Sacral Chordoma: An Advanced Orthopedic Case Study on Diagnosis & Imaging
Key Takeaway
Sacral chordoma diagnosis integrates patient history (sacral pain, radiculopathy, bladder/bowel changes), detailed neurological examination revealing deficits, and advanced imaging (lytic lesion on CT, hyperintense "soap bubble" on T2 MRI). Definitive diagnosis relies on CT-guided biopsy confirming pathognomonic physaliferous cells, guiding precise surgical planning and management.
Patient Presentation and History
Demographics and Clinical Onset
A 58-year-old male presented to the orthopedic oncology clinic with an 18-month history of progressively worsening sacral pain. The pain was initially insidious, dull, and localized to the gluteal region, but over the past 6 months, it had become constant, more intense, and radiated bilaterally into the posterior thighs, indicative of S1-S2 radiculopathy. He reported exacerbated pain with prolonged sitting and standing, and minimal relief with maximum recommended doses of non-steroidal anti-inflammatory drugs or acetaminophen. He denied any history of high-energy or low-energy trauma.
The insidious onset of sacral pain is a hallmark of primary sacral tumors, frequently leading to a significant delay in diagnosis. Because the sacral canal is capacious and the presacral space allows for extensive anterior tumor expansion without immediate visceral compression, patients often present with vague lumbopelvic symptoms that are initially misdiagnosed as degenerative lumbar spondylosis or sacroiliac joint dysfunction. In this case, the 18-month delay from symptom onset to orthopedic oncology evaluation is highly typical of sacral chordoma literature.
Neurological and Systemic Review
Of critical importance, the patient had recently developed new-onset constipation and experienced occasional urinary hesitancy, suggesting potential S3-S4 neurological involvement and autonomic dysfunction. The preservation of bowel and bladder function is a primary determinant of postoperative quality of life, making the baseline assessment of these autonomic pathways essential. He reported a 5 kg unintentional weight loss over the past year, attributing it to decreased appetite secondary to chronic, unremitting pain rather than systemic cachexia.
Past medical history was significant for well-controlled hypertension and dyslipidemia. He was a non-smoker and consumed alcohol occasionally. No family history of musculoskeletal malignancies, gastrointestinal carcinomas, or genetic tumor syndromes was reported. The patient’s functional status prior to the exacerbation of symptoms was excellent, rendering him an appropriate candidate for potentially morbid surgical intervention.
Clinical Examination Findings
Inspection and Palpation
On general inspection, the patient appeared in moderate discomfort, adopting a distinctly antalgic gait characterized by a shortened stance phase on the bilateral lower extremities to minimize axial loading through the pelvis. Inspection of the lumbosacral region revealed no obvious skin changes, erythema, venous engorgement, or gross asymmetry.
Palpation of the sacrum elicited significant point tenderness, particularly over the midline and bilateral sacroiliac joints. A digital rectal examination was performed, which is an indispensable component of the physical examination for any suspected sacral pathology. A deep, firm, non-mobile, and rubbery mass was palpable anterior to the sacrum, confirming the suspicion of a substantial presacral component displacing the rectal vault anteriorly. The rectal mucosa felt intact, suggesting the mass was extra-luminal and arising from the presacral space or the sacrum itself.
Neurological Assessment
Range of motion of the lumbar spine was full but painful at the extremes of flexion and extension, likely due to dural tension and mechanical loading of the sacropelvic junction. Hip range of motion was unrestricted, ruling out primary coxofemoral pathology. A comprehensive neurological examination was conducted to map the extent of sacral root compromise:
- Motor Function: Bilateral lower extremity strength was intact (5/5) for hip flexion (L2-L3), knee extension (L3-L4), ankle dorsiflexion (L4-L5), and great toe extension (L5). However, ankle plantarflexion was subtly weakened (4+/5) bilaterally, consistent with early S1 radiculopathy.
- Sensory Function: Light touch and pinprick sensation were diminished in the bilateral S1 and S2 dermatomes along the posterior thigh and calf. Perianal sensation (S3-S5) was significantly reduced bilaterally, correlating directly with the patient's reports of altered bowel and bladder function.
- Reflexes: Patellar reflexes (L3-L4) were 2+ and symmetrical. Achilles reflexes (S1) were diminished (1+) bilaterally.
