Proximal Humerus Cartilaginous Tumors: Navigating the Enchondroma-Chondrosarcoma Dilemma

Key Takeaway
Differentiating enchondroma from low-grade chondrosarcoma in the proximal humerus involves assessing progressive pain, clinical findings, and detailed imaging. Key diagnostic indicators on X-ray and CT include chondroid matrix calcifications and the depth of endosteal scalloping. Significant scalloping (e.g., 3-4mm) with persistent pain strongly suggests potential for low-grade chondrosarcoma.
Introduction and Epidemiology
The proximal humerus is a common anatomical site for the development of primary bone tumors, particularly those of cartilaginous origin. Within this region, the diagnostic differentiation between a benign enchondroma and a low-grade chondrosarcoma represents a significant clinical challenge for orthopedic surgeons and musculoskeletal oncologists. This dilemma is amplified by overlapping radiological features and often subtle histological distinctions, especially in the context of an asymptomatic versus a painful lesion. The case of a 48-year-old male presenting with insidious onset, progressively worsening dull aching pain in the left proximal humerus, exacerbated by overhead activities and eventually disturbing sleep, epitomizes this diagnostic and management conundrum. His absence of trauma, constitutional symptoms, or prior radiation exposure, coupled with localized swelling, directs immediate clinical suspicion toward an underlying neoplastic process.
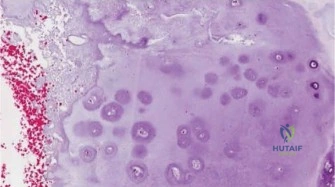
Enchondromas are benign cartilaginous tumors, representing one of the most common primary bone tumors. They are typically asymptomatic, discovered incidentally, and characterized by mature hyaline cartilage within the medullary cavity. The proximal humerus, along with the femur and small bones of the hands and feet, is a predilection site. While generally solitary, enchondromas can occur as multiple lesions in genetic syndromes such as Ollier disease or Maffucci syndrome, which carry a significantly higher risk of malignant transformation.
Chondrosarcomas, conversely, are malignant cartilaginous tumors and are the second most common primary bone malignancy after osteosarcoma. They are classified into conventional, dedifferentiated, mesenchymal, and clear cell subtypes, with conventional chondrosarcomas further graded I, II, or III based on cellularity, nuclear atypia, and mitotic activity. Low-grade (Grade I) chondrosarcomas, particularly central chondrosarcomas, pose the greatest diagnostic challenge due to their close resemblance to enchondromas, both radiographically and histologically. The World Health Organization classification now frequently categorizes appendicular Grade I chondrosarcomas as Atypical Cartilaginous Tumors to reflect their locally aggressive nature but negligible metastatic potential. The incidence of malignant transformation of a solitary enchondroma into a secondary chondrosarcoma is estimated to be less than 1 percent, but this risk increases substantially in syndromic cases. De novo primary low-grade chondrosarcomas, though rare, must always be considered in adults with a symptomatic cartilaginous lesion in the proximal humerus.
The insidious onset and progression of pain in an adult with a proximal humerus lesion should be considered a red flag, prompting a thorough diagnostic workup to differentiate benign from malignant processes. The management strategy for these lesions diverges significantly based on the definitive diagnosis, ranging from observation for asymptomatic enchondromas to aggressive surgical resection for chondrosarcomas.
Surgical Anatomy and Biomechanics
A comprehensive understanding of the surgical anatomy of the proximal humerus and its biomechanical properties is paramount for accurate diagnosis, meticulous surgical planning, and successful execution of interventions for cartilaginous tumors. The proximal humerus is a complex region comprising the humeral head, anatomical neck, surgical neck, greater tuberosity, and lesser tuberosity.
