Labral Support (Shelf) Procedure for Perthes Disease: An Intraoperative Masterclass
Key Takeaway
Join us in the OR for an immersive masterclass on the labral support (shelf) procedure for Perthes disease. We'll meticulously cover patient selection, detailed surgical anatomy, the precise step-by-step intraoperative technique, including arthrography and shelf construction, critical pearls for avoiding pitfalls, and comprehensive postoperative management. This guide ensures a granular understanding of every surgical nuance for optimal patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues, to the operating theater. Today, we are undertaking a critical intervention for a profoundly challenging pediatric hip condition: the Labral Support (Shelf) Procedure for Legg-Calvé-Perthes Disease. This operation is meticulously designed to address the catastrophic biomechanical instability and progressive deformity that arise from avascular necrosis (AVN) of the capital femoral epiphysis. The overarching goal of this procedure is to contain the necrotic, malleable femoral head within the acetabulum, definitively eliminate hinge subluxation, improve lateral coverage, support the acetabular labrum, and prevent secondary femoroacetabular impingement (FAI). Ultimately, this biological and mechanical optimization stimulates the concentric remodeling of both the femoral head and the acetabulum during the vulnerable reossification phase.
Legg-Calvé-Perthes disease is a complex, idiopathic condition of the immature hip, defined by a transient disruption of the blood supply to the capital femoral epiphysis and its adjacent physis. This ischemic event precipitates a cascade of necrosis, leading to the gradual osteoclastic resorption of dead subchondral bone, subsequent revascularization, and eventual reossification. However, it is during the fragmentation and early reossification phases that the epiphysis is mechanically compromised. Without intervention, normal weight-bearing forces cause the necrotic head to extrude laterally, leading to plastic deformation, flattening (coxa plana), and broadening (coxa magna). The natural history of untreated, severe Perthes disease is invariably linked to early-onset, debilitating osteoarthritis.

Epidemiologically, Perthes disease presents a distinct profile that guides our clinical suspicion and screening protocols. Symptoms typically manifest between the ages of 4 and 8 years, although the spectrum can extend from toddlers as young as 2 years up to skeletal maturity. There is a pronounced male predominance, with a male-to-female ratio of approximately 4:1. Furthermore, bilateral involvement is observed in up to 15% to 17% of cases, though typically occurring asynchronously. The etiology remains multifactorial and heavily debated, with theories implicating subtle coagulopathies (such as Factor V Leiden mutations or protein C/S deficiencies), passive smoking exposure, socioeconomic factors, and repetitive microtrauma to a susceptible vascular supply.
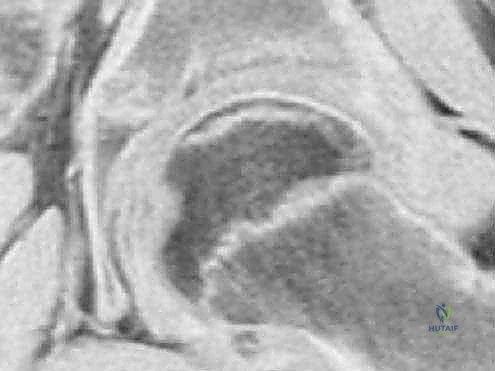
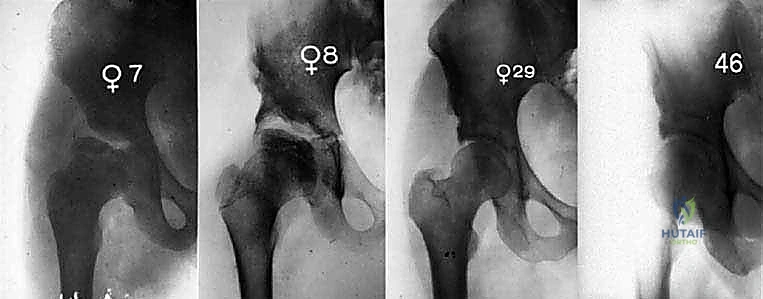
Our understanding of the disease's natural history is anchored by the Waldenström radiographic stages, which are paramount for surgical timing. The Necrosis stage features initial infarction and growth arrest, radiographically presenting as a dense, small epiphysis with a widened medial joint space (Waldenström's sign) and subchondral fracture (Crescent sign). The Fragmentation stage marks the onset of necrotic bone resorption and early revascularization; this is the period of maximum mechanical vulnerability where lateral extrusion and hinge subluxation occur. The Reossification stage involves the deposition of new bone on the fibrovascular stroma, dictating the final shape of the head. Finally, the Remodeling stage continues until skeletal maturity, during which the newly formed bone adapts to the mechanical stresses of the joint. Our surgical intervention today is optimally timed during the late fragmentation or early reossification phase to physically guide this remodeling process.
