Deep Posterior Thigh Liposarcoma: An Orthopedic Oncology Case Study & Diagnostic Approach
Key Takeaway
Diagnosing deep posterior thigh liposarcoma (ALT) involves patient history of an enlarging painless mass and clinical exam of a firm, deep tumor. MRI is critical, revealing a fatty mass with thick enhancing septations and non-lipomatous nodules, vital for distinguishing it from benign lipomas and guiding treatment.
Patient Presentation and History
We present the case of Mr. J.A., a 62-year-old male, generally fit and active, who presented to our orthopedic oncology clinic with a chief complaint of a slowly enlarging, painless mass in his right posterior thigh. The insidious nature of soft tissue masses often leads to delayed presentation, and this case was no exception. He first noticed the mass approximately 18 months prior to presentation, describing it initially as a small, deep-seated lump that he attributed to a minor muscle strain during recreational golf, despite denying any specific acute traumatic event.
Over the preceding six months, he reported a more noticeable and exponential increase in size. This rapid growth phase was accompanied by a subtle feeling of pressure and fullness within the posterior compartment, particularly noticeable when sitting on hard surfaces or driving for extended periods. Crucially, he denied any significant resting pain, radicular pain, paraesthesia, or motor weakness in the distal extremity. The absence of neurological symptoms initially suggested that the mass, while expanding, was either displacing rather than invading the sciatic nerve or originating from a location anatomically distinct from the immediate perineural sheath.
His past medical history was significant for well-controlled essential hypertension and hyperlipidemia, managed effectively with an Angiotensin-Converting Enzyme inhibitor and a statin, respectively. He had no history of prior malignancies, prior radiation therapy, or significant surgical interventions. A comprehensive review of systems was negative for systemic B-symptoms; he reported no unintentional weight loss, night sweats, or unexplained fevers.
His family history was meticulously reviewed and found to be non-contributory for soft tissue sarcomas, bone sarcomas, or known genetic syndromes predisposing to malignancy, such as Li-Fraumeni syndrome, Neurofibromatosis type 1, or Familial Adenomatous Polyposis. He was a non-smoker and consumed alcohol occasionally. Mr. J.A. remained functionally independent, maintaining an Eastern Cooperative Oncology Group performance status of 0. His primary concern was the increasing size of the mass and a growing apprehension regarding its underlying etiology, prompting referral from his primary care physician after an initial evaluation.
The presentation of a deep, painless, enlarging soft tissue mass greater than 5 centimeters in an adult must be considered a soft tissue sarcoma until proven otherwise. The historical details, specifically the deep subfascial location and the recent acceleration in growth, raise immediate suspicion for a malignant process, necessitating a highly protocolized diagnostic and therapeutic approach.
Clinical Examination Findings
A meticulous physical examination was conducted, focusing on the local tumor characteristics, regional anatomy, and distal neurovascular status. Upon initial inspection of the right lower extremity with the patient in the standing and prone positions, a prominent, asymmetrical bulge was noted in the posteromedial aspect of the right mid-to-distal thigh. The overlying cutaneous tissue appeared entirely normal. There was no evidence of erythema, ecchymosis, discoloration, ulceration, or dilated superficial venous collaterals. The skin demonstrated normal turgor with no evidence of tethering, dimpling, or peau d'orange changes, suggesting the superficial fascial planes and dermal layers were uninvolved. The mass appeared ovoid, with its long axis oriented longitudinally along the anatomical axis of the posterior thigh compartment.
Palpation was performed with the patient prone and the knee slightly flexed to relax the hamstring musculature. This revealed a deep-seated, firm-to-rubbery mass, approximately 10 by 8 centimeters in cross-sectional diameter, situated entirely within the subfascial muscle compartment. The mass felt subtly lobulated and was largely non-tender to deep palpation, though the patient reported a vague sensation of pressure.
Mobility testing is a critical component of the physical examination for soft tissue tumors. The mass exhibited severely limited mobility in the transverse plane, perpendicular to the muscle fibers. Longitudinally, there was slight mobility with passive movement of the knee, firmly suggesting its origin within or intimate association with the muscular or deep fascial planes of the posterior compartment, rather than being a superficially encapsulated subcutaneous lesion. There was no palpable warmth, thrill, or pulsatility to suggest a high-flow vascular malformation or aneurysm.
