Osteoblastoma of the Distal Femur: A Comprehensive Diagnostic Case Study

Key Takeaway
Diagnosing osteoblastoma, particularly in the distal femur, involves a comprehensive approach. It includes a detailed patient history and clinical examination, followed by advanced imaging (X-ray, CT, MRI, bone scan) to characterize the lesion. A definitive diagnosis is established through image-guided biopsy and histopathological confirmation, crucial for differentiating it from other bone tumors.
Patient Presentation and History
A 22-year-old male presented to the orthopedic oncology and trauma clinic with an 18-month history of progressive right knee pain. Initially described as an insidious, dull, and deep-seated ache, the pain gradually intensified over the preceding six months, becoming continuous and increasingly refractory to over-the-counter analgesic therapy. Unlike the classical presentation of an osteoid osteoma, the patient’s pain profile did not exhibit a profound nocturnal predominance, nor was it consistently or completely relieved by non-steroidal anti-inflammatory drugs (NSAIDs) or salicylates, although transient, partial symptomatic improvement was occasionally noted.
The patient reported a progressive decline in functional capacity, specifically noting increasing difficulty with weight-bearing activities. Prolonged standing, ambulation beyond a quarter-mile, and ascending or descending stairs exacerbated the localized discomfort in the lateral aspect of the distal femur. This mechanical pain component suggests structural compromise or localized periosteal irritation secondary to mass effect.
There was no history of acute macrotrauma, antecedent repetitive microtrauma, or prior surgical interventions to the ipsilateral lower extremity. A comprehensive review of systems was essentially negative; the patient denied any constitutional symptoms such as fever, chills, diaphoresis, night sweats, or unintentional weight loss, thereby lowering the pre-test probability of an infectious etiology (e.g., osteomyelitis) or a highly aggressive, high-grade systemic malignancy (e.g., Ewing sarcoma). He specifically denied any referred pain to the hip or ankle, paresthesias in the distribution of the common peroneal or tibial nerves, and any subjective motor weakness.
His past medical history was unremarkable, with no known drug allergies, bleeding diatheses, or significant medical comorbidities. He was a non-smoker and reported occasional, socially appropriate alcohol consumption. The family history was negative for primary bone tumors, soft tissue sarcomas, or known genetic cancer predisposition syndromes (such as Li-Fraumeni syndrome or hereditary multiple exostoses).
Clinical Examination Findings
Inspection and Palpation
Upon inspection of the right lower extremity in both the standing and supine positions, a subtle, localized fullness was appreciated over the distal metaphysis of the femur, slightly proximal to the lateral femoral condyle. The overlying integument was pristine; there was no visible erythema, ecchymosis, venous engorgement, or trophic skin changes. Muscle bulk appeared symmetrical when compared to the contralateral limb, with quantitative thigh circumference measurements yielding a negligible 0.5 cm deficit on the right, indicating an absence of significant disuse atrophy of the quadriceps musculature.
Palpation of the lateral distal femur revealed a localized, firm, and exquisitely tender area corresponding to the underlying metaphyseal bone. The warmth of the overlying skin was symmetrical to the contralateral limb, lacking the marked calor often associated with aggressive inflammatory or infectious processes. There was no palpable soft tissue mass distinct from the osseous structures, strongly suggesting an intraosseous or juxtacortical lesion rather than a primary soft tissue sarcoma invading the bone.
Range of Motion and Joint Stability
Range of motion (ROM) of the right knee was evaluated and found to be minimally restricted, primarily dictated by pain at the extreme arcs of motion. Active and passive flexion measured 0 to 120 degrees, compared to a full 0 to 140 degrees on the asymptomatic contralateral side. Extension was full and symmetric at 0 degrees, with no extensor lag.