Detailed Surgical Anatomy and Biomechanics
A profound mastery of the regional anatomy and the unique biomechanics of the dysplastic Perthes hip is non-negotiable for the successful execution of the labral support procedure. Biomechanically, the fundamental principle of "containment" dictates that the femoral head must be deeply seated within the acetabulum to ensure that the forces of weight-bearing are distributed evenly across a spherical template. In Perthes disease, lateral extrusion of the femoral head leads to point-loading on the lateral acetabular rim. This creates a fulcrum effect—known as hinge abduction—where the extruded, enlarged femoral head impinges against the acetabular margin during abduction, levering the medial head out of the joint. The shelf procedure directly neutralizes this by creating an extracapsular bony buttress that extends the acetabular roof laterally, supporting the labrum and preventing this disastrous lateral translation.
Navigating the neurovascular landscape of the anterior hip requires meticulous precision, particularly concerning the Lateral Femoral Cutaneous Nerve (LFCN). Arising from the L2-L3 nerve roots, the LFCN courses obliquely across the iliacus, passes beneath or through the inguinal ligament just medial to the anterior superior iliac spine (ASIS), and bifurcates. The posterior branch is at paramount risk during our bikini incision and superficial dissection. It typically traverses beneath the sartorius muscle and pierces the fascia lata approximately 1 to 2 cm inferior to the ASIS. Iatrogenic injury to this nerve results in meralgia paresthetica—a highly distressing dysesthesia over the anterolateral thigh that can significantly mar an otherwise mechanically successful operation.
The muscular intervals utilized in this approach are designed to maximize exposure while preserving the dynamic stabilizers of the hip. We exploit the internervous plane between the tensor fasciae latae (TFL), innervated by the superior gluteal nerve, and the sartorius/rectus femoris complex, innervated by the femoral nerve. As we deepen the dissection toward the anterior inferior iliac spine (AIIS), we encounter the reflected head of the rectus femoris. A cardinal rule of this specific shelf technique is the absolute preservation of the superior origins of the gluteus medius and minimus muscles along the outer iliac wing. Unlike a traditional Salter innominate osteotomy that requires extensive subperiosteal stripping of the abductors, the shelf procedure minimizes abductor trauma, thereby reducing postoperative Trendelenburg gait and accelerating rehabilitation.
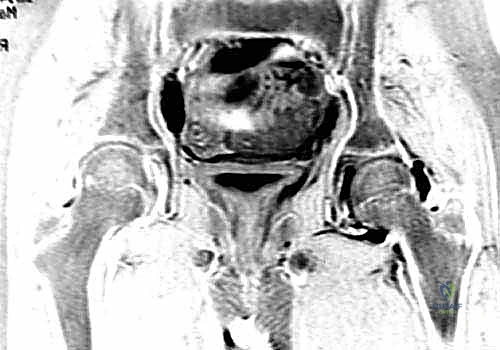
The acetabular labrum is the biological keystone of this procedure. Situated at the peripheral margin of the acetabulum, this fibrocartilaginous ring deepens the socket by 21%, increases the articular surface area by nearly 30%, and creates a critical fluid seal that maintains intra-articular hydrostatic pressure for cartilage nutrition. In the Perthes hip undergoing lateral extrusion, the labrum is subjected to immense shear forces, often becoming hypertrophic, inverted, or torn. The shelf procedure is inherently extracapsular; we create a bony slot just superior to the joint capsule and the labrum. The grafted bone is impacted into this slot to act as a rigid awning, physically supporting the capsulolabral complex from above. It is imperative that the surgeon recognizes the location of the labral growth plate, as iatrogenic penetration into the joint or damage to the physis will arrest acetabular development and precipitate early joint failure.