- Sphincter Tone: Resting rectal tone was reduced, and the bulbocavernosus reflex was completely absent. The loss of the bulbocavernosus reflex indicates disruption of the S2-S4 reflex arc.
These examination findings collectively pointed towards a massive space-occupying lesion affecting the distal sacral nerve roots and the sacral plexus, necessitating immediate advanced cross-sectional imaging.
Imaging and Diagnostics
Plain Radiography
Initial diagnostic workup commenced with standard plain radiographs of the lumbosacral spine and pelvis. Anteroposterior and lateral views of the sacrum demonstrated a lytic, expansile lesion centered in the S2-S4 segments of the sacrum. Cortical destruction was evident, particularly on the lateral view, with an associated soft tissue mass projecting anteriorly. Calcification within the lesion was absent, which, while not pathognomonic, is a less common finding in chordomas compared to chondrosarcomas. The bowel gas pattern was displaced anteriorly, further corroborating the rectal examination findings of a presacral mass.
Computed Tomography and Templating
A high-resolution, multi-detector Computed Tomography scan of the pelvis and lumbosacral spine was subsequently performed to better delineate the bony architecture, tumor extent, and matrix characteristics. The CT revealed a destructive, lytic lesion involving S2 to S4, with significant anterior cortical breach and an extensive anterior sacral soft tissue component. Areas of internal septation and a hypodense mucoid matrix were noted. There was no evidence of overt chondroid or osteoid matrix calcification.
The CT scan provided crucial information regarding the extent of bony involvement, potential for pathological fracture, and proximity to critical structures such as the mesorectal fascia, rectum, and major pelvic vessels (internal iliac artery and vein branches). Based on the CT images, 3D templating was initiated to map the precise osteotomy planes required for an en bloc resection.
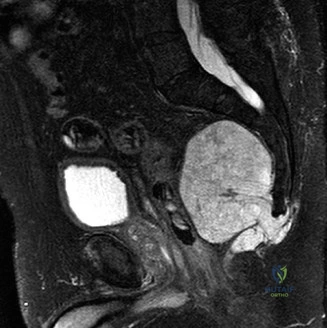
Magnetic Resonance Imaging
To fully characterize the soft tissue extent, epidural involvement, and neural element compromise, a contrast-enhanced Magnetic Resonance Imaging study of the lumbosacral spine and pelvis was obtained. MRI is the gold standard for evaluating sacral tumors.
The MRI demonstrated a large, lobulated mass centered in the lower sacrum. On T1-weighted images, the lesion was isointense to hypointense relative to skeletal muscle. On T2-weighted and STIR sequences, the tumor exhibited striking hyperintensity, characteristic of the high water content found in the myxoid and mucinous matrix of a chordoma. The classic "thumbprint sign" or lobular architecture with low-signal internal septations was clearly visible. Following gadolinium administration, the lesion demonstrated heterogeneous enhancement.
Crucially, the MRI detailed the tumor's relationship with the sacral nerve roots. The S1 roots were displaced superiorly but appeared uninvolved by gross tumor. The S2 roots were encased bilaterally, and the S3-S5 roots were completely obliterated by the mass. The tumor extended anteriorly, pushing against the presacral fascia, but a distinct fat plane was maintained between the tumor capsule and the posterior wall of the rectum, suggesting that a clear anterior margin could be achieved without necessitating a bowel resection.

Biopsy and Histopathology
Following comprehensive imaging, a CT-guided percutaneous core needle biopsy was performed. The biopsy approach is a critical step in orthopedic oncology. The tract was carefully planned via a direct posterior approach through the midline soft tissues that would be excised en bloc during the definitive surgical procedure. Transrectal or transvaginal biopsies are strictly contraindicated as they contaminate the presacral space and compromise the ability to achieve a clean anterior margin, potentially converting a resectable tumor into an unresectable one.