Joint line tenderness was notably absent along both the medial and lateral meniscal intervals. Comprehensive ligamentous stress testing—including varus and valgus stress at 0 and 30 degrees of flexion, the Lachman test, anterior and posterior drawer tests, and the pivot-shift test—demonstrated a stable articulation, effectively ruling out primary ligamentous pathology or intra-articular derangement as the source of the patient's symptomatology. Patellar tracking was centralized, and the patellar apprehension sign was negative.
Neurological and Vascular Assessment
A meticulous neurological examination of the right lower extremity was performed to rule out compressive neuropathy or radiculopathy. Motor strength was graded 5/5 according to the Medical Research Council (MRC) scale in all major muscle groups, including hip flexion (iliopsoas), extension (gluteus maximus), abduction (gluteus medius), knee flexion (hamstrings), knee extension (quadriceps), ankle dorsiflexion (tibialis anterior), plantarflexion (gastrocnemius-soleus complex), and extensor hallucis longus function.
Sensory examination to light touch and pinprick was completely intact across all dermatomes from L2 through S1. Deep tendon reflexes, specifically the patellar (L4) and Achilles (S1) reflexes, were symmetrical and graded 2+ bilaterally. No pathologic reflexes (e.g., Babinski sign or sustained clonus) were elicited.
Vascular assessment confirmed excellent perfusion to the distal extremity. Peripheral pulses, including the femoral, popliteal, dorsalis pedis, and posterior tibial arteries, were strong, bounding, and palpable bilaterally. Capillary refill in the digits of the right foot was brisk (less than 2 seconds). There was no clinical evidence of deep vein thrombosis, and no regional lymphadenopathy was detected in the inguinal or popliteal nodal basins.
Imaging and Diagnostics
Plain Radiography
Initial radiographic evaluation of the right knee, including weight-bearing anteroposterior (AP), lateral, and bilateral skyline views, revealed a well-circumscribed, expansile lytic lesion situated in the distal lateral metaphysis of the femur. The lesion measured approximately 3.5 x 2.8 cm in its maximum orthogonal dimensions. It demonstrated a distinct, continuous sclerotic rim, indicative of a slow-growing, benign biological process allowing the host bone sufficient time to mount a reactive osteoblastic response (Lodwick Grade 1A or 1B).
Mild cortical thickening was apparent along the lateral margin, but there was no overt cortical breach, pathological fracture, or aggressive periosteal reaction (such as a Codman triangle, sunburst, or onion-skinning appearance). No definite matrix mineralization (neither chondroid rings-and-arcs nor osteoid cloud-like densities) was unequivocally observed within the central radiolucency of the lesion on plain films. The tibiofemoral and patellofemoral joint spaces were perfectly preserved, and there were no ancillary findings suggestive of degenerative osteoarthritis or inflammatory arthropathy.

Computed Tomography
A multi-detector Computed Tomography (CT) scan of the distal femur without intravenous contrast was subsequently performed. The primary indications for this modality were to better characterize the lesion's internal bony architecture, meticulously evaluate the integrity of the lateral cortex, and delineate the precise three-dimensional extent of the tumor for preoperative templating.
The CT scan confirmed an intramedullary, eccentric lesion with a well-defined, thick sclerotic border, originating squarely in the metaphyseal region. It demonstrated significant endosteal scalloping and cortical expansion, leading to marked thinning of the lateral femoral cortex; however, the cortex remained continuously intact without frank macroscopic breach. The nidus, highly characteristic of osteoblastoma, was identified as hyperdense relative to surrounding marrow, exhibiting heterogeneous attenuation and subtle, stippled internal calcifications that were not readily apparent on plain radiography. Critically, the CT provided invaluable detail regarding the spatial relationship of the lesion to the closed physis and the articular surface, confirming that the tumor was entirely extra-articular and metaphyseal, thereby informing surgical planning for intralesional curettage and marginal excision.
