Grand Rounds: Giant Cell Tumor of Bone – A Detailed Diagnostic Case Study
Key Takeaway
Giant Cell Tumor (GCT) diagnosis involves a comprehensive approach, typically starting with clinical presentation like pain and swelling. Imaging, including X-ray, CT, and MRI, reveals characteristic lytic, expansile lesions and assesses soft tissue extension. Definitive diagnosis relies on histopathological confirmation via biopsy, showing multinucleated osteoclast-like giant cells within mononuclear stromal cells.
Patient Presentation and History
A 28-year-old active male presented to the orthopedic oncology clinic complaining of insidious onset, progressively worsening right knee pain over the preceding six months. The pain was dull, aching in nature, localized to the lateral aspect of the distal femur, and exacerbated by weight-bearing and strenuous activity. He reported nocturnal pain that occasionally disturbed sleep, partially relieved by over-the-counter non-steroidal anti-inflammatory drugs. There was no history of acute trauma or specific mechanism of injury. He denied fevers, chills, night sweats, or unintentional weight loss, effectively lowering the clinical suspicion for highly aggressive systemic malignancies or infectious etiologies at initial presentation.
His past medical history was unremarkable, and he was not on any regular medications. He reported no known family history of bone tumors, soft tissue sarcomas, or endocrine disorders such as hyperparathyroidism, which is critical to note when evaluating lytic bone lesions. Occupational history involved moderate physical activity, which correlated with the mechanical exacerbation of his symptoms. The patient noted that over the last three weeks, the pain had escalated to the point of causing an antalgic gait, prompting his referral to the tertiary care orthopedic oncology service. The transition from purely mechanical pain to nocturnal, rest-associated pain is a hallmark of an expanding intraosseous lesion causing endosteal pressure and cortical micro-fractures.
Clinical Examination Findings
On general examination, the patient was afebrile and hemodynamically stable. Local examination of the right knee revealed a mild, non-tense joint effusion and a subtle fullness in the distal suprapatellar region laterally. There was no overlying skin erythema, warmth, or palpable soft tissue mass, suggesting the lesion remained primarily intraosseous or had only a minimal extraosseous component without massive soft tissue invasion. Tenderness was elicited on deep palpation over the lateral femoral condyle, specifically along the metaphyseal-epiphyseal junction.
Range of motion of the right knee was actively from 0 degrees of extension to 120 degrees of flexion, with terminal flexion causing exacerbation of lateral-sided pain. Passive range of motion was similarly restricted by pain rather than a mechanical block. Patellar tracking was normal, and there were no signs of patellofemoral crepitus. Ligamentous stability testing, including anterior and posterior drawer, Lachman test, and varus and valgus stress at 0 degrees and 30 degrees of flexion, demonstrated no significant laxity. Meniscal tests, including McMurray and Apley grind tests, were negative, effectively ruling out common intra-articular derangements as the primary source of his symptoms.
Neurological assessment revealed intact sensation to light touch and pinprick in the distribution of the femoral, sciatic, superficial peroneal, deep peroneal, and tibial nerves. Motor strength was graded 5/5 in all major muscle groups around the knee and ankle. Distal pulses, specifically the dorsalis pedis and posterior tibial arteries, were bilaterally palpable, bounding, and symmetric. Capillary refill was brisk in all digits. Examination of the regional lymph node basins, specifically the inguinal and popliteal regions, revealed no significant lymphadenopathy.
Imaging and Diagnostics
Initial diagnostic workup commenced with plain radiographs of the right knee, followed by advanced cross-sectional imaging to define the local extent of the disease and plan potential surgical intervention.
Radiographic Evaluation
Anteroposterior and lateral radiographs of the right knee demonstrated an eccentric, purely lytic lesion in the metadiaphyseal region of the distal femur, extending into the subchondral bone of the lateral femoral condyle. The lesion was expansile, with a narrow zone of transition, and showed no significant sclerotic rim, a classic radiographic hallmark of an active benign or low-grade malignant bone tumor. Cortical thinning and a pathological stress fracture line were noted along the lateral cortex. There was no evidence of periosteal reaction, Codman triangle, or internal matrix mineralization (no chondroid rings/arcs or osteoid cloudiness). The epiphyseal plate was noted to be completely fused, consistent with the patient's skeletal maturity.
