Orthopedic Case Study: Diagnosing Pigmented Villonodular Synovitis (PVNS) of the Knee
Key Takeaway
PVNS in the knee is diagnosed through a comprehensive approach. Key indicators include chronic knee pain and effusion, specific MRI findings like diffuse synovitis and hemosiderin deposition ('blooming artifact'), serosanguinous synovial fluid, and confirmed by arthroscopic synovial biopsy demonstrating characteristic villous proliferation with hemosiderin-laden macrophages.
Patient Presentation and History
A 38-year-old male presented to the orthopedic clinic with a 2-year history of insidious onset left knee pain, intermittent swelling, and mechanical symptoms including occasional catching and giving way. Initially, the patient’s symptoms were mild, episodic, and attributed to sports-related overuse following recreational basketball. However, over the past 6 months, the clinical picture evolved significantly. The pain transitioned from intermittent to constant, the joint effusion became persistent rather than activity-dependent, and functional limitations markedly increased. The patient reported specific difficulty with deep knee flexion, squatting, and ascending stairs, noting a sensation of "fullness" and mechanical block within the joint.
He reported no specific inciting trauma, twisting injury, or direct blow to the knee that correlated with the onset of symptoms. A comprehensive review of systems was negative for fever, chills, night sweats, fatigue, or unintentional weight loss, effectively lowering the clinical suspicion for an acute infectious process or highly aggressive systemic malignancy.
His past medical history was unremarkable. He denied any personal or family history of inflammatory arthritides, such as rheumatoid arthritis, seronegative spondyloarthropathies, or gout. Furthermore, there was no history of bleeding diatheses, coagulopathies, or spontaneous hemarthroses that would suggest hemophilic arthropathy.
Prior to this consultation, the patient had undergone two separate joint aspirations by a primary care physician. Both aspirations yielded large volumes of dark, serosanguinous fluid. Fluid analysis on both occasions was negative for monosodium urate and calcium pyrophosphate dihydrate crystals. Gram stain and aerobic/anaerobic cultures were negative, ruling out septic arthritis. A trial of non-steroidal anti-inflammatory drugs (NSAIDs), specifically Meloxicam 15 mg daily, provided minimal and purely temporary symptomatic relief. Physical therapy focusing on quadriceps strengthening had been attempted but was poorly tolerated due to pain and the mechanical block secondary to the persistent effusion.
Clinical Examination Findings
Left Knee Assessment
Inspection of the left lower extremity revealed a noticeable, palpable joint effusion. Unlike the tense, fluid-filled effusion typical of an acute ligamentous tear or traumatic hemarthrosis, this effusion was distinctly "boggy" and doughy in consistency, highly suggestive of synovial hypertrophy rather than simple synovial fluid accumulation. There was no obvious overlying erythema, ecchymosis, or localized warmth to suggest acute inflammation or infection. Mild to moderate quadriceps atrophy was visually apparent and confirmed with circumferential thigh measurements (2 cm deficit compared to the contralateral limb measured 10 cm proximal to the superior pole of the patella), indicative of chronic joint pathology and secondary muscle inhibition.
Palpation elicited diffuse tenderness along both the medial and lateral joint lines, extending proximally over the suprapatellar pouch. Significant synovial thickening was palpable, particularly in the suprapatellar region and the medial gutter. The thickened synovium felt nodular and irregular. No discrete, freely mobile intra-articular bodies or large extra-articular masses were immediately obvious on superficial palpation.
Range of motion (ROM) testing demonstrated significant mechanical restriction. Active ROM was limited, demonstrating a 5° flexion contracture and maximal flexion to 100° (normal expected ROM: 0-140°). Pain was exacerbated at the extremes of both flexion and extension. Passive ROM closely mirrored active motion, terminating with a firm, mechanical end-feel rather than guarding, suggesting a physical block from the hypertrophic synovial tissue. Palpable and audible crepitus was noted throughout the arc of motion, localized primarily to the anterior compartment.
Ligamentous stability testing was unremarkable. The medial and lateral collateral ligaments demonstrated firm endpoints to valgus and varus stress at both 0° and 30° of knee flexion. Anterior and posterior drawer tests, as well as the Lachman test, were negative with firm endpoints, indicating intact cruciate ligaments. The pivot shift test was negative.
Meniscal provocative testing yielded equivocal results. McMurray's test elicited diffuse, non-specific pain during both internal and external rotation with axial load, but no distinct, palpable mechanical click was appreciated. Apley's grind test was positive for pain, suggestive of meniscal irritation or, more likely in this context, compression of the inflamed synovium between the articular surfaces.