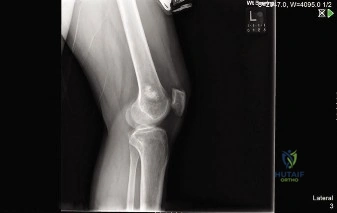
The humeral head, covered by articular cartilage, articulates with the glenoid fossa. Beneath this cartilage lies the cancellous bone, which is the common origin site for enchondromas and chondrosarcomas. The cortex of the proximal humerus, particularly the medial cortex, is critical for weight-bearing and stability. Tumors originating within the medullary canal can progressively thin and erode the cortex (endosteal scalloping), leading to compromised structural integrity and increased risk of pathological fracture.
The greater tuberosity serves as the insertion site for the supraspinatus, infraspinatus, and teres minor tendons (forming the posterior rotator cuff), while the lesser tuberosity serves as the insertion site for the subscapularis tendon. The bicipital groove, housing the long head of the biceps tendon, acts as an anatomical landmark separating the tuberosities. The surgical neck of the humerus is an area of profound biomechanical importance, representing a transition zone between the stiff diaphyseal cortical bone and the more compliant metaphyseal cancellous bone. This transition zone is inherently susceptible to stress concentration.
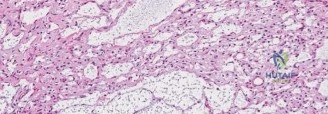
Neurovascular proximity dictates surgical approaches. The axillary nerve and posterior circumflex humeral artery traverse the quadrangular space, wrapping around the surgical neck posteriorly. Any cortical windowing or wide resection in this region mandates absolute protection of these structures. The musculocutaneous nerve, piercing the coracobrachialis, must also be respected during medial retraction.
Biomechanically, the proximal humerus is subjected to significant torsional and bending forces during upper extremity use. A cartilaginous lesion that causes endosteal scalloping exceeding two-thirds of the cortical thickness dramatically reduces the polar moment of inertia, creating a profound stress riser. Torsional strength is proportional to the fourth power of the radius; thus, even minimal endosteal erosion by a slowly expanding Atypical Cartilaginous Tumor exponentially increases the risk of a pathological fracture.
Indications and Contraindications
The decision to operate on a cartilaginous lesion of the proximal humerus hinges upon the differentiation between an actively growing, symptomatic tumor and a static, incidental finding. Pain is the most critical symptom, provided it can be definitively attributed to the tumor and not to concomitant shoulder pathology such as rotator cuff tendinopathy or glenohumeral osteoarthritis.
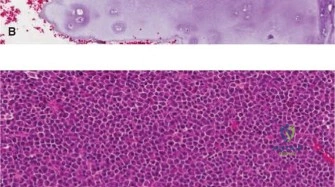
Asymptomatic lesions with classic "rings and arcs" or "popcorn" calcifications, without significant endosteal scalloping or periosteal reaction, are highly indicative of enchondroma and warrant serial observation. Conversely, lesions demonstrating deep scalloping, cortical breakthrough, soft tissue extension, or progressive enlargement require surgical intervention.
| Clinical Scenario | Operative Indications | Non Operative Indications |
|---|---|---|
| Symptomatic Presentation | Pain directly attributable to the lesion, night pain, pathological fracture | Pain clearly originating from alternative pathology (e.g., rotator cuff tear) |
| Radiographic Features | Endosteal scalloping > 2/3 cortical thickness, cortical disruption, soft tissue mass, > 5 cm size | Dense internal calcification, well-defined margins, no scalloping, stable size over time |
| Histological Findings | Biopsy-proven Atypical Cartilaginous Tumor (Grade I), Grade II/III Chondrosarcoma | Biopsy-proven Enchondroma (if biopsy was deemed necessary) |
| Patient Factors | High risk of impending fracture, progressive functional deficit | Poor surgical candidate, severe medical comorbidities, asymptomatic incidentaloma |
Contraindications to extensive surgical resection include profound medical comorbidities precluding anesthesia, or metastatic disease where local control will not alter the overall prognosis. In cases of high-grade chondrosarcoma with massive soft tissue extension encasing the neurovascular bundle, limb salvage may be contraindicated, necessitating forequarter amputation.