Figure 1: Initial AP radiograph demonstrating an expansile lytic lesion in the distal femur, extending to the subchondral bone, with cortical thinning and lack of sclerotic margin.
Computed Tomography Assessment
A computed tomography scan of the right femur with intravenous contrast was subsequently obtained to further characterize the bony architecture, evaluate cortical integrity, and assess the true extent of the lesion in three dimensions. CT images confirmed a geographic, purely lytic lesion originating in the lateral femoral condyle, measuring approximately 4.5 x 3.0 x 3.5 cm. The scan highlighted significant cortical thinning and multiple areas of cortical breach, particularly along the lateral and posterior aspects, with an associated subtle extraosseous soft tissue component. The subchondral bone plate was severely effaced, with the tumor extending to within 2 millimeters of the articular cartilage surface. No significant internal calcification or osteoid matrix was evident on the bone windows.
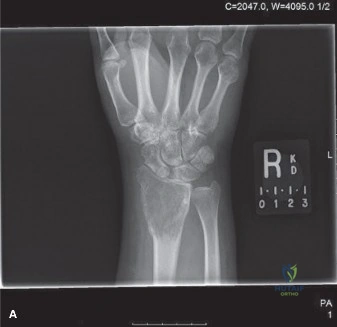
Figure 2: Coronal CT image showing the lytic, expansile nature of the tumor, severe cortical thinning, and subtle cortical breach, indicating local aggression and high risk for impending intra-articular fracture.
Magnetic Resonance Imaging
To fully evaluate the marrow extent, soft tissue involvement, and proximity to the neurovascular bundle, a magnetic resonance imaging scan with and without gadolinium contrast was performed.

Figure 3: Axial T2-weighted MRI demonstrating heterogeneous signal intensity, solid tumor components, and the extent of extraosseous soft tissue expansion laterally, while confirming the neurovascular bundle remains uninvolved posteriorly.
The MRI demonstrated a well-defined lesion that was homogeneously low-to-intermediate signal on T1-weighted sequences and heterogeneously hyperintense on T2-weighted and STIR sequences. Notably, there were focal areas of low signal intensity on T2-weighted images, which typically correspond to areas of hemosiderin deposition or dense fibrosis, a common finding in Giant Cell Tumor of Bone. The scan confirmed the extraosseous extension laterally into the vastus lateralis muscle belly, though a pseudocapsule appeared to contain the soft tissue mass. There were no fluid-fluid levels identified, reducing the likelihood of a secondary aneurysmal bone cyst component. The popliteal neurovascular bundle was clearly separated from the posterior margin of the tumor by a healthy layer of fat and the plantaris muscle origin.
Histopathological Confirmation
Following imaging, an image-guided core needle biopsy was performed via a lateral approach, ensuring the biopsy tract was positioned perfectly within the planned definitive surgical incision. Histopathological analysis revealed a highly cellular lesion characterized by a dual-cell population. There was a proliferation of mononuclear stromal cells interspersed with numerous, uniformly distributed, large multinucleated osteoclast-like giant cells. The background stroma was highly vascular. There was no significant nuclear atypia, abnormal mitoses, or frank sarcomatous stroma to suggest a malignant giant cell tumor or telangiectatic osteosarcoma. Immunohistochemistry was positive for H3F3A G34W mutation, definitively confirming the diagnosis of Giant Cell Tumor of Bone.
Differential Diagnosis
The differential diagnosis for a lytic, epiphyseal/metaphyseal lesion in a skeletally mature young adult is relatively narrow but requires careful distinction due to vastly different treatment paradigms. The primary differentials include Giant Cell Tumor of Bone, Aneurysmal Bone Cyst, Chondroblastoma, Clear Cell Chondrosarcoma, and Telangiectatic Osteosarcoma.