A comprehensive neurovascular assessment was paramount given the proximity to the sciatic nerve and the superficial femoral/popliteal vessels. Distal arterial pulses, including the femoral, popliteal, dorsalis pedis, and posterior tibial arteries, were all graded as 2+ and symmetrical with the contralateral limb. Capillary refill in the digits was brisk, occurring in under two seconds.
Neurological examination was systematically performed. Sensation to light touch, pinprick, and proprioception was completely intact throughout the right lower extremity, corresponding accurately to the L2 through S1 dermatomes. Motor strength was graded at a full 5/5 across all major muscle groups of the hip, knee, and ankle. Specifically, hip extension (gluteus maximus and hamstrings), knee flexion (hamstrings), ankle dorsiflexion (tibialis anterior), and ankle plantarflexion (gastrocnemius-soleus complex) were tested against maximal resistance and found to be symmetric. The preservation of normal motor and sensory function in the sciatic distribution (both tibial and common peroneal divisions) was a reassuring prognostic sign regarding the potential for nerve-sparing resection. No pathological reflexes, such as a positive Babinski sign, were elicited.
Range of motion assessment revealed full active and passive range of motion of the right hip and knee joints. There was no pain elicited with terminal flexion or extension of the knee or hip, nor was there any mechanical block to motion. Examination of the regional lymphatic basins, specifically the superficial and deep inguinal lymph nodes, revealed no palpable lymphadenopathy. While soft tissue sarcomas typically metastasize hematogenously rather than lymphatically (with notable exceptions such as synovial sarcoma, epithelioid sarcoma, clear cell sarcoma, rhabdomyosarcoma, and angiosarcoma), a thorough nodal examination remains a standard component of the oncologic evaluation.
Given the deep subfascial location, firm consistency, progressive enlargement, and size exceeding 5 centimeters, the clinical picture was highly suspicious for a primary soft tissue sarcoma of the extremity.
Imaging and Diagnostics
The diagnostic algorithm for a suspected soft tissue sarcoma follows a strict sequence to ensure accurate characterization, staging, and biopsy planning without compromising subsequent surgical extirpation.
Plain Radiography and Ultrasonography
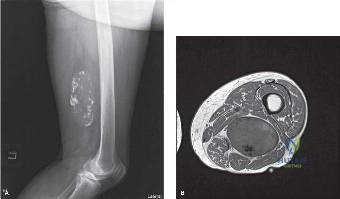
Initial imaging consisted of orthogonal plain radiographs of the right femur and knee. These films demonstrated a large, relatively radiolucent soft tissue mass in the posterior thigh, displacing the surrounding muscle shadows. The radiolucency was consistent with a lesion containing macroscopic fat. Importantly, there was no overt evidence of periosteal reaction, cortical erosion, or direct bony involvement of the adjacent femoral diaphysis. No internal calcifications, phleboliths, or ossifications within the mass were identified, which helped lower the suspicion for entities such as synovial sarcoma, hemangioma, or myositis ossificans. While plain films are rarely diagnostic for soft tissue tumors, they are essential to rule out primary bone tumors with soft tissue extension and to assess for matrix mineralization.
An initial focused ultrasound performed at the referring facility confirmed a large, hyperechoic mass with internal septations. While ultrasound can confirm the solid or cystic nature of a mass and estimate its depth, it is insufficient for surgical planning or definitive characterization of deep soft tissue sarcomas.
Advanced Magnetic Resonance Imaging
The gold standard for local staging and characterization of soft tissue masses is Magnetic Resonance Imaging with and without intravenous gadolinium contrast. MRI provides superior soft tissue contrast, delineates the anatomical relationship of the tumor to critical neurovascular structures, and aids in characterizing the internal architecture of the lesion.

A dedicated MRI of the right thigh was obtained. The imaging protocol included axial, coronal, and sagittal T1-weighted, T2-weighted, and Short Tau Inversion Recovery sequences, followed by fat-suppressed contrast-enhanced T1-weighted images.
The MRI revealed a large, complex, well-circumscribed but lobulated mass centered within the posterior compartment of the right thigh, deep to the fascia lata, measuring 11.2 x 8.5 x 14.0 centimeters. The lesion was primarily located within the substance of the biceps femoris muscle, displacing the semitendinosus and semimembranosus medially.