Pre Operative Planning and Patient Positioning
Thorough preoperative planning relies heavily on advanced imaging modalities. Plain radiographs in orthogonal planes are the initial step, assessing for the classic chondroid matrix mineralization. Computed Tomography provides superior resolution of cortical integrity, accurately quantifying the depth of endosteal scalloping and identifying subtle cortical breaches.
Magnetic Resonance Imaging is indispensable for evaluating the intramedullary extent of the tumor and any extraosseous soft tissue involvement. Cartilaginous tumors typically exhibit lobulated, high signal intensity on T2-weighted sequences due to their high water content, and intermediate to low signal on T1-weighted images. Dynamic contrast-enhanced MRI can aid in differentiation; enchondromas typically show slow, peripheral enhancement, whereas higher-grade chondrosarcomas may demonstrate rapid, diffuse enhancement due to increased neoangiogenesis.
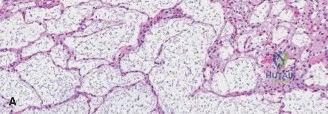
If a biopsy is indicated to rule out high-grade malignancy prior to definitive surgery, it must be performed meticulously. The biopsy tract must be planned so that it can be completely excised during definitive surgical resection. Core needle biopsy is preferred over open incisional biopsy to minimize the risk of tumor seeding.
For surgical intervention, the patient is typically placed in a modified beach chair position. This allows excellent access to the anterior and lateral aspects of the proximal humerus and facilitates intraoperative fluoroscopy. The arm should be draped free to allow for full manipulation, rotation, and assessment of stability during reconstruction. A sterile tourniquet is generally not feasible for proximal humerus lesions, necessitating meticulous hemostasis throughout the procedure.
Detailed Surgical Approach and Technique
The surgical approach is dictated by the tumor grade and the planned procedure. For enchondromas and Atypical Cartilaginous Tumors, extended intralesional curettage with local adjuvant therapy is the gold standard. For Grade II or III chondrosarcomas, wide en bloc resection is mandatory.
Deltopectoral Approach and Exposure
The standard deltopectoral approach provides versatile and extensile access to the proximal humerus. An incision is made starting from the coracoid process, extending distally and laterally along the deltopectoral groove.
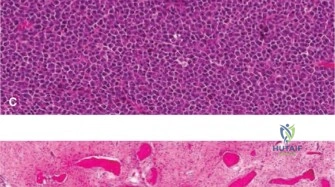
The cephalic vein is identified and typically retracted laterally with the deltoid to preserve its major tributaries, though medial retraction is acceptable if dictated by venous anatomy. The internervous plane between the deltoid (axillary nerve) and the pectoralis major (medial and lateral pectoral nerves) is developed. The clavipectoral fascia is incised lateral to the conjoint tendon. The subscapularis and the anterior circumflex humeral vessels are identified. Depending on the distal extent of the lesion, the proximal insertion of the pectoralis major may be partially released, leaving a cuff for later repair.
Intralesional Curettage and Adjuvant Application
For Atypical Cartilaginous Tumors, a cortical window is created over the lesion. The window should be oval or rectangular with rounded corners to minimize stress risers. The window must be large enough to allow complete visualization of the tumor cavity.
Intralesional curettage is performed systematically. Hand curettes are used to remove the gross macroscopic tumor, which typically appears as glistening, bluish-white, avascular lobules of cartilage. Following gross removal, a high-speed mechanical burr is utilized to extend the curettage 1 to 2 millimeters into normal-appearing cancellous and cortical bone. This mechanical adjuvant is critical for removing microscopic tumor extensions within the trabecular bone.