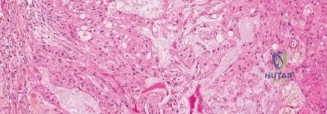
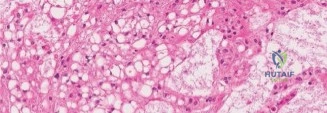
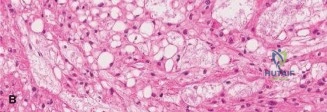
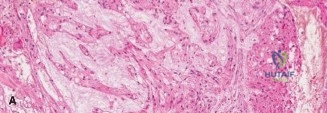
Histopathological analysis revealed lobules of tumor cells separated by fibrous septa. The cells possessed abundant, vacuolated cytoplasm, classically described as physaliferous cells, set within a myxoid stroma. Immunohistochemistry was strongly positive for cytokeratin, epithelial membrane antigen, S-100 protein, and, most definitively, brachyury. Brachyury is a transcription factor crucial for notochordal development and is highly sensitive and specific for chordoma, confirming the diagnosis.
Differential Diagnosis
The differential diagnosis for a destructive sacral lesion in an adult is broad and encompasses primary benign, primary malignant, and metastatic etiologies. A systematic approach utilizing patient demographics, radiographic appearance, and clinical presentation is required to narrow the differential prior to biopsy.
Sacral Lesion Comparative Analysis
| Pathology | Radiographic Appearance | Clinical Hallmarks | Histology and Markers |
|---|---|---|---|
| Chordoma | Midline, lytic, expansile, large anterior soft tissue mass. T2 hyperintense with lobules and septations. | Most common primary sacral malignancy. Insidious onset, bowel/bladder dysfunction. Arises from notochordal remnants. | Physaliferous cells in myxoid stroma. Brachyury positive, Cytokeratin positive, S-100 positive. |
| Giant Cell Tumor | Eccentric or midline, purely lytic, expansile, lacks sclerotic rim. "Soap bubble" appearance. | Usually younger demographic (20-40 years). Painful. Can cross sacroiliac joint. | Multinucleated giant cells uniformly distributed among mononuclear stromal cells. H3F3A mutation positive. |
| Chondrosarcoma | Lytic lesion with characteristic "rings and arcs" or "popcorn" chondroid calcifications. | Eccentric location (sacral ala). Painful mass. Can arise secondary to osteochondroma. | Atypical chondrocytes in hyaline cartilage matrix. S-100 positive, Brachyury negative. |
| Metastatic Disease | Multiple lesions, destructive, can be lytic (lung, thyroid, kidney) or blastic (prostate, breast). | History of primary carcinoma. Older demographic. Rapid onset of mechanical pain. | Histology matches primary tumor. Specific markers (e.g., PSA, TTF-1, ER/PR) dictate origin. |
| Multiple Myeloma / Plasmacytoma | "Punched out" lytic lesions, severe osteopenia. No reactive sclerosis. | Systemic symptoms, anemia, hypercalcemia, renal insufficiency. Bence-Jones proteinuria. | Monoclonal plasma cell proliferation. CD138 positive, Kappa/Lambda light chain restriction. |
| Schwannoma | Well-circumscribed, smooth bony remodeling, enlargement of neural foramina. Target sign on MRI. | Radicular pain, sensory deficits. Slow growing. Arises from peripheral nerve sheath. | Antoni A (cellular) and Antoni B (hypocellular) areas. Verocay bodies. S-100 strongly positive. |
Surgical Decision Making and Classification
Oncological Staging
Complete staging was performed to rule out disseminated disease. A CT scan of the chest, abdomen, and pelvis, along with a whole-body technetium-99m bone scintigraphy, revealed no evidence of pulmonary or skeletal metastases. Chordomas are locally aggressive tumors with a high propensity for local recurrence, but they metastasize late in the disease course, most commonly to the lungs, bone, and liver.
Based on the Enneking staging system for malignant musculoskeletal tumors, this lesion was classified as Stage IB (low-grade, extracompartmental). The tumor had breached the sacral cortex to form a presacral mass, extending beyond the bony compartment of the sacrum.
Operative vs Non Operative Management
The management of sacral chordoma mandates a multidisciplinary approach. Chordomas are notoriously radioresistant and chemoresistant. While advanced modalities such as proton beam therapy and carbon ion radiotherapy have shown efficacy in local control, particularly for unresectable disease or as adjuvant therapy, surgical resection remains the cornerstone of curative intent.
The primary oncological goal is en bloc resection with wide, negative margins (R0 resection). Intralesional curettage or piecemeal resection is universally associated with near 100% local recurrence rates and significantly diminished overall survival.