Signal characteristics were highly informative. The majority of the tumor exhibited high signal intensity on T1-weighted images that suppressed on STIR sequences, confirming the presence of macroscopic adipose tissue. However, unlike a simple benign lipoma, the mass demonstrated significant heterogeneity. There were thickened, irregular internal septations (>2 mm) and distinct nodular areas of non-fatty tissue that were isointense to muscle on T1, hyperintense on T2, and demonstrated avid, heterogeneous enhancement following gadolinium administration.
The sciatic nerve was identified and appeared displaced anteriorly and medially by the mass. While the tumor abutted the epineurium of the sciatic nerve over a length of approximately 5 centimeters, there was a preserved fat plane in some areas, and no definitive encasement or gross invasion of the nerve sheath was visualized. The profound femoral vessels and their perforating branches were identified anterior to the mass, separated by the adductor magnus muscle, with no evidence of vascular invasion. The underlying femoral cortex showed no abnormal marrow signal or cortical breakthrough.
The presence of a predominantly fatty tumor with thick septations and nodular, enhancing, non-lipomatous components is the classic radiological hallmark of an atypical lipomatous tumor or a dedifferentiated liposarcoma.
Systemic Staging Protocols
Given the high suspicion for a malignant soft tissue sarcoma based on MRI findings, systemic staging was immediately initiated. Soft tissue sarcomas of the extremity most commonly metastasize hematogenously to the lungs. Therefore, a high-resolution Computed Tomography scan of the chest without intravenous contrast was performed. The CT chest revealed clear lung fields bilaterally with no evidence of pulmonary nodules, pleural effusions, or mediastinal lymphadenopathy.
For certain histological subtypes, such as myxoid liposarcoma, there is a known propensity for extrapulmonary metastases, particularly to the axial skeleton and abdominal cavity. If the subsequent biopsy were to reveal a myxoid component, a full spine MRI and a CT of the abdomen and pelvis would be mandated. At this juncture, based on the classic imaging appearance of a well-differentiated/dedifferentiated spectrum liposarcoma, staging was limited to the chest.
Image Guided Core Needle Biopsy
The final, and most critical, step in the diagnostic pathway is obtaining tissue for histopathological diagnosis. An incisional biopsy or a poorly planned core needle biopsy can contaminate tissue planes, potentially turning a limb-sparing procedure into an amputation.
A percutaneous, ultrasound-guided core needle biopsy was performed by the musculoskeletal radiology team in direct consultation with the orthopedic oncology service. The biopsy tract was meticulously planned to ensure it fell entirely within the planned future surgical excision ellipse. The needle was introduced through the posterior thigh skin, traversing only the subcutaneous fat and the underlying muscle immediately adjacent to the tumor, avoiding any uninvolved anatomical compartments.
Multiple cores were obtained using a 14-gauge coaxial system, specifically targeting the solid, enhancing, non-fatty nodular components identified on the MRI, as these areas harbor the highest grade of malignancy and dictate the overall prognosis and treatment strategy. Biopsying only the fatty components could lead to a false-negative diagnosis of a simple lipoma or under-grading the lesion as merely an atypical lipomatous tumor. Meticulous hemostasis was achieved to prevent a post-biopsy hematoma, which could spread tumor cells along fascial planes.
Differential Diagnosis
The differential diagnosis for a large, deep, fatty soft tissue mass in the posterior thigh is nuanced. While the presence of fat narrows the differential, the heterogeneous nature of the lesion on MRI demands careful consideration of both benign and malignant entities.