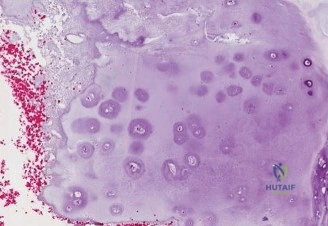
Chemical or physical adjuvants are subsequently applied to the cavity to induce necrosis of any remaining microscopic disease. Phenol (followed by alcohol neutralization), hydrogen peroxide, or liquid nitrogen (cryotherapy) are commonly employed. When using cryotherapy, careful protection of the surrounding soft tissues and neurovascular structures is imperative to prevent iatrogenic thermal injury. Multiple freeze-thaw cycles provide the maximum tumoricidal effect.
Defect Reconstruction
The resulting cavitary defect must be reconstructed to restore biomechanical integrity. Polymethylmethacrylate bone cement is frequently utilized. The exothermic reaction of PMMA polymerization provides an additional thermal adjuvant effect. Furthermore, PMMA provides immediate structural stability, allowing for early postoperative rehabilitation.
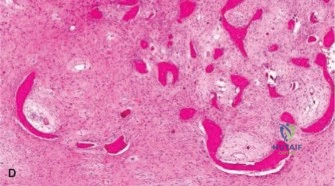
Alternatively, autograft or allograft bone can be used, particularly in younger patients where long-term cement-bone interface issues are a concern. However, bone graft does not provide an adjuvant effect and requires prolonged protected weight-bearing until incorporation occurs. The radiolucency of bone graft can also complicate postoperative surveillance for local recurrence, whereas the radiodense PMMA allows easy identification of new radiolucent osteolysis indicative of recurrence.
Wide Resection and Endoprosthetic Reconstruction
If preoperative biopsy or intraoperative frozen section indicates a high-grade chondrosarcoma (Grade II or III), intralesional procedures are contraindicated due to the unacceptably high risk of local recurrence and metastasis. Wide en bloc resection with negative margins is required.
The resection involves osteotomy of the humeral diaphysis distal to the MRI-defined intramedullary extent of the tumor. The rotator cuff tendons must be transected, and the glenohumeral joint capsule is incised to remove the proximal humerus en bloc. Reconstruction is typically achieved using a modular proximal humeral endoprosthesis. If the rotator cuff and deltoid function are compromised, a reverse total shoulder arthroplasty configuration may be utilized to optimize postoperative functional mechanics. Soft tissue reconstruction, including securing the remaining rotator cuff or conjoint tendon to the prosthesis using synthetic mesh or specialized suture holes, is critical for dynamic stability.
Complications and Management
Surgical management of proximal humerus cartilaginous tumors carries inherent risks, primarily related to the structural compromise of the bone and the proximity of neurovascular structures. The incidence of complications varies significantly between intralesional curettage and wide resection.

Local recurrence is the most concerning oncologic complication. For Atypical Cartilaginous Tumors treated with curettage and adjuvants, the recurrence rate is generally less than 10 percent. Recurrences are typically managed with repeat curettage or wide resection depending on the aggressiveness of the recurrent lesion. Pathological fracture or intraoperative iatrogenic fracture can occur due to the stress riser created by the cortical window or the underlying endosteal scalloping.
| Complication | Estimated Incidence | Prevention and Salvage Strategies |
|---|---|---|
| Local Recurrence | 5 to 15 percent (ACT) | Prevention: Meticulous high-speed burring, use of chemical/thermal adjuvants. Salvage: Repeat curettage, wide resection. |
| Pathological Fracture | 2 to 8 percent | Prevention: Prophylactic stabilization with plates/screws if cortical defect > 50 percent of diameter. Salvage: Open reduction internal fixation with PMMA augmentation. |
| Infection | 1 to 3 percent | Prevention: Strict sterile technique, perioperative antibiotics, meticulous hemostasis. Salvage: Surgical debridement, targeted intravenous antibiotics, hardware removal if necessary. |
| Axillary Nerve Injury | 1 to 2 percent | Prevention: Careful dissection, avoidance of excessive traction on the deltoid, identification during wide resection. Salvage: Observation for neurapraxia, nerve grafting if transected. |
| Prosthetic Instability | 5 to 10 percent (Wide Resection) | Prevention: Meticulous soft tissue repair, appropriate component version, use of reverse geometry if cuff deficient. Salvage: Revision arthroplasty, constrained liners. |
Post Operative Rehabilitation Protocols
Rehabilitation protocols must be tailored to the specific surgical intervention and the method of reconstruction. The primary goals are to protect the surgical construct, restore glenohumeral range of motion, and eventually regain upper extremity strength.