Patellofemoral joint assessment revealed mild patellofemoral crepitus during active extension. There was no patellar instability, apprehension, or abnormal patellar tracking. The patellar grind test (Clarke's sign) was positive for pain.
Neurological and Vascular Assessment
A comprehensive neurovascular examination of the left lower extremity was performed. Distal pulses, including the dorsalis pedis and posterior tibial arteries, were palpable, bounding, and symmetric to the contralateral limb. Capillary refill was brisk (less than 2 seconds) in all toes. Sensation was intact to light touch and pinprick throughout the L2 to S1 dermatomes of the lower extremity. Motor strength was graded 5/5 in all major muscle groups, including hip flexors, knee extensors, knee flexors, ankle dorsiflexors, ankle plantarflexors, and extensor hallucis longus, despite the aforementioned quadriceps atrophy.
Systemic and Contralateral Evaluation
Examination of the contralateral right knee and other major appendicular joints revealed no other joint effusions, synovial thickening, or signs of systemic polyarticular disease. Cardiovascular, pulmonary, and abdominal examinations were unremarkable, further isolating the pathology to the left knee joint.
Imaging and Diagnostics
Plain Radiography Analysis
Standard orthogonal weight-bearing radiographs of the left knee were obtained, including Anteroposterior (AP), Lateral, 45-degree posteroanterior flexion (Rosenberg), and Merchant patellofemoral views.
The initial radiographic evaluation demonstrated a moderate to large joint effusion, visible as a dense soft tissue shadow in the suprapatellar pouch on the lateral view. Remarkably, despite the 2-year history of symptoms, the tibiofemoral and patellofemoral joint spaces appeared relatively preserved, with no evidence of gross joint space narrowing that would characterize advanced osteoarthritis. There were no obvious osteophytes, subchondral sclerosis, or significant degenerative changes.
Mild, diffuse osteopenia of the distal femur and proximal tibia was observed, likely secondary to chronic hyperemia associated with synovial inflammation and localized disuse osteopenia. No acute fracture lines, periosteal reactions, or aggressive osseous lesions were evident. However, close inspection revealed subtle, well-marginated radiolucent lesions—subchondral erosions—at the margins of the medial femoral condyle and the posterior aspect of the tibial plateau. These erosions, while not overtly destructive or expansile, occurred at the capsular reflections ("bare areas" where cartilage ends and synovium begins) and raised immediate clinical suspicion for an aggressive synovial process.
Magnetic Resonance Imaging
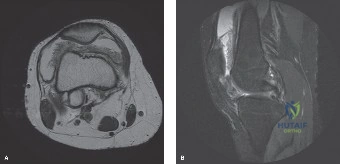
Given the chronicity of the symptoms, the boggy effusion, the history of serosanguinous aspirates, and the subtle marginal erosions on plain radiographs, a Magnetic Resonance Imaging (MRI) scan of the left knee was strictly indicated. Standard protocols including T1-weighted, T2-weighted, Proton Density (PD), and Gradient-Recalled Echo (GRE) sequences were obtained, both with and without intravenous Gadolinium contrast. The MRI proved to be the definitive diagnostic modality.

The MRI findings demonstrated diffuse, exuberant, hypertrophic synovitis involving all compartments of the knee joint. The synovial proliferation extended extensively into the suprapatellar pouch, the medial and lateral gutters, the intercondylar notch, and the posterior joint recesses. The synovium exhibited a striking frond-like, villonodular architecture.
The most critical and pathognomonic finding was the presence of extensive areas of low signal intensity on both T1- and T2-weighted sequences within the hypertrophic synovial masses. On the Gradient-Recalled Echo (GRE) sequences, these areas demonstrated a prominent "blooming artifact." This magnetic susceptibility artifact is caused by the paramagnetic properties of extensive hemosiderin deposition within the synovial tissue, a hallmark of Pigmented Villonodular Synovitis (PVNS).
Following the administration of intravenous Gadolinium, the thickened synovial tissue demonstrated avid, heterogeneous enhancement, confirming the highly vascular nature of the proliferative process.
Furthermore, the MRI allowed for precise evaluation of the articular cartilage and subchondral bone. The articular cartilage appeared focally thinned in several areas, particularly over the medial femoral condyle. The MRI clearly delineated the subchondral bone erosions previously suspected on plain radiographs, confirming their presence on the medial femoral condyle and the posterior aspect of the tibial plateau. These erosions were filled with the same low-signal, hemosiderin-laden synovial tissue. Notably, there was no significant surrounding bone marrow edema adjacent to these erosions, a finding that helps differentiate chronic, slow-growing erosions of PVNS from acute inflammatory or infectious erosions.