Exhaustive Indications and Contraindications
The decision to proceed with a labral support (shelf) procedure requires a nuanced synthesis of the patient's chronological age, skeletal maturity, radiographic stage, and dynamic joint mechanics. The ideal candidate is a child between the ages of 6 and 11 years who is in the fragmentation or early reossification stage of the disease. Children in this age bracket possess sufficient remaining growth potential for the acetabulum to remodel around the contained femoral head, yet they are old enough that the disease process is likely to cause severe deformity if left unchecked. The primary mechanical indication is the presence of lateral subluxation of the femoral head with demonstrable "hinge abduction" on dynamic arthrography.
Radiographically, we rely heavily on the Lateral Pillar Classification described by Herring. Patients with Lateral Pillar Group B (where >50% of the lateral pillar height is maintained) or Group B/C border hips are the prime beneficiaries of containment surgery. If the lateral pillar is completely collapsed (Group C), the femoral head has often lost its spherical template entirely, and containment may be less effective, though a shelf can still be employed as a salvage procedure to relieve pain and provide a fulcrum for the hip. Furthermore, we look for signs of "head-at-risk" as described by Catterall, including Gage's sign, calcification lateral to the epiphysis, and a horizontal growth plate.
Contraindications must be strictly respected to avoid subjecting the child to unnecessary morbidity with no functional gain. The absolute contraindication is a stiff, incongruent hip where the femoral head cannot be concentrically reduced into the acetabulum even after extensive soft tissue releases (such as adductor or psoas tenotomies). If the hip hinges in abduction and cannot be seated, creating a shelf will simply lock the deformed head in a subluxated position, accelerating joint destruction. Chronological age also dictates contraindications; children under 6 years of age with mild to moderate involvement generally have an excellent prognosis with non-operative management due to their immense remodeling potential. Conversely, patients nearing skeletal maturity (e.g., >12-13 years) lack the biological plasticity required for the shelf to induce meaningful acetabular remodeling, making them better candidates for redirectional osteotomies (like a periacetabular osteotomy) or eventual arthroplasty.
| Parameter | Indications for Shelf Procedure | Contraindications for Shelf Procedure |
|---|---|---|
| Age | 6 to 11 years (optimal remodeling window) | < 6 years (spontaneous healing likely) or > 12 years (insufficient growth remaining) |
| Disease Stage | Fragmentation or early reossification | Late remodeling stage with established, fixed deformity (Stulberg V) |
| Lateral Pillar | Group B or B/C border (>50% height maintained) | Group C with severe, uncorrectable collapse (relative contraindication) |
| Joint Mechanics | Reducible lateral subluxation; dynamic hinge abduction | Irreducible subluxation; fixed incongruity preventing concentric seating |
| Necrosis Extent | > 50% involvement of the capital femoral epiphysis | < 50% involvement (often resolves with conservative management) |
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the bedrock of a flawless intraoperative execution. Our diagnostic workup begins with high-quality, orthogonal plain radiographs: a true anteroposterior (AP) view of the pelvis and a frog-leg lateral view of the affected hip. These images allow us to stage the disease according to Waldenström, assess the lateral pillar height, and quantify the degree of lateral extrusion using the Reimers migration index. However, plain films only tell a fraction of the story in the cartilaginous pediatric hip. We heavily rely on Magnetic Resonance Imaging (MRI) to delineate the true extent of the cartilaginous deformity, assess the viability of the unossified epiphysis, and evaluate the integrity of the labrum. MRI is unparalleled in differentiating true avascular necrosis from transient bone marrow edema syndromes.