Following intralesional curettage and PMMA cementation, the construct is generally stable immediately. Patients are placed in a sling for comfort but are encouraged to begin passive and active-assisted range of motion exercises within the first postoperative week. Pendulum exercises and supine forward elevation are initiated early to prevent adhesive capsulitis. Active strengthening is typically delayed until 6 weeks postoperatively to allow for soft tissue healing of the deltopectoral interval and any partial tendon releases.
If bone graft is utilized instead of PMMA, the rehabilitation must be significantly slower. The proximal humerus remains mechanically vulnerable until graft incorporation and cortical remodeling occur, which can take 3 to 6 months. During this period, torsional loading and heavy lifting must be strictly avoided.
For patients undergoing wide resection and endoprosthetic reconstruction, rehabilitation is prolonged and complex. The primary concern is protecting the soft tissue reattachments to the prosthesis. Patients are typically immobilized in an abduction sling for 4 to 6 weeks. Passive range of motion is initiated early, but active motion is strictly prohibited until the soft tissues have healed to the implant. The ultimate functional outcome is heavily dependent on the preservation and rehabilitation of the deltoid muscle, especially if the rotator cuff has been resected.
Summary of Key Literature and Guidelines
The management of proximal humerus cartilaginous tumors is guided by evolving literature regarding histological classification, advanced imaging, and surgical techniques. The National Comprehensive Cancer Network guidelines provide a robust framework for the workup and treatment of these lesions.

Recent updates to the World Health Organization classification of soft tissue and bone tumors have been pivotal in reshaping our understanding of low-grade cartilaginous lesions. The reclassification of Grade I chondrosarcoma of the appendicular skeleton as an "Atypical Cartilaginous Tumor" reflects the consensus that these lesions behave in a locally aggressive manner but do not possess the metastatic potential of higher-grade sarcomas. This nomenclature shift supports the utilization of joint-preserving intralesional curettage rather than morbid wide resections for these specific tumors.
Literature regarding the efficacy of various local adjuvants remains a topic of academic debate. Studies comparing phenol, cryotherapy, and argon beam coagulation generally show equivalent oncologic outcomes, emphasizing that meticulous mechanical burring is the most critical step in minimizing local recurrence. The choice of adjuvant is often driven by surgeon preference and institutional availability.
Advanced imaging, particularly dynamic contrast-enhanced MRI, has been heavily investigated as a non-invasive tool to differentiate enchondromas from malignant lesions. Geirnaerdt et al. demonstrated that early and exponential enhancement patterns are highly sensitive for higher-grade chondrosarcomas, whereas enchondromas exhibit slow, gradual enhancement. Furthermore, the identification of IDH1 and IDH2 mutations in a vast majority of central cartilaginous tumors has opened avenues for targeted molecular therapies, although surgical management remains the cornerstone of treatment.
Ultimately, navigating the enchondroma-chondrosarcoma dilemma requires a multidisciplinary approach. Clinical presentation, radiographic findings, and histological analysis must be synthesized to formulate a treatment plan that eradicates the neoplastic process while maximizing upper extremity function and preserving the biomechanical integrity of the proximal humerus. Continuous postoperative surveillance with serial radiographs is mandatory, regardless of the treatment modality, to monitor for local recurrence or hardware failure.
