually guaranteeing a deep space infection.
Anteriorly, the external iliac vessels must be mobilized to access the superior pubic ramus and quadrilateral plate. Surgeons must maintain a high index of suspicion for the corona mortis, an anastomotic connection between the obturator and external iliac (or inferior epigastric) systems, located on the posterior aspect of the superior pubic ramus. Inadvertent avulsion of this vessel can result in massive, difficult-to-control intrapelvic hemorrhage that quickly retracts into the true pelvis, complicating hemostasis.
Myofascial Anatomy of the Gluteus Maximus
The gluteus maximus is the largest and most superficial muscle of the gluteal region, characterized by its coarse fascicular architecture. It originates from the posterior gluteal line of the ilium, the posterior surface of the lower sacrum, the side of the coccyx, the aponeurosis of the erector spinae, the sacrotuberous ligament, and the fascia covering the gluteus medius. It inserts into the iliotibial tract and the gluteal tuberosity of the femur. This extensive origin must be systematically detached during a posterior pelvic approach, often utilizing electrocautery to minimize bleeding from the highly vascularized muscle belly.
In pelvic oncology, the gluteus maximus is elevated as a massive myocutaneous flap. Its preservation is the single most important soft-tissue consideration in limb-sparing internal hemipelvectomy. The muscle provides essential bulk to obliterate the dead space created by the osseous resection, acts as a biological barrier against infection for the metallic reconstruction, and provides powerful hip extension for postoperative ambulation. The deep surface of the muscle must be dissected with extreme caution to preserve the inferior gluteal neurovascular bundle entering its deep central belly.
Biomechanics of the Reconstructed Pelvic Ring
The pelvic ring is a complex biomechanical structure designed to transfer the weight of the axial skeleton from the sacrum to the lower extremities. The posterior sacroiliac complex is the strongest ligamentous structure in the body, responsible for the majority of vertical and rotational stability. Resection of the ilium (Type I) or the periacetabular region (Type II) completely disrupts this load-bearing axis. The biomechanical forces acting upon the pelvis during normal ambulation can exceed three to four times body weight, concentrating immense stress on any reconstructive construct.
Reconstruction must aim to restore the mechanical continuity between the axial skeleton and the femur. Failure to achieve stable fixation results in proximal migration of the femur, leg length discrepancy, and severe mechanical pain. The biomechanical forces across a pelvic reconstruction are immense, subjecting plates, screws, and massive endoprostheses to significant cyclical loading and a high risk of fatigue failure. Modern custom 3D-printed implants utilize highly porous titanium interfaces to encourage osteointegration at the sacral and remaining iliac margins, attempting to transition the construct from a purely mechanical fixation to a biologically stable union.
Exhaustive Indications and Contraindications
The decision to proceed with a limb-sparing internal hemipelvectomy versus a hindquarter amputation is highly nuanced and represents one of the most challenging dilemmas in orthopedic oncology. It requires a delicate balance between achieving definitive oncologic clearance and preserving a functional, sensate limb. The multidisciplinary tumor board must weigh tumor biology, anatomical extent, patient physiology, and expected functional outcomes.
The primary indication for limb salvage is the ability to achieve wide (R0) surgical margins while preserving the major neurovascular structures of the lower extremity—specifically, the sciatic nerve and the external iliac/femoral vessels. If the tumor abuts but does not encase these structures, epineural or adventitial dissection may be permissible in select low-grade tumors or highly responsive high-grade sarcomas following robust neoadjuvant cytoreduction. However, if the tumor encases the sciatic nerve, the nerve must be resected en bloc with the tumor. While sciatic nerve resection is not an absolute contraindication to limb salvage, the resulting flail extremity, insensate plantar surface, and high risk of neuropathic ulceration often render the functional outcome inferior to a well-fitted hemipelvectomy prosthesis.