| Diagnosis | Typical Age Group | Typical Location | Radiographic Features | Histologic Hallmarks |
|---|---|---|---|---|
| Giant Cell Tumor of Bone | 20 - 40 years | Epiphyseal/Metaphyseal (extends to subchondral bone) | Eccentric, purely lytic, expansile, narrow zone of transition, no sclerotic rim. | Mononuclear neoplastic stromal cells (H3F3A mutated) with abundant reactive multinucleated giant cells. |
| Aneurysmal Bone Cyst (ABC) | 10 - 20 years | Metaphyseal (can expand into epiphysis) | Expansile, lytic, "soap bubble" appearance, fluid-fluid levels on MRI. | Blood-filled cystic spaces lacking endothelial lining, separated by fibrous septa containing giant cells. |
| Chondroblastoma | 10 - 20 years | Epiphyseal (often prior to physeal closure) | Well-defined lytic lesion, often with a thin sclerotic rim, internal chondroid calcifications. | Chondroblasts ("cobblestone" appearance), pericellular "chicken-wire" calcification, scattered giant cells. |
| Clear Cell Chondrosarcoma | 30 - 50 years | Epiphyseal (proximal femur/humerus predilection) | Lytic lesion, often with distinct margins, may have subtle central calcifications. | Sheets of cells with abundant clear cytoplasm, distinct cytoplasmic membranes, central round nuclei, malignant cartilage. |
| Telangiectatic Osteosarcoma | 15 - 25 years | Metaphyseal | Destructive, permeative lytic lesion, wide zone of transition, cortical destruction. | Blood-filled spaces similar to ABC, but septa contain highly pleomorphic, malignant cells producing osteoid. |
In this case, the patient's age (28), the extreme subchondral location without a sclerotic rim, the lack of fluid-fluid levels on MRI, and the definitive H3F3A G34W mutation on core biopsy firmly established the diagnosis of a primary, benign but locally aggressive Giant Cell Tumor of Bone.
Surgical Decision Making and Classification
Oncologic Staging and Classification
The tumor was classified according to the Campanacci Grading System for Giant Cell Tumors.
* Grade I: Inactive, intraosseous lesions with intact cortices and a sclerotic rim.
* Grade II: Active lesions, expansile, with thinned cortices but no frank cortical breach.
* Grade III: Aggressive lesions with cortical destruction and extraosseous soft tissue extension.
Based on the CT and MRI findings demonstrating lateral cortical breach and subtle extraosseous extension into the vastus lateralis, this lesion was classified as a Campanacci Grade III Giant Cell Tumor. Furthermore, under the Enneking Staging System for benign bone tumors, this represents a Stage 3 (Aggressive) lesion.
Treatment Rationale
The management of Campanacci Grade III GCTB in the distal femur presents a significant reconstructive challenge. The goals of surgery are complete eradication of the macroscopic tumor, prevention of local recurrence, preservation of joint function, and maintenance of the structural integrity of the distal femur.
Historically, En Bloc resection with endoprosthetic reconstruction (distal femoral replacement) was favored for Grade III lesions due to high recurrence rates with simple curettage. However, modern techniques utilizing Extended Intralesional Curettage combined with high-speed burring and local chemical/thermal adjuvants have drastically reduced local recurrence rates to acceptable levels (10-15%), allowing for joint salvage even in cases with minimal subchondral bone stock.
For this patient, despite the cortical breach, the articular cartilage remained intact, and the extraosseous component was well-contained within a pseudocapsule. Therefore, the decision was made to proceed with extended intralesional curettage, adjuvant treatment with hydrogen peroxide and phenol, and reconstruction of the defect utilizing polymethylmethacrylate (PMMA) bone cement augmented with structural internal fixation.
The use of PMMA serves three critical purposes:
1. Thermal Adjuvant: The exothermic reaction during cement polymerization generates local temperatures exceeding 70 degrees Celsius, inducing thermal necrosis of any remaining microscopic tumor cells in the cavity walls.
2. Structural Support: It provides immediate, high-compressive-strength mechanical support to the compromised subchondral bone plate, allowing for early weight-bearing.
3. Surveillance: The radiopaque cement mantle provides an excellent contrast to the surrounding radiolucent host bone. Any future local recurrence will manifest as a progressive radiolucent halo at the cement-bone interface, making radiographic surveillance highly sensitive.
Neoadjuvant Denosumab (a monoclonal antibody against RANKL) was considered. Denosumab inhibits the recruitment and maturation of the osteoclast-like giant cells, leading to rapid ossification of the tumor margin. While highly effective at downstaging tumors, we elected to bypass neoadjuvant Denosumab in this specific case. Denosumab can induce a thickened, "eggshell-like" woven bone rim that traps microscopic neoplastic stromal cells, paradoxically making complete curettage more difficult and potentially increasing the risk of local recurrence if joint salvage is attempted. Given the lesion was deemed resectable via curettage upfront, primary surgery was preferred.