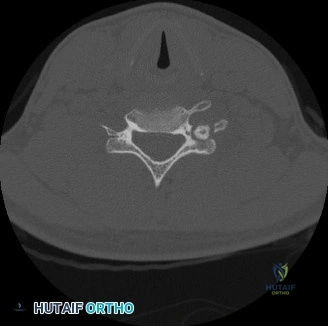
Magnetic Resonance Imaging
Magnetic Resonance Imaging (MRI) of the right knee with and without intravenous gadolinium contrast was obtained to evaluate the extent of intramedullary marrow involvement, assess for any extraosseous soft tissue extension, and further characterize the lesion's tissue composition.
On T1-weighted sequences, the lesion appeared predominantly isointense to hypointense relative to normal skeletal muscle. On T2-weighted and Short Tau Inversion Recovery (STIR) fluid-sensitive sequences, the central nidus demonstrated heterogeneous hyperintensity. Most notably, there was an extensive, disproportionate zone of perilesional bone marrow edema and adjacent soft tissue edema in the vastus lateralis. This extensive inflammatory response, often termed the "flare phenomenon," is a hallmark of osteoblastoma (and osteoid osteoma) and can frequently mimic the aggressive local behavior of a malignant sarcoma or acute osteomyelitis.
Following the administration of intravenous gadolinium, the lesion exhibited intense, homogeneous enhancement of the central nidus, while the surrounding sclerotic rim remained non-enhancing as a dark halo. The adjacent marrow edema also demonstrated diffuse enhancement. The MRI definitively confirmed the absence of soft tissue mass extension beyond the periosteum and verified that the neurovascular bundle in the popliteal fossa was completely uninvolved and anatomically distant from the pathology.
Differential Diagnosis
The presentation of a painful, expansile, lytic metaphyseal bone lesion in a young adult necessitates a rigorous and broad differential diagnosis. The distinction between benign aggressive lesions and low-grade malignancies is paramount for determining the appropriate surgical intervention and avoiding catastrophic undertreatment or unnecessary morbidity.
| Pathology | Radiographic Features | Clinical Presentation | Histological Characteristics |
|---|---|---|---|
| Osteoblastoma | Expansile lytic lesion > 2.0 cm, eccentric, metaphyseal/diaphyseal, variable internal mineralization, surrounding sclerosis. | Progressive, dull ache. Less profound nocturnal pain. Inconsistent or partial relief with NSAIDs. | Interlacing trabeculae of woven bone, prominent osteoblastic rimming, loose fibrovascular stroma. No atypical mitoses. |
| Osteoid Osteoma | Small radiolucent nidus < 2.0 cm, surrounded by a massive zone of reactive cortical sclerosis and cortical thickening. | Intense pain, classically worse at night. Dramatic and rapid relief with NSAIDs or salicylates. | Histologically identical to osteoblastoma but strictly defined by a nidus size of less than 2.0 cm. |
| Aneurysmal Bone Cyst (ABC) | Eccentric, expansile, purely lytic lesion, "soap bubble" appearance, thin cortical shell. Fluid-fluid levels on MRI. | Pain and swelling, often presenting with a pathological fracture. Rapid growth phase. | Blood-filled cystic spaces lacking endothelial lining, separated by fibrous septa containing multinucleated giant cells. |
| Giant Cell Tumor of Bone (GCT) | Eccentric, purely lytic, epiphyseal-metaphyseal location (extends to subchondral bone), non-sclerotic margins. | Pain, swelling, restricted joint motion. Typically occurs after physeal closure. | Mononuclear stromal cells interspersed with numerous, uniformly distributed osteoclast-like multinucleated giant cells. |
| Chondroblastoma | Well-defined, lytic lesion, classically epiphyseal or apophyseal. Chondroid matrix calcifications (rings and arcs). | Joint pain, effusion, restricted ROM. Often presents before skeletal maturity. | Chondroblasts ("cobblestone" appearance), scattered giant cells, "chicken-wire" pericellular calcification. |
| Telangiectatic Osteosarcoma | Destructive, permeative lytic lesion, aggressive periosteal reaction, cortical destruction. Fluid-fluid levels on MRI. | Rapidly progressive pain, swelling, high risk of early pathological fracture. | Highly malignant, pleomorphic, spindle-shaped cells producing disorganized malignant osteoid within blood-filled spaces. |
In this clinical scenario, the lesion's size (3.5 cm) definitively excludes osteoid osteoma. The presence of a thick sclerotic rim and the metaphyseal location make Giant Cell Tumor less likely, as GCTs typically lack a sclerotic margin and invariably abut the articular surface. The lack of fluid-fluid levels on MRI reduces the probability of a primary Aneurysmal Bone Cyst, though secondary ABC-like changes can occur within an osteoblastoma. Ultimately, the radiographic signature of an expansile, mineralizing lesion with a sclerotic halo and extensive perilesional edema in a young adult is highly characteristic of an Enneking Stage 2 (active) Osteoblastoma.