Contraindications to limb salvage include massive tumor extension into the intrapelvic viscera precluding complete resection, encasement of both the sciatic nerve and the external iliac vessels, and extensive fungating tumors with severe secondary infection. Additionally, patients with severe medical comorbidities who cannot tolerate the physiological insult of a 10-to-14-hour surgery with massive fluid shifts and potential blood loss are better served with a more expeditious amputation. A poorly executed limb salvage that results in local recurrence or chronic, intractable infection is a disservice to the patient and ultimately leads to a delayed, technically more difficult amputation.
Below is an exhaustive summary of the operative versus non-operative (or limb salvage versus amputation) decision-making matrix utilized by advanced orthopedic oncology centers.
| Clinical Variable | Favors Limb Salvage (Internal Hemipelvectomy) | Favors Hindquarter Amputation |
|---|---|---|
| Neurovascular Involvement | Sciatic nerve and external iliac vessels free of tumor | Encasement of both sciatic nerve and major vessels |
| Oncologic Margins | High probability of achieving R0 margins | Inability to achieve negative margins without amputation |
| Soft Tissue Envelope | Adequate gluteus maximus and local flaps available | Massive fungating tumor, skin involvement, radiation necrosis |
| Visceral Extension | Tumor confined to musculoskeletal structures | Massive invasion of bladder, rectum, or major retroperitoneal structures |
| Patient Physiology | Robust health, able to tolerate massive fluid shifts | Severe cardiopulmonary comorbidities, malnutrition |
| Tumor Response | Excellent response to neoadjuvant chemotherapy (>90% necrosis) | Progression of disease during neoadjuvant therapy |
| Expected Function | Preservation of at least partial motor control and plantar sensation | Complete loss of lumbosacral plexus function (insensate, flail limb) |
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning is the bedrock of a successful pelvic resection. The complexity of the pelvic anatomy and the proximity of vital structures demand an uncompromising, multi-modal approach to surgical preparation. The modern orthopedic oncologist relies heavily on advanced imaging, virtual surgical planning (VSP), and patient-specific instrumentation (PSI) to execute these high-stakes procedures.
Advanced Imaging Modalities and 3D Modeling
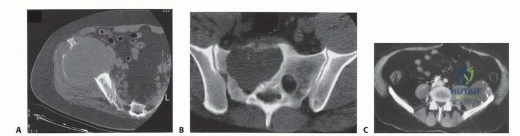
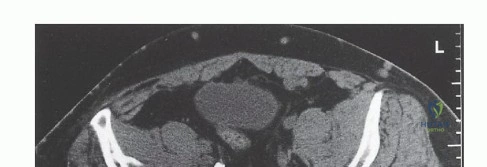
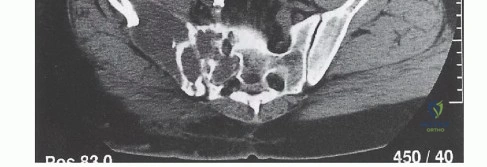
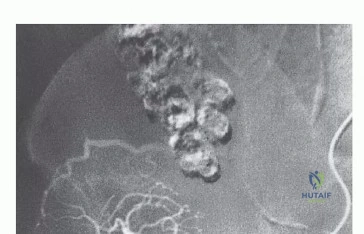
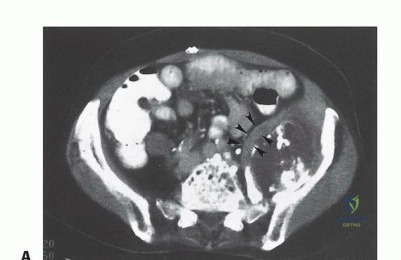
High-resolution, multi-planar Magnetic Resonance Imaging (MRI) is the gold standard for delineating the intraosseous and extraosseous extent of the tumor. T1-weighted images are critical for assessing marrow replacement, while fluid-sensitive sequences (STIR or T2 fat-suppressed) highlight peritumoral edema and soft-tissue mass extension. Computed Tomography (CT) with fine cuts provides unparalleled detail of cortical destruction and is essential for 3D reconstruction. CT angiography or MR angiography is highly recommended to map the vascular displacement caused by the tumor and to definitively identify the patency of the superior and inferior gluteal arteries.