Synovial Fluid Analysis and Laboratory Studies
While previous aspirations were performed elsewhere, a repeat diagnostic arthrocentesis was performed in the clinic under strict aseptic technique. The aspiration yielded 45 cc of dark, opaque, reddish-brown fluid, often classically described as resembling "crankcase oil" or liquid chocolate.
Fluid analysis demonstrated a high red blood cell count and a moderately elevated white blood cell count with a mononuclear predominance. The fluid was negative for crystals under polarized light microscopy. Gram stain was negative. Subsequent aerobic, anaerobic, mycobacterial, and fungal cultures all remained negative at final read.
Peripheral blood laboratory studies were obtained to complete the workup. Complete Blood Count (CBC), basic metabolic panel, Erythrocyte Sedimentation Rate (ESR), and C-Reactive Protein (CRP) were all within normal laboratory limits. A coagulation panel (PT, aPTT, INR) was normal, definitively excluding an underlying coagulopathy as the etiology for the recurrent hemarthroses.
Histopathological Evaluation
While the MRI findings were considered pathognomonic, the definitive diagnosis of PVNS—now more accurately classified by the World Health Organization (WHO) as Tenosynovial Giant Cell Tumor (TGCT), diffuse type—relies on histopathological examination of the excised tissue.
In cases where diagnostic ambiguity remains despite MRI, an image-guided core needle biopsy or a diagnostic arthroscopic biopsy may be indicated prior to definitive surgical planning. In this case, the clinical and radiographic picture was sufficiently classic to proceed directly to definitive operative intervention, with histopathology to be confirmed from the surgical specimen.
Microscopically, PVNS is characterized by a hypercellular proliferation of mononuclear cells (the actual neoplastic cells) admixed with a reactive infiltrate of multinucleated osteoclast-like giant cells, foamy lipid-laden macrophages (histiocytes), and extensive intracellular and extracellular hemosiderin deposits. The mononuclear cells frequently demonstrate a specific genetic translocation, t(1;2)(p13;q37), which results in the fusion of the Colony Stimulating Factor 1 (CSF1) gene with the Collagen Type VI Alpha 3 (COL6A3) promoter. This translocation leads to the localized overproduction of CSF1, which acts as a chemoattractant, recruiting the massive influx of non-neoplastic macrophages and giant cells that form the bulk of the tumor mass.
Differential Diagnosis
The presentation of chronic monoarticular knee pain, recurrent serosanguinous effusions, and synovial hypertrophy requires a systematic approach to rule out several distinct pathologies. The following table outlines the primary differential diagnoses considered in this case.
| Condition | Clinical Presentation | Radiographic Findings | MRI Characteristics | Synovial Fluid Analysis |
|---|---|---|---|---|
| Pigmented Villonodular Synovitis (Diffuse TGCT) | Insidious onset, chronic pain, boggy effusion, mechanical symptoms, recurrent hemarthrosis. | Preserved joint space, dense soft tissue shadow, marginal subchondral erosions. | Diffuse synovial thickening, low T1/T2 signal, prominent GRE blooming artifact (hemosiderin). | Serosanguinous to dark brown ("crankcase oil"), negative for crystals/infection. |
| Hemophilic Arthropathy | History of known bleeding diathesis, recurrent spontaneous acute hemarthroses since childhood. | Epiphyseal overgrowth (in children), squared patella, widened intercondylar notch, advanced secondary OA. | Synovial hypertrophy, hemosiderin deposition (blooming on GRE), severe cartilage loss. | Grossly bloody, high RBC count. Patient will have abnormal coagulation profile. |
| Synovial Chondromatosis | Chronic pain, mechanical locking/catching, palpable mobile bodies, insidious onset. | Multiple intra-articular calcified or ossified loose bodies ("snowstorm" appearance). | Multiple nodular loose bodies, variable signal depending on calcification, no massive hemosiderin blooming. | Clear to slightly yellow, highly viscous, negative for crystals/infection. |
| Rheumatoid Arthritis (Monoarticular presentation) | Morning stiffness, boggy effusion, typically polyarticular but can present monoarticularly early on. | Periarticular osteopenia, symmetric joint space narrowing, marginal erosions. | Synovial hypertrophy, intense post-contrast enhancement, bone marrow edema, no massive hemosiderin. | Inflammatory fluid (WBC > 2,000), cloudy, yellow. Positive Rheumatoid Factor / anti-CCP. |
| Tuberculous Arthritis | Extremely insidious onset, chronic swelling, mild pain, possible constitutional symptoms. | Phemister's triad: severe periarticular osteoporosis, peripheral erosions, gradual joint space narrowing. | Synovial thickening, large effusions, bone marrow edema, cold abscesses. | Inflammatory, mononuclear predominance. Requires AFB stain and mycobacterial culture. |
Surgical Decision Making and Classification
World Health Organization Classification of TGCT
To formulate an appropriate treatment plan, it is imperative to classify the lesion accurately. The World Health Organization (WHO) classifies these lesions under the umbrella term Tenosynovial Giant Cell Tumor (TGCT), which is subdivided based on growth pattern and biological behavior:
1. Localized TGCT (formerly Giant Cell Tumor of Tendon Sheath or localized PVNS): Typically presents as a discrete, well-circumscribed, pedunculated nodule. It is most common in the digits of the hand but can occur in large joints. Surgical excision is generally curative with a low recurrence rate.
