Mastering Kyphectomy in Myelomeningocele: An Intraoperative Guide
Key Takeaway
This intraoperative masterclass guides fellows through kyphectomy for myelomeningocele kyphosis. We cover comprehensive preoperative planning, meticulous surgical anatomy, and granular, real-time execution from incision to rod placement. Learn critical pearls, pitfalls, and post-operative care, ensuring optimal outcomes for these complex pediatric spinal deformities.
Welcome, colleagues, to the definitive discourse on one of the most mechanically demanding and physiologically profound interventions in pediatric spinal deformity surgery: the kyphectomy for severe congenital kyphosis in the setting of myelomeningocele (MMC). This is not merely a technical exercise in osteology; it is a life-altering reconstructive endeavor designed to restore global sagittal balance, mitigate progressive visceral compromise, and prevent devastating, potentially fatal complications. The surgical management of MMC-associated kyphosis demands a masterful understanding of dysplastic anatomy, meticulous preoperative optimization, and flawless intraoperative execution. Let us systematically deconstruct this complex pathology and its surgical remedy.
Comprehensive Introduction and Patho-Epidemiology
The kyphotic deformity observed in patients with myelomeningocele is a complex, early-onset, and rigidly progressive osseous defect. It most frequently manifests at the thoracolumbar junction, the mid-lumbar spine, or the lumbosacral junction. Unlike standard developmental or idiopathic kyphosis, the deformity in MMC is inherently congenital, driven by a fundamental failure of posterior vertebral migration and neural tube closure during early embryogenesis. This distinct congenital etiology dictates the rigidity of the curve, its relentless progression, and its profound resistance to non-operative management.
Embryologically, the development of the axial skeleton is inextricably linked to the closure of the neural tube. The notochord is designed to be covered dorsally by the progressive cephalic-to-caudal closure of the ectoderm. In myelomeningocele, a localized failure of this closure occurs, typically at the caudal aspect of the neural axis, leaving the neural placode exposed to the amniotic environment. This primary neuroectodermal failure precipitates a secondary mesodermal failure; it physically obstructs the normal posterior migration of the developing somites, which are destined to become the posterior bony elements and paraspinal musculature.
Consequently, the paraspinal muscles (primarily the erector spinae group) form in an anomalous lateral and anterior position relative to the instantaneous axis of rotation of the spine. With somatic growth and the child's attempt to adopt an upright sitting posture, these aberrantly positioned muscles act as active flexors rather than extensors. Compounded by the relentless vector of gravity, this muscular imbalance pulls the upper torso into an increasingly kyphotic, forward-flexed posture. This creates a biomechanical "vicious cycle" where the deformity exacerbates the mechanical advantage of the deforming forces, leading to rapid, inexorable progression.
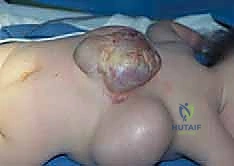
The consequences of untreated severe MMC kyphosis are systemic and catastrophic. The apex of the rigid kyphosis becomes a prominent, high-pressure bony fulcrum against insensate skin, leading to chronic breakdown, decubitus ulcers, and deep wound infections. Given the frequently tenuous dural closure from neonatal placode repair, these apical ulcers serve as a direct conduit for fatal central nervous system infections, including recurrent meningitis. Furthermore, the severe kyphosis radically diminishes the vertical height of the abdominal cavity. This visceral compression leads to disastrous kinking of the great vessels (aorta and inferior vena cava), severe gastrointestinal compromise (early satiety, intractable gastroesophageal reflux, malabsorption), and genitourinary dysfunction (urinary hydrostasis, recurrent pyelonephritis, and nephrolithiasis). Finally, the forward-flexed posture forces the abdominal contents cephalad, compressing the diaphragm and restricting lung expansion, culminating in Thoracic Insufficiency Syndrome. These children often become "functional quadriplegics," forced to rely entirely on their upper extremities to prop themselves up in a "marionette maneuver" just to breathe and maintain a visual horizon, severely stunting their cognitive and motor development.
Detailed Surgical Anatomy and Biomechanics
Navigating the surgical landscape of a myelomeningocele kyphectomy requires a profound appreciation for the highly dysplastic and distorted anatomy. The standard landmarks relied upon in routine spinal deformity surgery are either absent, grossly malformed, or perilously displaced.
Osteology and Bony Dysplasia
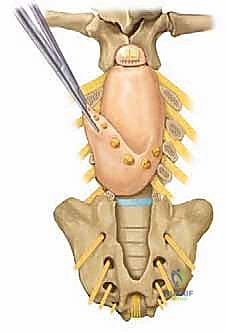
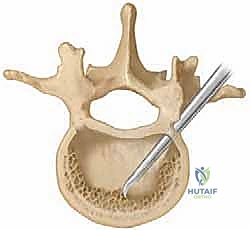
The kyphotic apex is characterized by an absence of posterior elements, including the spinous processes, laminae, and often the medial aspects of the pedicles. The pedicles that are present are typically widely splayed laterally, exhibiting a "teardrop" or severely flattened morphology on cross-sectional imaging. This lateral splaying creates significant bony ridges in the area of the diastasis, representing the rudimentary lateral elements that failed to migrate posteriorly. These ridges leave a paucity of viable bone stock for achieving a robust posterior fusion mass, severely complicating our reconstructive and instrumentation efforts. The vertebral bodies at the apex are often wedged anteriorly, further locking the deformity into a rigid configuration. In contrast, the sacral ala and the ilium often remain relatively well-formed, serving as critical, robust anchor points for our foundational pelvic fixation.