| Diagnosis | Clinical Presentation | Imaging Characteristics (MRI) | Histopathology |
|---|---|---|---|
| Simple Lipoma | Painless, slow-growing mass. Often incidental. Usually superficial, but can be deep (intramuscular). | Homogeneous high T1 signal, suppresses on STIR. Thin septations (<2mm). No nodular enhancement. | Mature adipocytes. No cellular atypia, no lipoblasts. Normal karyotype. |
| Atypical Lipomatous Tumor (ALT) / Well-Differentiated Liposarcoma (WDLPS) | Painless, enlarging mass. Deep subfascial location common. | Predominantly fatty. Thickened, irregular septations (>2mm). Mild to moderate enhancement of septations. | Mature fat with variation in cell size. Hyperchromatic, atypical stromal cells. MDM2 and CDK4 gene amplification. |
| Dedifferentiated Liposarcoma (DDLPS) | Rapidly enlarging mass, often arising within a pre-existing long-standing ALT. | Biphasic appearance. Fatty regions adjacent to prominent, solid, non-fatty, avidly enhancing nodular components. | Transition from WDLPS to high-grade, non-lipogenic sarcoma (often resembling undifferentiated pleomorphic sarcoma). MDM2/CDK4 amplification. |
| Myxoid Liposarcoma | Deep mass, typically in younger adults (30-50 years). Often in the thigh. | Lacks macroscopic fat on T1. High T2 signal (cyst-like due to myxoid matrix). Avid enhancement. "Bowl of fruit" appearance. | Small round/oval cells, prominent myxoid stroma, delicate plexiform "chicken-wire" capillary network. t(12;16) translocation (FUS-DDIT3). |
| Pleomorphic Liposarcoma | Rapidly growing, aggressive mass in older adults. | Highly heterogeneous, necrotic, hemorrhagic. Minimal to no visible macroscopic fat on MRI. | High-grade pleomorphic sarcoma with scattered, pleomorphic lipoblasts. Complex, unbalanced karyotype. |
| Intramuscular Hemangioma | Chronic mass, may fluctuate in size. Painful after exercise. | Heterogeneous. High T1 and T2 signal. Presence of phleboliths (signal voids). Serpiginous vascular channels. | Proliferation of benign vascular channels (capillary, cavernous, or mixed) within skeletal muscle. |
Based on the clinical and radiological findings, the primary working diagnosis was a Dedifferentiated Liposarcoma, given the biphasic nature of the tumor on MRI showing both macroscopic fat and solid enhancing nodules.
Surgical Decision Making and Classification
The management of soft tissue sarcomas requires a highly coordinated, multidisciplinary approach. The decision-making process is dictated by the histopathological diagnosis, the biological grade of the tumor, its anatomical location, and the presence or absence of metastatic disease.
Histopathologic Classification and Molecular Profiling
The core needle biopsy specimens were analyzed by a dedicated soft tissue musculoskeletal pathologist. The histomorphology revealed a biphasic tumor. Areas of the core demonstrated mature adipose tissue interspersed with atypical, hyperchromatic stromal cells and characteristic lipoblasts, consistent with a Well-Differentiated Liposarcoma component. However, other cores taken from the solid nodules showed an abrupt transition to a high-grade, pleomorphic spindle cell proliferation with high mitotic activity and focal areas of tumor necrosis.
To definitively confirm the diagnosis, molecular profiling utilizing Fluorescence In Situ Hybridization was performed. The tumor cells demonstrated high-level amplification of the MDM2 (Murine Double Minute 2) and CDK4 (Cyclin-Dependent Kinase 4) genes located on chromosome 12q13-15. This molecular signature is the diagnostic hallmark of the ALT/WDLPS and Dedifferentiated Liposarcoma spectrum, distinguishing it definitively from pleomorphic liposarcoma or undifferentiated pleomorphic sarcoma.
The final histopathological diagnosis was a Dedifferentiated Liposarcoma. The tumor was graded using the Fédération Nationale des Centres de Lutte Contre le Cancer (FNCLCC) grading system. Based on tumor differentiation (score 3 for dedifferentiated sarcoma), mitotic count (score 2 for 10-19 mitoses per 10 HPF), and tumor necrosis (score 1 for <50% necrosis), the total score was 6, classifying this as a Grade 2 (Intermediate-to-High Grade) soft tissue sarcoma.
Oncologic Staging Systems
Staging was determined using the American Joint Committee on Cancer staging system for Soft Tissue Sarcoma of the Trunk and Extremities.
* T (Tumor): The tumor was greater than 10 cm and deep to the superficial fascia, classifying it as T3.
* N (Node): There was no clinical or radiological evidence of regional lymph node involvement (N0).
* M (Metastasis): Staging CT chest was negative for distant metastasis (M0).
* G (Grade): FNCLCC Grade 2.
Consequently, the patient was staged as Stage IIIB (T3, N0, M0, Grade 2). This stage indicates a large, deep, intermediate-to-high-grade tumor with a significant risk of both local recurrence and distant hematogenous metastasis.
Multidisciplinary Treatment Strategy
The case was presented at our institutional multidisciplinary Musculoskeletal Tumor Board, attended by orthopedic oncologists, medical oncologists, radiation oncologists, musculoskeletal radiologists, and pathologists.