Surgical Technique and Intervention
Patient Positioning and Approach
The patient was brought to the operating room and placed supine on a radiolucent Jackson table. General endotracheal anesthesia was administered. A sterile tourniquet was applied to the proximal right thigh. The right lower extremity was prepped and draped in a standard sterile oncologic fashion. Intravenous prophylactic antibiotics (Cefazolin) were administered prior to tourniquet inflation.
A lateral longitudinal approach to the distal femur was utilized, incorporating the previous core biopsy tract en bloc. The iliotibial band was incised in line with its fibers. The vastus lateralis was elevated anteriorly from the lateral intermuscular septum. The extraosseous component of the tumor, contained within its pseudocapsule, was carefully identified. The pseudocapsule was meticulously dissected from the surrounding healthy muscle, ensuring no tumor spillage.
Cortical Window and Extended Curettage
A large, oval cortical window was created on the lateral aspect of the lateral femoral condyle using a high-speed burr and osteotomes. The window must be large enough to allow direct visualization and instrument access to all recesses of the tumor cavity, particularly the subchondral bone and the posterior condylar recesses.
Gross tumor removal was performed using a series of progressively sized curettes. The macroscopic tumor tissue was friable, highly vascular, and characteristic of GCTB. Once gross tumor was removed, the critical step of extended curettage commenced. A high-speed spherical burr was systematically swept across all bony walls of the cavity. This breaks down the reactive bony ridges and removes an additional 2-3 millimeters of host bone, effectively expanding the margin and eliminating microscopic disease residing in the Haversian canals. Extreme care was taken when burring against the subchondral bone plate to prevent iatrogenic penetration into the knee joint.
Adjuvant Application
Following mechanical burring, the cavity was subjected to chemical adjuvants.
1. Hydrogen Peroxide: The cavity was repeatedly lavaged with 3% hydrogen peroxide. The effervescent action helps to mechanically debride cellular debris from the cancellous bone interstices, while the oxidative stress induces cellular necrosis.
2. Phenol: The surrounding soft tissues were meticulously protected with moist laparotomy sponges. An 89% phenol solution was applied to the bony walls of the cavity using cotton-tipped applicators for three successive cycles of 2 minutes each. Phenol acts as a potent protein coagulant, causing chemical necrosis of residual microscopic tumor cells.
3. Alcohol Wash: The phenol was subsequently neutralized and washed out with copious amounts of 70% isopropyl alcohol, followed by pulsatile lavage with normal saline.
Reconstruction and Fixation
Given the extensive volume of the defect and the proximity to the articular surface, a thin layer of cancellous allograft bone chips was packed against the subchondral plate. This "subchondral bone graft" acts as a biologic buffer, protecting the articular cartilage from the extreme thermal necrosis generated by the curing PMMA cement, which could otherwise lead to premature secondary osteoarthritis.
Following the graft placement, liquid PMMA bone cement (mixed with Tobramycin) was pressurized into the remaining defect. Pressurization ensures interdigitation of the cement into the cancellous bone trabeculae, maximizing mechanical stability and the depth of the thermal adjuvant effect.
Due to the significant cortical destruction, the stress fracture, and the large lateral cortical window, prophylactic internal fixation was mandated to prevent postoperative peri-prosthetic or pathological fracture. A pre-contoured distal femoral locking compression plate was applied to the lateral femur. Locking screws were placed proximally into the diaphyseal bone and distally through the plate, traversing the PMMA cement mantle and anchoring into the medial femoral condyle.
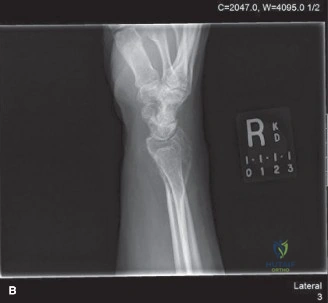
Figure 4: Postoperative AP radiograph demonstrating complete filling of the lytic defect with radiopaque PMMA cement, preservation of the joint space, and robust structural augmentation with a lateral distal femoral locking plate.
The wound was irrigated, a closed suction drain was placed deep to the vastus lateralis, and the soft tissues were closed in layered fashion. The skin was closed with surgical staples. The patient was placed in a hinged knee brace locked in extension.
Post Operative Protocol and Rehabilitation
The immediate postoperative course was uneventful. The intra-articular drain was removed on postoperative day two when output decreased to less than 30 cc per shift. Deep vein thrombosis prophylaxis was initiated with low-molecular-weight heparin (Enoxaparin) for 14 days, given the extensive lower extremity surgery and the patient's transient immobility.