Surgical Decision Making and Classification
Biological Behavior and Classification
Osteoblastomas are classified as benign, bone-forming neoplasms, but they exhibit a spectrum of biological behavior. Utilizing the Enneking Staging System for benign bone tumors, this lesion is classified as a Stage 2 (active) tumor. Stage 2 lesions demonstrate progressive growth, cause symptomatic pain, and expand the host bone, but they remain contained within a reactive osseous shell or the periosteum. They do not typically exhibit the destructive, permeative growth patterns of Stage 3 (aggressive) benign tumors, nor do they metastasize.
Rationale for Operative Intervention
Non-operative management or observation is contraindicated in this scenario for several critical reasons. First, the patient is experiencing progressive, debilitating pain that is refractory to conservative pharmacological measures, significantly impairing his quality of life and functional independence.
Second, the structural integrity of the distal lateral femur is compromised. The lesion has caused substantial endosteal scalloping and cortical thinning. The distal femur is subjected to immense biomechanical forces, including high axial compressive loads during the stance phase of gait and significant tensile and torsional forces generated by the pull of the vastus lateralis and the iliotibial band. A 3.5 cm defect in this critical load-bearing metaphysis places the patient at an unacceptably high risk for a catastrophic pathological fracture, which would drastically complicate subsequent management and compromise the articular surface.
Third, while the clinical and radiographic picture strongly favors a benign osteoblastoma, a definitive histopathological diagnosis is absolutely mandatory. Aggressive variants of osteoblastoma (epithelioid osteoblastoma) can locally mimic low-grade osteosarcoma. Tissue diagnosis via surgical extirpation is required to definitively rule out malignancy.
Treatment Options and Selection
Treatment options for osteoblastoma include radiofrequency ablation (RFA), intralesional curettage with bone grafting, and en bloc marginal or wide resection.
While RFA is the gold standard for osteoid osteoma, its utility in osteoblastoma is limited by the larger size of the lesion (>2 cm). Achieving complete thermal necrosis in a 3.5 cm lesion is technically challenging and carries a high risk of incomplete ablation, leading to recurrence, or excessive thermal damage to adjacent normal bone, leading to structural failure.
En bloc wide resection would guarantee tumor removal but is excessively morbid for a benign lesion, requiring complex reconstructive procedures (such as an osteoarticular allograft or a distal femoral replacement endoprosthesis) and resulting in significant, permanent functional impairment.
Therefore, the gold standard and selected surgical strategy for this patient is aggressive intralesional curettage, supplemented by high-speed burring and local chemical or thermal adjuvants, followed by structural reconstruction of the osseous defect and prophylactic internal fixation. This approach balances the need for complete tumor extirpation to minimize recurrence (which can be 10-20% in osteoblastomas) with the preservation of native host bone and joint biomechanics.
Surgical Technique and Intervention
Patient Positioning and Preparation
The patient was brought to the operating theater and placed in the supine position on a radiolucent Jackson flat table. Following the induction of general endotracheal anesthesia, a prophylactic dose of intravenous cefazolin was administered. A non-sterile tourniquet was applied to the proximal right thigh. The right lower extremity was meticulously prepped and draped in the standard sterile orthopedic fashion, allowing for full, unencumbered manipulation of the knee joint. The limb was exsanguinated using an Esmarch bandage, and the tourniquet was inflated to 250 mmHg.