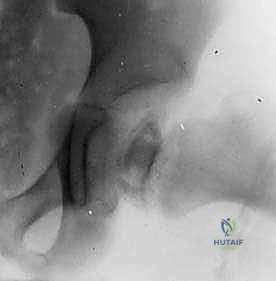
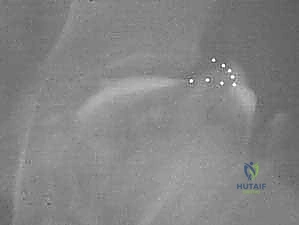
While advanced imaging is critical, the definitive decision-making tool is often the dynamic intraoperative arthrogram. Preoperatively, we plan for this crucial step. We anticipate injecting radio-opaque contrast directly into the joint space under fluoroscopy to visualize the cartilaginous profile of the femoral head and the acetabulum. The arthrogram dynamically assesses "hinge abduction"—we will actively abduct the hip under live fluoroscopy to see if the extruded lateral margin of the femoral head impinges on the acetabular rim, causing the medial joint space to widen as the head is levered out of the socket. If hinge abduction is present but the head can be concentrically reduced in flexion and internal rotation, the shelf procedure is perfectly indicated to provide lateral coverage and neutralize the hinge.
Patient positioning and operating room setup must be orchestrated with precision to facilitate both the surgical approach and unimpeded fluoroscopic access. The patient is placed supine on a fully radiolucent operating table. A critical step is the placement of a longitudinal gel roll or bump underneath the ipsilateral shoulder, thorax, and upper pelvis. This elevates the operative hemipelvis slightly, preventing the hip from rolling into external rotation and providing a more direct trajectory to the lateral ilium. Crucially, this bump must not extend distally beneath the greater trochanter or the hip joint itself, as this would artificially alter the hip's resting position, impede our ability to perform dynamic reduction maneuvers, and potentially compress posterior neurovascular structures.
The sterile preparation and draping must be expansive. We prep the entire affected lower extremity from the toes up to the costal margin, extending from the anterior midline of the abdomen to the posterior midline of the spine. The leg is draped free in a sterile stockinette to allow for unhindered, multidirectional manipulation of the hip during the arthrogram and subsequent graft placement. The fluoroscopic C-arm is brought in from the contralateral side of the table. Before the first incision is made, we must ensure that the C-arm can smoothly transition between true AP and cross-table lateral views of the hip without compromising the sterile field or encountering interference from the table's pedestal.
Step-by-Step Surgical Approach and Fixation Technique
With the patient optimally positioned and the surgical plan verified, we commence the procedure. The precision of our approach dictates the viability of the abductor musculature and the overall stability of the hip postoperatively.
1. Incision and Superficial Dissection:
We utilize a cosmetically favorable "bikini" incision. The incision begins approximately 1 cm inferior and slightly medial to the anterior superior iliac spine (ASIS) and extends laterally along the natural skin lines of Langer for about 3 to 5 cm, depending on the child's size.
Using electrocautery, we sharply divide the subcutaneous adipose tissue down to the glistening white fascia lata. At this juncture, surgical vigilance is paramount. We must identify and protect the lateral femoral cutaneous nerve (LFCN). We carefully incise the fascia lata in line with the skin incision, utilizing blunt dissection with a peanut sponge to sweep the subcutaneous fat and any traversing nerve branches medially. We identify the interval between the tensor fasciae latae (TFL) laterally and the sartorius medially.
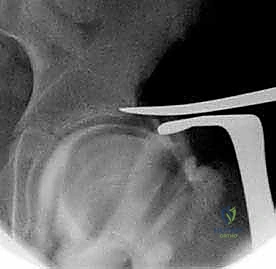
2. Deep Dissection and Capsular Exposure:
Developing the TFL-sartorius interval, we retract the TFL laterally and the sartorius medially. This exposes the rectus femoris muscle. We identify the direct head of the rectus originating from the AIIS and the reflected head coursing laterally over the anterior hip capsule. We carefully elevate the reflected head of the rectus femoris and retract it inferiorly. We then develop a triangular working interval bordered by the iliac wing medially, the hip capsule inferiorly, and the gluteus minimus laterally. We meticulously sweep the gluteus minimus off the superior capsule using a Cobb elevator, strictly avoiding any elevation of the abductor origins from the outer table of the ilium.