In contemporary practice, the integration of CT and MRI data into virtual surgical planning software allows the surgeon to simulate the resection in a three-dimensional virtual environment. This technology facilitates the design of patient-specific cutting guides that precisely match the patient's osseous anatomy, ensuring that planned margins are executed flawlessly in the operating room. Furthermore, 3D-printed anatomical models can be sterilized and brought into the surgical field, providing a tactile and visual reference that significantly enhances spatial orientation during complex osteotomies.
Neoadjuvant Considerations and Timing
For high-grade sarcomas (e.g., osteosarcoma, Ewing sarcoma), neoadjuvant chemotherapy is standard protocol. The timing of surgery following the completion of a chemotherapy cycle is critical; surgery is typically scheduled 3 to 4 weeks post-chemotherapy to allow for bone marrow recovery and optimization of absolute neutrophil counts, thereby mitigating the risk of catastrophic perioperative infection. If neoadjuvant radiation therapy is employed (often in the setting of soft tissue sarcomas invading the pelvis or specific Ewing sarcoma protocols), the surgeon must be acutely aware of the resulting tissue friability, microvascular thrombosis, and compromised wound healing potential.
Patient Positioning and Surgical Preparation
Patient positioning dictates the surgical approach and the ease of access to the anterior and posterior pelvic compartments. The "floppy lateral" or lateral decubitus position is most commonly utilized for internal hemipelvectomies. The patient is secured on a peg board or bean bag, allowing the surgical table to be tilted to provide access to the anterior ilioinguinal region and the posterior gluteal region without the need for intraoperative repositioning.
Rigorous padding of all bony prominences is mandatory to prevent pressure necrosis and neuropraxia during these marathon procedures. The entire hemipelvis, lower extremity, and lower abdomen are prepped and draped free to allow for full manipulation of the limb. A Foley catheter is essential for monitoring urine output during massive fluid shifts, and central venous access combined with an arterial line is required for invasive hemodynamic monitoring. Blood conservation strategies, including cell salvage (if oncologically permissible) and acute normovolemic hemodilution, should be coordinated with the anesthesia team.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution of a pelvic resection with gluteus maximus preservation is a masterclass in anatomic dissection. The procedure is typically divided into distinct phases: superficial exposure, neurovascular isolation, tumor mobilization, osseous resection, and finally, reconstruction and closure.
Incision and Superficial Dissection
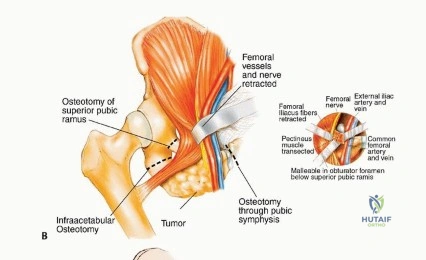
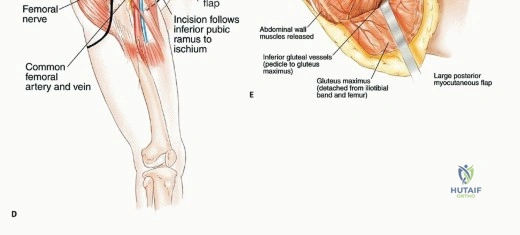
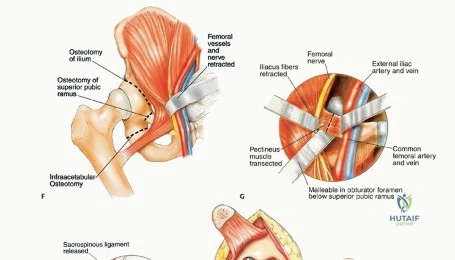
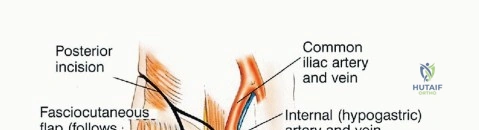
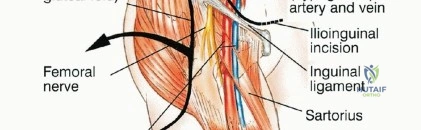
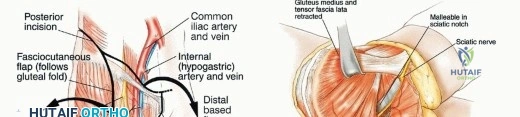
The utilitarian approach to the hemipelvis frequently utilizes a modified "question mark" incision or a triradiate incision. The incision begins at the posterior superior iliac spine (PSIS), extends along the iliac crest to the anterior superior iliac spine (ASIS), and then curves distally along the inguinal ligament or drops vertically down the lateral thigh, depending on the required distal exposure. The skin and subcutaneous tissues are incised down to the fascia lata and the gluteal aponeurosis.