For a large (>5 cm), deep, high-grade soft tissue sarcoma of the extremity, the standard of care involves a combination of surgical resection and radiation therapy. The timing of radiation therapy—neoadjuvant (preoperative) versus adjuvant (postoperative)—was debated.
Neoadjuvant radiation therapy (typically 50 Gy in 25 fractions) offers several distinct oncologic advantages: it treats a smaller target volume, potentially sterilizes the reactive pseudocapsule, thickens the tumor pseudocapsule making surgical resection easier, and requires a lower total dose, thereby reducing long-term tissue fibrosis and joint stiffness. However, as demonstrated by the landmark O'Sullivan trial, neoadjuvant radiation carries a significantly higher risk of major acute postoperative wound complications (up to 35%) compared to adjuvant radiation.
Adjuvant radiation therapy (typically 60-66 Gy) has a lower risk of acute wound dehiscence but is associated with a higher incidence of long-term morbidities, including severe fibrosis, lymphedema, and joint contractures, due to the higher dose and larger treatment field required to cover the entire surgical bed.
Given the tumor's proximity to the sciatic nerve and the desire to maximize local control while potentially downstaging the tumor margins, the consensus recommendation was for Neoadjuvant External Beam Radiation Therapy followed by wide surgical resection. The patient was counseled extensively on the risks of postoperative wound complications associated with this approach. Systemic chemotherapy is generally not considered standard upfront therapy for localized dedifferentiated liposarcoma, as these tumors exhibit relative chemoresistance compared to other subtypes like synovial or Ewing sarcoma, though it may be considered in the metastatic setting or in highly selected high-risk cases.
The patient successfully completed 50 Gy of neoadjuvant radiotherapy over 5 weeks. A restaging MRI performed 4 weeks post-radiation demonstrated a slight decrease in the overall size of the mass, increased internal necrosis, and a thickened, more defined pseudocapsule. The relationship with the sciatic nerve remained unchanged, with close abutment but no gross invasion. Surgery was scheduled for 6 weeks post-completion of radiation to allow for acute radiation-induced tissue inflammation to subside while operating before the onset of dense chronic fibrosis.
Surgical Technique and Intervention
The primary goal of oncologic surgery for soft tissue sarcoma is an R0 resection, defined as microscopically negative margins in all directions. This requires an en bloc resection of the tumor along with a cuff of normal, healthy tissue surrounding it.
Patient Positioning and Preparation
Following the induction of general endotracheal anesthesia, a Foley catheter was placed. The patient was positioned prone on a radiolucent Jackson table with chest and pelvic bolsters to ensure the abdomen hung free, reducing venous pressure and intraoperative bleeding. The bilateral upper extremities were positioned on arm boards in 90 degrees of abduction. All bony prominences were meticulously padded.
A prophylactic dose of intravenous cefazolin was administered 30 minutes prior to skin incision. A non-sterile pneumatic tourniquet was placed high on the proximal right thigh, as close to the gluteal fold as possible, but not inflated. The use of a tourniquet is generally avoided if possible to allow for accurate assessment of tissue perfusion and hemostasis, but it is applied as a rescue measure in the event of catastrophic hemorrhage from the profunda femoris vessels. The entire right lower extremity, from the iliac crest to the toes, was prepped and draped in a standard sterile fashion, allowing for free manipulation of the limb during the procedure.
Surgical Approach and Exposure
The surgical approach was a longitudinal posterior thigh incision. The incision was designed as an ellipse to incorporate the previous core needle biopsy tract, the biopsy entry site on the skin, and any potentially contaminated subcutaneous tissues. The longitudinal incision extended from the subgluteal crease proximally to the apex of the popliteal fossa distally, providing extensile exposure to the entire posterior compartment.
Skin and subcutaneous tissues were incised down to the level of the fascia lata. Full-thickness fasciocutaneous flaps were raised medially and laterally. It is imperative to avoid dissecting into the tumor pseudocapsule; the dissection must remain in normal, uninvolved tissue planes. The posterior cutaneous nerve of the thigh was identified and, due to its intimate association with the superficial aspect of the tumor mass, was sacrificed en bloc with the specimen.
Tumor Extirpation and Margin Control
The deep fascia was incised longitudinally. The hamstrings musculature was identified. The mass was found to be primarily located within the substance of the long head of the biceps femoris. To achieve an adequate margin, the decision was made to resect the involved portion of the biceps femoris muscle from its origin near the ischial tuberosity to its musculotendinous junction distally.