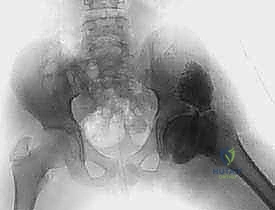
3. Intraoperative Arthrogram and Slot Creation:
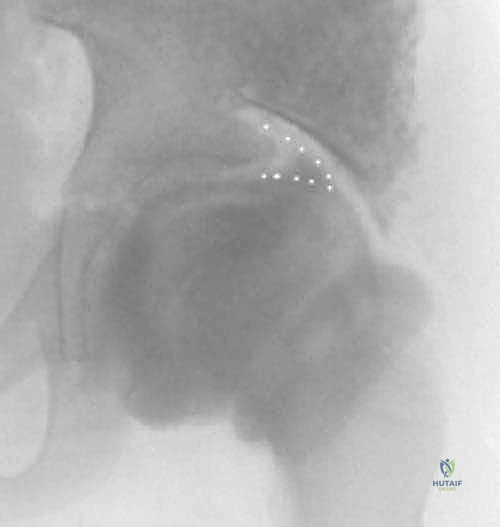
Before creating the shelf, we perform the dynamic arthrogram. A spinal needle is introduced into the joint space, and contrast is injected. Under live fluoroscopy, we assess the sphericity of the cartilaginous head and confirm the presence of hinge abduction. Once confirmed, we locate the exact superior margin of the acetabulum. Using a scalpel, we make a transverse incision through the pericapsular tissues precisely 2 to 3 mm superior to the capsulolabral junction.
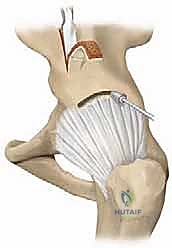
Using a sharp, curved osteotome, we create an extracapsular slot in the lateral cortex of the ilium. The osteotome is directed medially and slightly superiorly, parallel to the weight-bearing dome of the acetabulum, to a depth of approximately 10 to 15 mm. Fluoroscopy is used continuously to ensure the osteotome does not penetrate the joint space or violate the acetabular physis.
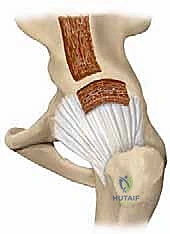
4. Graft Harvesting and Impaction:
We typically harvest an autologous bone graft from the outer table of the ipsilateral ilium, slightly superior to our working field, or utilize a structural cortical-cancellous allograft to minimize donor site morbidity. The graft is meticulously sculpted into a rectangular block, roughly 1.5 cm wide and 2.5 cm long, with a beveled medial edge.
The hip is placed in approximately 20 degrees of abduction and slight internal rotation to concentrically seat the femoral head. The sculpted graft is then firmly impacted into the prepared extracapsular slot. The graft should cantilever laterally, forming a robust roof directly over the extruded portion of the femoral head and intimately supporting the underlying capsule and labrum. The fixation is primarily achieved via a tight press-fit within the slot. In some variations, the reflected head of the rectus femoris is sutured over the graft to provide a vascularized soft-tissue tension band, further securing the construct. We confirm the final position and the elimination of hinge abduction with a final fluoroscopic sweep.
Complications, Incidence Rates, and Salvage Management
While the labral support procedure is a highly effective biologic intervention, it is not without inherent risks. A comprehensive understanding of potential complications is essential for preoperative patient counseling and intraoperative vigilance. The complications range from transient neuropraxias to structural failures of the graft construct, each requiring specific management strategies.
The most frequent complication, though often transient, is injury to the Lateral Femoral Cutaneous Nerve (LFCN). Given the nerve's variable arborization and proximity to the bikini incision, neuropraxia or complete transection can occur. Incidence rates vary in the literature but are reported between 5% and 15%. Patients present with numbness, tingling, or burning pain over the anterolateral thigh (meralgia paresthetica). Intraoperative prevention via meticulous blunt dissection is paramount. If neuropraxia occurs, management is largely expectant, with gabapentinoids utilized for severe dysesthesia; symptoms typically resolve within 3 to 6 months as the nerve regenerates.