In this case, the tumor extended to the S2 level. Achieving a wide margin necessitated a high sacral amputation (trans-sacral osteotomy at the S1-S2 disc space or through the S1 body). This level of resection inherently requires the sacrifice of the S2-S5 nerve roots bilaterally. The patient was extensively counseled regarding the absolute certainty of permanent postoperative bowel, bladder, and sexual dysfunction. The necessity of a permanent colostomy and urostomy was discussed, although in some cases, a bowel management program can be attempted if the external anal sphincter retains any residual innervation, which was unlikely here given the bilateral S2 sacrifice. The patient prioritized oncological clearance and survival over neurological preservation and consented to the en bloc resection.
Surgical Technique and Intervention
Preoperative Preparation and Embolization
Given the highly vascular nature of the pelvic anatomy and the potential for life-threatening hemorrhage during sacral resection, preoperative angiographic embolization was performed 24 hours prior to surgery. The bilateral middle sacral arteries and branches of the internal iliac arteries supplying the tumor were selectively embolized using polyvinyl alcohol particles and coils. This step significantly reduces intraoperative blood loss and improves visualization during the presacral dissection.
The surgical strategy utilized a sequential anterior-posterior approach, performed during a single anesthetic event by a combined team of orthopedic oncology, colorectal surgery, and vascular surgery.
Anterior Approach and Mobilization
The patient was positioned supine, and a lower midline laparotomy was performed. The colorectal team mobilized the sigmoid colon and rectum. The mesorectum was carefully dissected off the presacral fascia (Waldeyer's fascia). Maintaining the integrity of the presacral fascia over the tumor is the most critical oncological step in the anterior stage, as it serves as the anterior surgical margin.
The internal iliac vessels were identified, and the lateral sacral arteries and veins were ligated. The sympathetic trunks were identified and transected. The dissection was carried laterally to the sacroiliac joints. A laparotomy pad was placed deep in the presacral space to protect the rectum and pelvic viscera during the subsequent posterior osteotomies. Once the anterior mobilization was complete and the tumor was completely freed from the visceral structures, the abdomen was closed temporarily, and the patient was carefully flipped into the prone position on a Jackson table.
Posterior Approach and En Bloc Resection
A wide posterior midline incision was made, incorporating the previous biopsy tract in an elliptical fashion to ensure it remained en bloc with the specimen. The gluteus maximus muscles were sharply detached from their sacral origins and reflected laterally. The sacrotuberous and sacrospinous ligaments were identified and transected, releasing the pelvic floor musculature from the lower sacrum.
The sciatic nerves were identified at the greater sciatic notch and carefully protected. The piriformis muscles were transected at their musculotendinous junctions. A laminectomy of L5 and S1 was performed to expose the thecal sac. The dura was ligated and transected distal to the S1 nerve roots, effectively sacrificing the S2-S5 nerve roots along with the filum terminale. The S1 nerve roots were meticulously dissected free and mobilized laterally.
Using a combination of osteotomes and a high-speed burr, the posterior osteotomies were initiated. Based on the preoperative 3D templating, trans-iliac osteotomies were performed just lateral to the sacroiliac joints to ensure a wide margin around the tumor laterally. A transverse osteotomy was then performed through the mid-body of S1, connecting the bilateral trans-iliac cuts. The osteotomy was completed through the anterior cortex, meeting the plane created during the anterior stage. The entire specimen, including the lower sacrum, tumor, and adherent presacral fascia, was delivered en bloc from the surgical field.
Spinopelvic Reconstruction and Soft Tissue Coverage
High sacral amputations that resect the sacroiliac joints completely disrupt the spinopelvic ring, leading to spinopelvic dissociation. Because the osteotomy in this case was through the S1 body, a portion of the sacroiliac joint remained intact, maintaining some innate pelvic stability. However, to prevent late kyphotic deformity, sacral settling, and to facilitate early mobilization, a robust lumbopelvic fixation construct was applied.
Pedicle screws were placed bilaterally in L4 and L5. Dual iliac screws were placed bilaterally, extending deep into the supra-acetabular corridor to maximize pull-out strength. The construct was connected using dual titanium rods, contoured to restore lumbar lordosis. Cross-links were applied to increase torsional rigidity. Structural allograft was placed across the osteotomy site to promote long-term arthrodesis.