The medial border of the resection involved mobilizing the semitendinosus and semimembranosus muscles. These muscles were retracted medially, exposing the deep aspect of the tumor. The most critical and challenging portion of the dissection was the lateral and deep margin, which abutted the sciatic nerve.
The sciatic nerve was identified proximally, well above the superior pole of the tumor, in pristine, unviolated tissue planes beneath the gluteus maximus. The nerve was carefully traced distally. As anticipated from the preoperative MRI, the tumor pseudocapsule was densely adherent to the epineurium of the sciatic nerve over a distance of approximately 6 centimeters.
In oncologic surgery, when a tumor abuts a critical major motor nerve without directly invading the fascicles, an epimesial dissection (or "peel") is performed. This involves sharply dissecting the outermost layer of the nerve sheath (the epineurium) away from the underlying fascicles, leaving the epineurium attached to the tumor specimen. This constitutes a planned marginal (R1) resection at this specific anatomical location. The rationale is that the combination of this marginal resection and the neoadjuvant radiation therapy provides excellent local control rates while preserving limb function, avoiding the devastating morbidity of a complete sciatic nerve resection.
Using loupe magnification and meticulous microsurgical technique, the epineurium was incised longitudinally adjacent to the tumor. The tumor, along with the adherent epineurium, was sharply peeled away from the intact sciatic nerve fascicles. The tibial and common peroneal divisions of the nerve were preserved in their entirety.
Anteriorly, the dissection proceeded along the posterior aspect of the adductor magnus. The perforating branches of the profunda femoris artery and vein, which supply the posterior compartment and were feeding the tumor, were systematically identified, ligated with non-absorbable silk sutures, and divided.
Once the tumor was completely mobilized circumferentially, it was delivered en bloc from the surgical field. The specimen was oriented with sutures (e.g., short stitch superior, long stitch lateral) and sent to pathology for immediate gross evaluation and subsequent comprehensive margin analysis.
Dead Space Management and Closure
Following tumor extirpation, the surgical bed was copiously irrigated with sterile saline. Meticulous hemostasis was achieved using bipolar electrocautery and argon beam coagulation.
Given the administration of neoadjuvant radiation and the planned marginal resection along the sciatic nerve, the surgical bed was marked with small titanium clips. These clips serve as vital radiographic landmarks for the radiation oncologist should a localized postoperative radiation boost be deemed necessary, or for future surveillance imaging.
The resection of a substantial portion of the biceps femoris created a significant dead space in the posterior thigh. To prevent the formation of a seroma or hematoma, which could serve as a nidus for infection or delay wound healing, two large-bore (19 French) fluted closed-suction drains were placed deep within the resection cavity and brought out through separate stab incisions distally.
Closure of irradiated wounds requires meticulous technique. The deep fascial layer was closed loosely with interrupted heavy absorbable sutures to reduce tension. The subcutaneous layer was closed in multiple layers using 2-0 and 3-0 absorbable sutures, ensuring obliteration of superficial dead space and eversion of the skin edges. The skin was approximated using surgical staples. A sterile, compressive dressing was applied from the toes to the proximal thigh to minimize postoperative edema.
Post Operative Protocol and Rehabilitation
The postoperative management of orthopedic oncology patients is complex, particularly when neoadjuvant radiation has been utilized, as the risk of wound complications is paramount.
Acute Postoperative Management
The patient was transferred to the surgical ward. Neurological checks of the right lower extremity were performed every four hours to ensure the integrity of the sciatic nerve following the epimesial dissection. The patient demonstrated intact motor function in ankle dorsiflexion, plantarflexion, and EHL extension, with no new sensory deficits, confirming the functional preservation of the nerve.
Deep Vein Thrombosis prophylaxis was initiated on postoperative day one with low molecular weight heparin, given the high risk associated with malignancy and major lower extremity surgery. Intravenous antibiotics were continued for 24 hours postoperatively.
Drain management is critical. The closed-suction drains were left in place until the output decreased to less than 30 cc per 24-hour period for two consecutive days. In irradiated fields, lymphatic leakage can be prolonged, and premature drain removal frequently leads to seroma formation and subsequent wound dehiscence. In this case, the drains were removed on postoperative day 8.
Functional Rehabilitation Phases
Rehabilitation