2. Diffuse TGCT (formerly Diffuse PVNS): Involves the entire synovial lining of a joint, bursa, or tendon sheath. It is locally aggressive, infiltrative, and carries a notoriously high recurrence rate (ranging from 20% to 50% in the literature) even after extensive surgical synovectomy.
Based on the MRI findings demonstrating involvement of the suprapatellar pouch, gutters, intercondylar notch, and posterior recesses, this patient presented with Diffuse TGCT of the knee.
Operative Versus Non Operative Management
The natural history of untreated diffuse TGCT is progressive joint destruction. The hyperplastic synovium acts as a space-occupying lesion, causing mechanical damage, while the biochemical environment (metalloproteinases and inflammatory cytokines) degrades articular cartilage. The subchondral erosions eventually lead to structural collapse and severe secondary osteoarthritis, often necessitating premature total joint arthroplasty in young patients.
Therefore, non-operative management is not a viable definitive strategy for diffuse TGCT. The primary goal of treatment is the complete eradication of the abnormal synovial tissue to relieve symptoms, halt joint destruction, and prevent recurrence.
Surgical synovectomy is the gold standard. The critical decision lies in the surgical approach: arthroscopic, open, or a combined approach.
* All-Arthroscopic Synovectomy: Offers lower morbidity, faster rehabilitation, and excellent visualization of the anterior compartments and intercondylar notch. However, visualizing and accessing the extreme posterior compartments (posteromedial and posterolateral recesses) can be technically demanding and historically associated with higher recurrence rates if tissue is left behind.
* Open Synovectomy (Anterior and Posterior approaches): Historically the standard for diffuse disease. Allows for direct visualization and en bloc resection of massive synovial tissue. However, it is associated with significant morbidity, prolonged rehabilitation, high risk of severe arthrofibrosis, and wound healing complications.
* Combined Approach: Utilizing arthroscopy for the anterior compartments and an open posterior approach for the posterior recesses.
Given modern arthroscopic techniques, advanced instrumentation (such as angled shavers and radiofrequency wands), and the surgeon's comfort level, an all-arthroscopic extensive anterior and posterior synovectomy was chosen for this patient. The MRI confirmed that while the posterior disease was extensive, there was no extra-articular extension (such as into the popliteal fossa neurovascular bundle) that would mandate an open posterior approach.
Role of Adjuvant and Targeted Therapies
Because the recurrence rate of diffuse TGCT remains unacceptably high despite meticulous surgery, adjuvant therapies must be considered.
* Radiosynoviorthesis (Intra-articular Yttrium-90): Injection of a beta-emitting radioisotope into the joint post-operatively to ablate microscopic residual synovial tissue. This is highly effective but requires specialized nuclear medicine facilities and carries a risk of radionecrosis if the isotope leaks extra-articularly.
* External Beam Radiation Therapy (EBRT): Reserved for highly recurrent, aggressive cases. Carries risks of post-radiation sarcoma and joint stiffness.
* Targeted Systemic Therapy (Pexidartinib): A relatively recent breakthrough. Pexidartinib is a selective Tyrosine Kinase Inhibitor (TKI) that targets the CSF1 receptor. By blocking the CSF1 receptor on the recruited macrophages, it effectively starves the tumor of its cellular bulk. It is FDA-approved for adults with symptomatic TGCT associated with severe morbidity or functional limitations, and not amenable to improvement with surgery. Due to the risk of severe hepatotoxicity (requiring a REMS program), it is generally reserved for unresectable cases or massive recurrences.
For this patient, the plan was meticulous surgical resection. Adjuvant radiosynoviorthesis would be considered if histopathology confirmed highly active diffuse disease or if early recurrence was detected on surveillance MRI.