Following skeletal reconstruction, attention was turned to soft tissue coverage. The large dead space created by the sacrectomy is highly prone to hematoma, infection, and wound breakdown. A plastic surgery team mobilized bilateral gluteus maximus advancement flaps. The flaps were approximated in the midline over closed suction drains, providing robust, well-vascularized muscle coverage over the spinal hardware and preventing dead space accumulation.
Post Operative Protocol and Rehabilitation
Immediate Postoperative Care
The patient was transferred to the Surgical Intensive Care Unit intubated and sedated for hemodynamic monitoring. Massive fluid shifts and coagulopathy are common following sacral resections. Resuscitation was guided by arterial line monitoring and targeted central venous pressures. Prophylactic broad-spectrum intravenous antibiotics were continued for 48 hours.
Strict flat bed rest was maintained for the first 72 hours to allow for initial soft tissue resting and to minimize tension on the gluteal flaps. Closed suction drains were closely monitored and removed sequentially once output decreased to less than 30 cc per 24 hours. Deep vein thrombosis prophylaxis with low molecular weight heparin was initiated on postoperative day 2, balancing the risk of epidural hematoma with the high risk of venous thromboembolism in orthopedic oncology patients.
Long Term Rehabilitation and Surveillance
Because the S2-S5 nerve roots were sacrificed, the patient required intensive postoperative management of his bowel and bladder. A suprapubic catheter was placed intraoperatively, and he was later transitioned to a clean intermittent self-catheterization protocol. A bowel management regimen utilizing daily enemas and suppositories was initiated, though he eventually required a diverting colostomy due to intractable fecal incontinence.
Physical therapy was initiated on postoperative day 4. He was mobilized with a walker, initially restricted to touch-down weight-bearing bilaterally to protect the lumbopelvic hardware until radiographic evidence of initial graft incorporation was observed at 6 weeks. He progressed to full weight-bearing by 12 weeks.
Oncological surveillance for chordoma requires lifelong commitment due to the high risk of late local recurrence. The protocol includes MRI of the pelvis and CT of the chest every 3 months for the first 2 years, every 6 months for years 3 to 5, and annually thereafter. At his 2-year follow-up, the patient remained disease-free with stable lumbopelvic hardware, mobilizing independently with a cane, and adapting to his altered bowel and bladder physiology.
Clinical Pearls and Pitfalls
- Pitfall - Inappropriate Biopsy: The most devastating error in the initial management of a sacral mass is a transrectal or transvaginal biopsy. This contaminates the presacral space, destroys the only natural barrier (presacral fascia), and frequently renders the tumor unresectable. Always utilize a posterior, midline CT-guided approach.
- Pearl - The Presacral Fascia: The anterior margin is the most difficult to achieve in sacral chordoma resection. The mesorectum must be dissected off Waldeyer's fascia, leaving the fascia intact on the tumor specimen. If the tumor has invaded through the fascia into the mesorectum, a concurrent abdominoperineal resection (APR) by colorectal surgery is mandatory to achieve a negative margin.
- Pitfall - Underestimating Blood Loss: The presacral venous plexus and tumor neovascularization can lead to catastrophic, uncontrollable hemorrhage. Preoperative embolization is highly recommended. Intraoperatively, early ligation of the internal iliac branches and meticulous hemostasis are critical.
- Pearl - Neurological Counseling: The level of sacral root sacrifice dictates postoperative function. Preservation of at least one S2 root is required for a 50% chance of functional bowel/bladder control. Preservation of S3 is required for normal sphincter function. Surgeons must have explicit, documented discussions with the patient regarding the trade-off between oncological margins and permanent neurological deficits. Intralesional surgery to spare nerve roots in chordoma is oncologically unsound.
- Pitfall - Inadequate Fixation: Failure to recognize the biomechanical demands of a high sacrectomy can lead to hardware failure, spinopelvic dissociation, and severe pain. Constructs must bypass the sacrum entirely, utilizing robust pedicle fixation in the lumbar spine and long, multi-point fixation in the ilium (e.g., Galveston technique or modern S2-alar-iliac/iliac screws).