Surgical Approach
A direct lateral approach to the distal femur was utilized. A 10 cm longitudinal incision was made centered over the lateral femoral epicondyle, extending proximally along the axis of the femoral shaft. Subcutaneous tissues were sharply divided in line with the skin incision to expose the iliotibial (IT) band.
The IT band was incised longitudinally. The vastus lateralis muscle was identified and carefully elevated anteriorly off the lateral intermuscular septum. Perforating branches from the profunda femoris artery and the superior lateral genicular artery were meticulously identified, ligated, and electrocoagulated to maintain a bloodless surgical field. The vastus lateralis was retracted anteriorly using Hohmann retractors, exposing the lateral cortex of the distal femoral metaphysis.
Tumor Extirpation and Adjuvant Therapy
Intraoperative fluoroscopy was utilized to precisely localize the lesion. A rectangular cortical window, measuring approximately 4 cm by 3 cm, was mapped out over the thinned lateral cortex. The corners of the window were pre-drilled with a 3.2 mm drill bit to prevent stress risers and iatrogenic propagation of fracture lines. The cortical window was then carefully elevated using a sharp osteotome, revealing a hypervascular, gritty, reddish-brown intramedullary tumor.
Aggressive intralesional curettage was systematically performed using a series of straight and angled curettes of ascending sizes. The macroscopic tumor was meticulously shelled out from the surrounding sclerotic host bone. Multiple representative tissue specimens were sent fresh to pathology for frozen section analysis, which confirmed a benign, bone-forming lesion consistent with osteoblastoma, with no evidence of high-grade sarcoma.
Following macroscopic clearance, a high-speed pneumatic burr with a spherical diamond tip was employed to aggressively extend the margins of the curettage. The entire cavity was burred down 1 to 2 mm into normal, bleeding cancellous bone to eradicate any microscopic disease residing in the surrounding trabeculae.
To further reduce the risk of local recurrence, local adjuvant therapy was applied. The cavity was thoroughly irrigated with pulsatile lavage using normal saline. Subsequently, 89% phenol was carefully applied to the walls of the cavity using cotton-tipped applicators. The phenol was left in situ for three cycles of two minutes each, followed immediately by copious irrigation with 70% isopropyl alcohol to neutralize the phenol and prevent collateral chemical necrosis of adjacent healthy tissues.
Reconstruction and Prophylactic Fixation
Following tumor extirpation and adjuvant treatment, a significant cavitary defect remained in the lateral distal femoral metaphysis, severely compromising the structural integrity of the lateral column. To reconstruct the defect and provide immediate biomechanical stability, the cavity was tightly packed with a combination of cancellous allograft chips and a biologically active synthetic calcium phosphate bone void filler.
Given the substantial size of the cortical window and the inherent biomechanical stresses placed on the distal femur during weight-bearing, prophylactic internal fixation was deemed mandatory to prevent a postoperative pathological fracture. A pre-contoured, titanium distal femoral locking compression plate (LCP) was selected. The plate was applied to the lateral cortex, bridging the defect. It was secured proximally to the femoral shaft with three bicortical locking screws and distally in the femoral condyles with four cancellous locking screws, ensuring robust, fixed-angle stability.
The surgical site was copiously irrigated. The tourniquet was deflated, and meticulous hemostasis was achieved. A deep subfascial suction drain was placed. The vastus lateralis was allowed to fall back into anatomical position, and the IT band was repaired using continuous #1 Vicryl sutures. The subcutaneous tissue was closed with 2-0 Vicryl, and the skin was approximated using surgical staples. A sterile, compressive modified Jones dressing was applied.