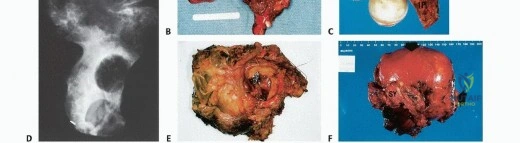
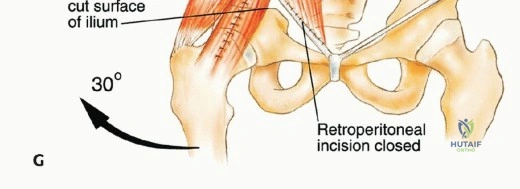
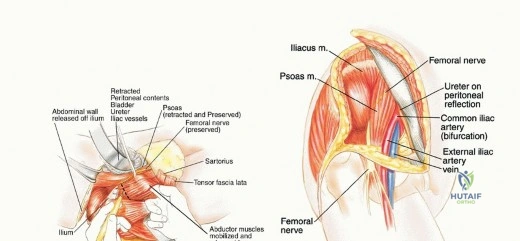
Thick fasciocutaneous flaps are raised to expose the entire gluteus maximus. It is critical to maintain the integrity of the subcutaneous fat attached to the skin to prevent marginal skin necrosis. The anterior dissection involves detaching the abdominal musculature (external oblique, internal oblique, transversus abdominis) from the iliac crest, thereby entering the retroperitoneal space. The iliacus muscle is typically resected en bloc with the tumor if the inner table of the ilium is involved.
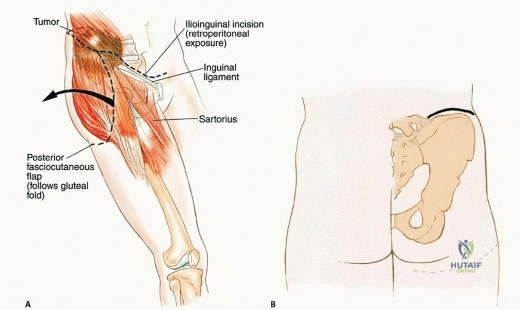
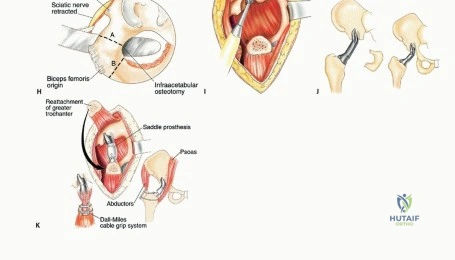
Gluteus Maximus Mobilization and Pedicle Preservation
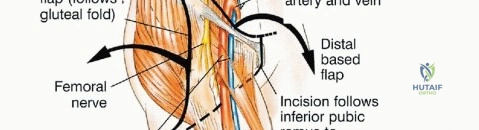
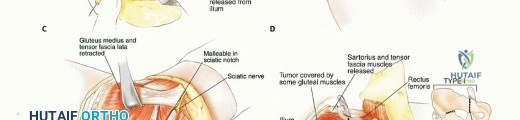
Elevating the gluteus maximus is the most critical step for soft-tissue preservation. The muscle is detached from its femoral insertion at the gluteal tuberosity and the iliotibial band. It is then carefully reflected medially and proximally. The surgeon must sharply dissect the deep fascia of the gluteus maximus off the underlying gluteus medius and piriformis muscles.