Surgical Technique and Intervention
Patient Positioning and Anesthesia
The patient was taken to the operating room and placed in the supine position on the operating table. General endotracheal anesthesia was induced to ensure complete muscle relaxation. A high-thigh tourniquet was applied to the left lower extremity. The leg was placed in a standard arthroscopic leg holder, allowing for full range of motion and valgus/varus stress. The contralateral leg was placed in a well-padded gynecologic stirrup. The surgical site was prepped and draped in a standard sterile orthopedic fashion. Esmarch bandage was used for exsanguination, and the tourniquet was inflated to 250 mmHg.
Anterior Compartment Arthroscopic Synovectomy
Standard anterolateral and anteromedial portals were established adjacent to the patellar tendon. Upon introduction of the arthroscope, a massive amount of dark, rusty-brown, villonodular synovial tissue was immediately encountered, obscuring normal intra-articular landmarks. The synovial fluid was evacuated, and extensive lavage was performed.
A systematic, compartmentalized approach to the synovectomy was initiated to ensure no area was overlooked.
1. Suprapatellar Pouch: A superolateral portal was established to allow for inflow and alternative viewing angles. Using a combination of a 4.5mm aggressive motorized shaver and a radiofrequency ablation wand, the hypertrophic synovium was systematically resected from the anterior femur, the undersurface of the quadriceps tendon, and the medial and lateral plicae. The tissue was notably friable and highly vascular; meticulous hemostasis with the radiofrequency wand was required continuously.
2. Medial and Lateral Gutters: The arthroscope was swept into the medial gutter. The villous tissue was adherent to the capsule and extended down to the meniscocapsular junction. A 70-degree arthroscope was utilized to visualize the extreme posterior reflections of the gutters. Resection was carried down to the bare capsular tissue. The process was repeated in the lateral gutter, paying careful attention to the popliteus hiatus, a common hiding place for residual TGCT.
3. Intercondylar Notch: The notch was completely choked with brown synovial tissue, obscuring the Anterior Cruciate Ligament (ACL) and Posterior Cruciate Ligament (PCL). Careful shaving was performed to skeletonize the cruciate ligaments without damaging their fibers. The tissue was cleared from the anterior aspect of the PCL and the medial aspect of the lateral femoral condyle.
4. Articular Cartilage and Erosions: Once the soft tissue was cleared, the articular surfaces were inspected. Outerbridge Grade II-III chondromalacia was noted on the medial femoral condyle. The MRI-identified subchondral erosions on the medial femoral condyle and tibial plateau were visualized. These defects were filled with tumor tissue. Small curettes and the arthroscopic shaver were used to meticulously debride the erosions down to healthy, bleeding subchondral bone.
Posterior Compartment Management
Thorough clearance of the posterior compartments is the most critical step in preventing recurrence in diffuse TGCT. Trans-notch visualization is insufficient for a complete posterior synovectomy.
- Posteromedial Compartment: The arthroscope was passed through the intercondylar notch, between the PCL and the medial femoral condyle, into the posteromedial compartment. An 18-gauge spinal needle was used to localize a posteromedial portal under direct intra-articular visualization, ensuring it was superior to the joint line and posterior to the medial collateral ligament to avoid the saphenous nerve and vein. An incision was made, and a cannula was introduced. The posteromedial compartment was packed with diseased synovium. Using the shaver through the posteromedial portal and viewing trans-notch (and subsequently switching viewing and working portals), the entire posterior capsule, the posterior horn of the medial meniscus, and the posterior aspect of the medial femoral condyle were cleared.
- Posterolateral Compartment: Similarly, the arthroscope was passed between the ACL and the lateral femoral condyle into the posterolateral compartment. A posterolateral portal was established under direct visualization, taking extreme care to stay anterior to the biceps femoris tendon and fibular head to protect the common peroneal nerve. The synovial tissue surrounding the posterior horn of the lateral meniscus, the posterior cruciate ligament insertion, and the posterior lateral capsule was meticulously resected.
Specimen Collection and Closure
Multiple representative samples of the resected synovial tissue were sent to pathology for definitive histological analysis.
Following the exhaustive synovectomy, the joint was copiously irrigated with 9 liters of normal saline to remove any microscopic debris and free-floating cells that could seed a recurrence. The tourniquet was deflated prior to closure to assess hemostasis. As expected with TGCT, there was diffuse capsular bleeding. Extensive electrocautery was utilized to achieve hemostasis.
Given the massive raw synovial surface area created by the resection, a medium-sized intra-articular Hemovac drain was placed through the superolateral portal to prevent the formation