Structural failure of the shelf construct—namely graft resorption, fracture, or displacement—represents a significant mechanical complication. Graft resorption occurs in approximately 5% to 8% of cases and is usually secondary to inadequate initial graft thickness, poor vascular bed preparation, or premature weight-bearing. If the graft resorbs before the femoral head has reossified, the hip will lose its lateral containment, and the hinge abduction will return. Prevention relies on using a robust, structurally sound graft (cortical-cancellous) and achieving a secure press-fit into a sufficiently deep (10-15mm) iliac slot. If significant displacement occurs early postoperatively, revision surgery to replace and secure the graft (potentially with K-wire augmentation) is indicated.
A late, yet critical, complication is the iatrogenic creation of Femoroacetabular Impingement (FAI), specifically pincer-type impingement. If the shelf graft is made excessively long or is angled too inferiorly, it can over-cover the femoral head. As the child reaches skeletal maturity, this prominent bony awning will impinge against the femoral neck during hip flexion and internal rotation, causing labral tears and early chondral damage. The incidence of symptomatic overcoverage requiring intervention is roughly 10%. Management involves arthroscopic or open surgical osteochondroplasty to trim the excess shelf bone and repair any associated labral pathology.
| Complication | Estimated Incidence | Prevention and Salvage Management |
|---|---|---|
| LFCN Neuropraxia | 5% - 15% | Prevention: Blunt dissection, identify/retract nerve. Management: Expectant, Gabapentin; usually resolves in 3-6 months. |
| Graft Resorption / Displacement | 5% - 8% | Prevention: Robust cortical-cancellous graft, deep slot (10-15mm), strict NWB post-op. Management: Revision grafting if early loss of containment occurs. |
| Iatrogenic FAI (Overcoverage) | ~10% (Late) | Prevention: Avoid excessive graft length; ensure parallel trajectory to acetabular dome. Management: Arthroscopic/open rim trimming (osteochondroplasty) at maturity. |
| Intra-articular Penetration | < 2% | Prevention: Continuous fluoroscopic guidance during slot creation; stay 2-3mm above capsule. Management: Immediate redirection; if cartilage damaged, monitor for early OA. |

Phased Post-Operative Rehabilitation Protocols
The success of the labral support procedure relies as much on disciplined postoperative rehabilitation as it does on intraoperative precision. The rehabilitation protocol is phased, designed to protect the vulnerable bone graft during its incorporation phase while aggressively preventing joint stiffness and muscle atrophy.
Phase I: Immediate Post-Operative Protection (Weeks 0-6)
The primary objective in the first six weeks is the absolute protection of the press-fit bone graft to ensure osseous integration. Historically, patients were placed in a 1.5 spica cast or an A-frame orthosis to maintain abduction. However, modern protocols favor a more forgiving approach, provided the patient is compliant. The patient is strictly Non-Weight-Bearing (NWB) on the operative extremity, utilizing bilateral axillary crutches or a pediatric walker. Pain is managed with a multimodal regimen including scheduled acetaminophen, NSAIDs (once the immediate risk of bleeding has passed, though some surgeons avoid NSAIDs to theoretically optimize bone healing), and short-acting opioids for breakthrough pain. Gentle, passive Range of Motion (ROM) exercises are initiated immediately, focusing on maintaining hip flexion, extension, and crucial internal rotation, while avoiding forced adduction that could stress the lateral graft.
Phase II: Intermediate Mobilization and Graft Incorporation (Weeks 6-12)
At the 6-week mark, AP and frog-leg lateral radiographs are obtained to assess graft incorporation and the maintenance of femoral head containment. If radiographic evidence of early bony union at the graft-host interface is present, the patient transitions to Partial Weight-Bearing (PWB). Physical therapy is intensified. Active-assisted and active ROM exercises are emphasized. Aquatic therapy is highly beneficial during this phase, as the buoyancy allows for concentric joint loading and gait mechanics training without subjecting the healing graft to full gravitational forces. Strengthening focuses on the gluteus medius and minimus to combat the inevitable abductor atrophy, utilizing isometric and isotonic exercises in a pain-free range.
Phase III: Full Weight-Bearing and Functional Restoration (Months 3-6)
Assuming complete graft incorporation is confirmed on the 12-week radiographs, the