Post Operative Protocol and Rehabilitation
Phase 1: Immediate Postoperative Period (Weeks 0-2)
The patient was admitted for 24-hour observation and intravenous pain management. The subfascial drain was removed on postoperative day 1, following an output of less than 30 cc over a 12-hour period. Chemical deep vein thrombosis (DVT) prophylaxis was initiated with low-molecular-weight heparin (Enoxaparin 40 mg subcutaneously daily) and continued for 14 days, supplemented by mechanical sequential compression devices while in bed.
Due to the robust nature of the locking plate construct, the patient was permitted toe-touch weight-bearing (TTWB) with the assistance of bilateral axillary crutches immediately postoperatively. Early mobilization is critical to prevent arthrofibrosis; therefore, continuous passive motion (CPM) and active-assisted range of motion exercises for the knee were initiated on postoperative day 1, with a goal of achieving 0 to 90 degrees of flexion prior to discharge.
Phase 2: Early Rehabilitation (Weeks 2-6)
At the two-week follow-up, surgical staples were removed, and the incision demonstrated excellent primary healing. Radiographs confirmed maintenance of hardware position and stable alignment of the bone graft.
The patient was transitioned to partial weight-bearing (50% body weight) and advanced to a structured outpatient physical therapy program. The focus during this phase was on restoring full, symmetrical knee range of motion, patellar mobilization to prevent infrapatellar contracture, and isometric strengthening of the quadriceps and hamstring musculature (e.g., straight leg raises, quad sets).
Phase 3: Advanced Rehabilitation (Weeks 6-12)
At the six-week postoperative mark, clinical examination revealed no pain with palpation over the lateral femur, and radiographs demonstrated early incorporation of the cancellous allograft and no evidence of hardware failure or peri-implant radiolucency.
The patient was cleared to progress to full weight-bearing as tolerated and was weaned off assistive devices. Physical therapy advanced to closed kinetic chain exercises (e.g., leg presses, shallow squats, step-ups) to restore lower extremity proprioception, muscular endurance, and functional strength.
Long-Term Surveillance
Given the known recurrence rate of osteoblastoma, long-term clinical and radiographic surveillance is mandatory. The patient is scheduled for serial orthogonal radiographs of the distal femur at 3 months, 6 months, 12 months, and annually thereafter for a minimum of five years. Any recurrence of the patient's characteristic deep ache or the appearance of new periosteal reaction or progressive lucency on radiographs will prompt immediate evaluation with a contrast-enhanced MRI.
Clinical Pearls and Pitfalls
- Pearl: The "Flare Phenomenon" on MRI—characterized by extensive, disproportionate perilesional bone marrow and soft tissue edema—is a classic hallmark of both osteoid osteoma and osteoblastoma. It must not be misinterpreted as aggressive local invasion or an indicator of a high-grade malignancy (such as osteosarcoma) or acute osteomyelitis.
- Pearl: Size is the primary differentiating factor between osteoid osteoma and osteoblastoma. Lesions with a nidus greater than 2.0 cm are classified as osteoblastomas. Furthermore, osteoblastomas typically exhibit a less predictable response to NSAIDs and lack the classic, severe nocturnal pain profile of osteoid osteomas.
- Pearl: The absolute necessity of the high-speed burr cannot be overstated. Simple curettage leaves microscopic tumor cells within the irregular trabecular bone of the cavity walls, leading to unacceptably high recurrence rates. Burring extends the margin by 1-2 mm, transforming an intralesional procedure into a marginal excision.
- Pitfall: Failing to prophylactically stabilize a large, mechanically critical defect in a weight-bearing bone. A large cortical window in the distal femoral metaphysis significantly alters the torsional and compressive strength of the bone. Relying solely on bone graft or cement without load-sharing hardware (like a locking plate) exposes the patient to a severe risk of a catastrophic postoperative pathological fracture.
- Pitfall: Proceeding with definitive surgical ablation or resection without obtaining a definitive tissue diagnosis. While imaging may be highly suggestive of osteoblast