Weight Bearing and Mobility
Because the defect was mechanically reconstructed with high-compressive-strength PMMA and augmented with a robust locking plate construct, the patient was permitted immediate weight-bearing as tolerated on the operative extremity with the use of crutches. The hinged knee brace was unlocked to allow active and active-assisted range of motion from 0 to 90 degrees immediately postoperatively to prevent intra-articular adhesions and arthrofibrosis. Physical therapy focused on quadriceps activation, straight leg raises, and gradual progression of knee flexion. By week six, the patient had achieved full, painless range of motion (0-130 degrees) and was ambulating without assistive devices.
Oncologic Surveillance
Giant Cell Tumor of Bone carries a local recurrence rate of roughly 10-15% following extended curettage and cementation. Furthermore, while technically a benign tumor, GCTB has a known potential for "benign pulmonary metastasis" in approximately 2-4% of cases. These metastases are histologically identical to the primary bone tumor and can be indolent or progressive.
Therefore, a strict, long-term oncologic surveillance protocol was instituted:
* Local Surveillance: Orthogonal radiographs of the right distal femur are obtained at 3 months, 6 months, 9 months, and 12 months postoperatively, then every 6 months for the next 2 years, and annually thereafter for a minimum of 10 years. The primary radiographic sign of recurrence is the development of a progressive, enlarging radiolucent zone measuring greater than 2 millimeters at the bone-cement interface.
* Systemic Surveillance: A non-contrast CT scan of the chest was obtained at the 3-month postoperative mark, followed by annual chest radiographs or low-dose chest CTs for 5 years to monitor for pulmonary metastases.
At the two-year postoperative follow-up, the patient remained completely asymptomatic, with full functional capacity and return to all pre-morbid activities. Serial radiographs demonstrated a stable bone-cement interface with no evidence of progressive radiolucency, and chest imaging remained clear of metastatic disease.
Clinical Pearls and Pitfalls
High-Yield Clinical Pearls
- The "Gold Standard" is Extended Curettage: Simple curettage of a GCTB yields unacceptably high recurrence rates (up to 50%). The use of a high-speed burr to break the reactive bony rim is the single most critical step in minimizing local recurrence.
- Thermal and Chemical Adjuvants: Always utilize adjuvants (Phenol, Hydrogen Peroxide, Argon Beam Coagulation, or Liquid Nitrogen) to address microscopic disease in the cancellous bone walls. PMMA cement acts as a secondary thermal adjuvant.
- Subchondral Protection: When cementing near a joint surface, interposing a layer of cancellous allograft or autograft between the subchondral bone plate and the PMMA cement can protect the articular cartilage from thermal necrosis, reducing the risk of early-onset osteoarthritis.
- Mandatory Chest Imaging: Never omit chest imaging in the workup and surveillance of GCTB. Despite being classified as benign, the 2-4% rate of pulmonary metastasis requires diligent screening.
Critical Pitfalls to Avoid
- Inadequate Cortical Window: Creating a small "keyhole" cortical window prevents adequate visualization of the tumor recesses. If you cannot see the cavity wall, you cannot adequately burr it. Make the window large enough to comfortably accommodate your instruments and line of sight.
- Misinterpreting Biopsy Results: Telangiectatic osteosarcoma and giant cell-rich osteosarcoma can mimic GCTB both radiographically and histologically on a small biopsy sample. Always correlate the pathology with the imaging. The presence of malignant osteoid or severe nuclear atypia must pivot the diagnosis toward sarcoma. The H3F3A mutation is highly specific for true GCTB.
- Over-reliance on Neoadjuvant Denosumab for Salvageable Joints: While Denosumab is excellent for downstaging massive, unresectable tumors or preparing for En Bloc resection, using it prior to planned curettage can be a pitfall. It induces a thick, hard rim of woven bone that traps tumor cells, making extended curettage technically difficult and potentially increasing the risk of local recurrence if the newly formed bone is not aggressively burred away.
- Failure to Prophylactically Fix Large Defects: Large distal femoral defects, especially those with cortical breaches or stress fractures, are at high risk for peri-prosthetic fracture if reconstructed with cement alone. Always consider augmenting massive defects with a locking plate construct to restore the biomechanical integrity of the metadiaphyseal junction.