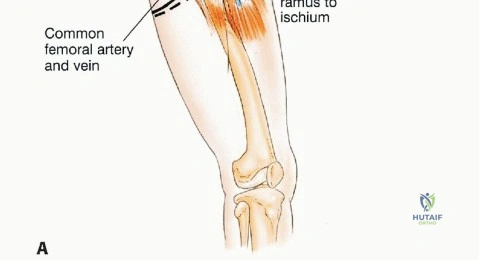
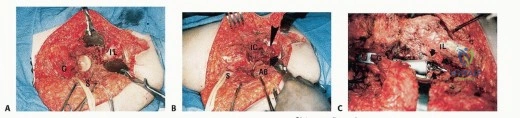
As the dissection approaches the greater sciatic notch, extreme caution is required. The inferior gluteal artery and nerve emerge from the notch inferior to the piriformis and enter the deep, central surface of the gluteus maximus. This neurovascular pedicle must be visually identified, mobilized, and protected throughout the remainder of the procedure. Any traction on the muscle flap must be strictly monitored to avoid avulsing these delicate vessels. Once the pedicle is secured, the sciatic nerve is identified and traced distally into the posterior thigh and proximally into the pelvis, ensuring it is completely free from the tumor mass.
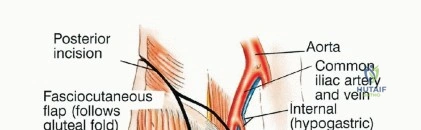
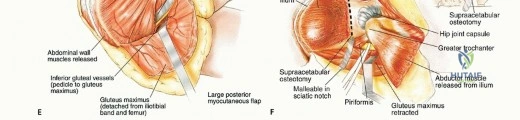
Deep Dissection and Osteotomies
With the neurovascular structures protected, the deep dissection focuses on isolating the osseous margins. Anteriorly, the external iliac vessels and the femoral nerve are mobilized and retracted medially with vascular loops. The pubic symphysis or the superior pubic ramus is exposed for the anterior osteotomy. Posteriorly, the sacroiliac joint or the sacral ala is exposed. The L4 and L5 nerve roots, which form the lumbosacral trunk draped over the sacral ala, must be meticulously dissected and protected.
Osteotomies are performed using a combination of Gigli saws, wide osteotomes, and ultrasonic bone scalpels. The use of patient-specific cutting guides greatly facilitates the accuracy of these cuts. The posterior osteotomy (often through the sacrum or the sacroiliac joint) is particularly treacherous due to the proximity of the presacral venous plexus and the sacral nerve roots. Once all osteotomies are complete, the entire specimen is mobilized en bloc, ensuring that the tumor pseudo-capsule is not breached. The specimen is immediately sent for margin analysis by the pathology team.
Endoprosthetic Reconstruction and Soft Tissue Closure
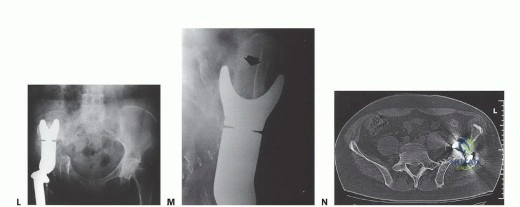
Following oncologic clearance, the reconstructive phase begins. Custom 3D-printed pelvic endoprostheses have largely replaced older saddle prostheses and massive allografts due to superior mechanical stability and lower infection rates. The prosthesis is positioned and secured to the remaining sacrum or lumbar spine using a combination of pedicle screws, massive iliac bolts, and porous titanium flanges designed for osseointegration. The femoral component is then implanted, and the hip joint is reduced, often utilizing a dual-mobility articulation to minimize the risk of postoperative dislocation, which is notoriously high in these patients due to the loss of the abductor mechanism.
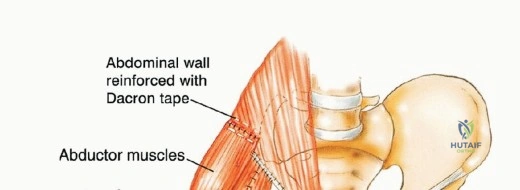
Closure is a formidable task. The preserved gluteus maximus is rotated anteriorly and sutured to the abdominal wall musculature and the remaining anterior structures to completely envelop the endoprosthesis. This muscular coverage is the primary defense against deep infection. Synthetic meshes (e.g., Marlex or Prolene) or biological matrices may be utilized to reinforce the abdominal wall and prevent herniation. Multiple large-bore closed suction drains are placed deep and superficial to the muscle flap. The skin is closed without tension; if tension is excessive, split-thickness skin grafting or additional rotational flaps (e.g., vertical rectus abdominis myocutaneous flap) may be required.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, pelvic resections are plagued by an exceptionally high complication profile. The combination of massive osseous defects, prolonged operative times, extensive dead space, and immunocompromised patients (due to neoadjuvant therapies) creates a perfect storm for perioperative morbidity.
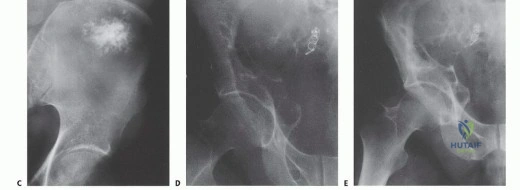
Deep surgical site infection (SSI) is the most dreaded complication, occurring in 20% to 40% of internal hemipelvectomy patients. The presence of a massive metallic endoprosthesis provides an ideal substrate for bacterial biofilm formation. Infections are frequently polymicrobial, involving skin flora (Staphylococcus species) as well as enteric organisms due to the proximity to the perineum. Management of deep SSI requires aggressive surgical debridement, copious pulsatile lavage, and exchange of modular implant components. In severe cases, complete removal of the endoprosthesis and conversion to a flail hip or a delayed amputation may be the only life-saving options.
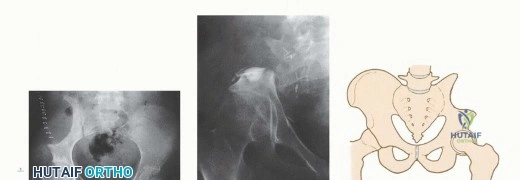
Flap necrosis, particularly of the gluteus maximus, occurs if the inferior gluteal pedicle is compromised or if the skin flaps are closed under excessive tension. Partial necrosis can sometimes be managed with serial debridements and negative pressure wound therapy (NPWT). However, full-thickness necrosis exposing the implant requires immediate coverage with a pedicled or free tissue transfer to salvage the limb. Mechanical failure, including aseptic loosening, screw breakage, and periprosthetic fracture, occurs in up to 30% of patients over a 10-year follow-up period due to the massive cyclical loads placed on
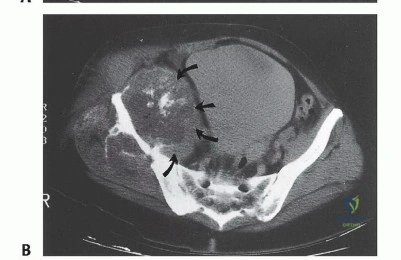
Clinical & Radiographic Imaging Archive