Soft Tissue and Muscular Intervals
The muscular anatomy is equally distorted. The erector spinae muscles, normally situated dorsal to the transverse processes to act as primary spinal extensors, are displaced laterally and ventrally. They lie anterior to the transverse processes, effectively converting them into powerful flexors of the spine. Dissecting these tissues requires meticulous technique; the surgeon must elevate these lateralized muscle bellies subperiosteally to expose the residual bony anatomy while preserving their vascular supply, as these muscle flaps are often critical for the final soft-tissue closure over the instrumentation. The overlying skin and subcutaneous tissues at the apex are frequently scarred, atrophic, and poorly vascularized from previous neonatal closures and chronic pressure, necessitating extreme care during the initial exposure and final closure.
Neurovascular Topography and Risks
The neural placode is typically tethered or physically incorporated into the dorsal aspect of the kyphotic apex. While the neural elements at and distal to the placode are generally non-functional in severe lumbar MMC, their preservation and careful handling are crucial to avoid ascending neurological deterioration or disruption of the fragile cerebrospinal fluid (CSF) dynamics. The dura mater surrounding the placode and the site of the previous myelomeningocele repair is characteristically thin, adherent, and exceedingly fragile. Incidental durotomies are a constant, high-probability risk and must be managed instantaneously with primary repair or patch grafting to prevent postoperative CSF fistulas.
Perhaps the most critical anatomical consideration involves the great vessels. The aorta and the inferior vena cava (IVC) generally do not follow the acute contours of the kyphotic apex. Instead, they "bowstring" across the concavity of the deformity. As the surgeon performs the apical resection and subsequently corrects the kyphosis by closing the osteotomy wedge, these vessels are subjected to acute stretching and potential kinking. This dynamic tension risks catastrophic rupture, intimal dissection, or complete occlusion of the vessels. Furthermore, the epidural venous plexus within the spinal canal and the basivertebral veins within the vertebral bodies are massively engorged due to altered hemodynamics and the loss of the normal epidural space. Aggressive decancellization or violation of the posterior vertebral body cortex can precipitate torrential, life-threatening hemorrhage.
Exhaustive Indications and Contraindications
The decision to proceed with a kyphectomy in a patient with myelomeningocele is monumental. It requires a delicate balance between the life-threatening natural history of the deformity and the profound physiological insult of the surgical intervention. Bracing is universally ineffective for congenital MMC kyphosis; it fails to halt progression and invariably causes devastating pressure necrosis over the insensate apical skin.
| Category | Specific Parameters | Rationale / Clinical Context |
|---|---|---|
| Primary Indications | Recurrent or recalcitrant apical skin breakdown | The rigid bony prominence acts as an internal pressure point against insensate skin. Chronic ulceration risks fatal CNS infection via the adjacent, often tenuous, dural closure. |
| Progressive Thoracic Insufficiency Syndrome | The severe forward flexion forces abdominal contents against the diaphragm, restricting pulmonary excursion. Correction restores abdominal height and improves vital capacity. | |
| Loss of sitting balance / "Marionette Maneuver" | Inability to sit independently without upper extremity support ("propping"). Correction frees the upper limbs for activities of daily living and environmental interaction. | |
| Severe visceral compression | Diminished abdominal height leads to gastroesophageal reflux, early satiety, malabsorption, and urinary stasis. Restoring trunk height alleviates these compressive phenomena. | |
| Relative Indications | Difficulty with hygiene and orthotic fitting | Severe deformity precludes the use of standard seating systems or lower extremity orthotics, severely limiting mobility and increasing the burden of care. |
| Absolute Contraindications | Active, untreated central nervous system infection | Operating in the presence of active meningitis or ventriculitis guarantees seeding of the massive spinal hardware construct. |
| Uncorrectable coagulopathy or profound malnutrition | The procedure entails massive blood loss and requires robust physiologic reserves for wound healing. Severe malnutrition (Albumin < 3.0 g/dL) guarantees wound failure. | |
| Medically unstable for prolonged anesthesia | Patients with severe, unoptimized congenital cardiac defects or end-stage pulmonary failure who cannot tolerate the hemodynamic shifts of major blood loss and positioning. |
Pre-Operative Planning, Templating, and Patient Positioning
Success in this high-stakes arena is forged in the crucible of meticulous preoperative planning. A multidisciplinary approach is mandatory, involving pediatric orthopedic surgery, neurosurgery, plastic surgery, urology, and specialized pediatric anesthesiology.
Multidisciplinary Assessment and Optimization
A thorough history and physical examination must specifically interrogate associated anomalies. The presence of a functioning ventriculoperitoneal (VP) shunt must be confirmed by neurosurgery, as altered CSF dynamics postoperatively can precipitate acute hydrocephalus. Urological evaluation, often including a renal ultrasound, is critical to rule out severe hydronephrosis or renal calculi resulting from chronic urinary stasis. Baseline pulmonary function tests (if the child is cooperative) and a thorough nutritional assessment are non-negotiable. Malnutrition must be aggressively treated, often requiring hyperalimentation via a gastrostomy tube for several months prior to surgical intervention to optimize the physiological envelope for healing. Plastic surgery consultation is paramount; local tissue rearrangement, rhomboid flaps, or the preoperative placement of tissue expanders in the posterolateral flanks may be required to ensure tension-free closure over the prominent hardware.
Radiographic Blueprinting
The imaging workup must be exhaustive. Standard upright and supine anteroposterior (AP) and lateral radiographs define the global sagittal balance and the magnitude of the deformity under the influence of gravity.
Supine bending or traction films are essential to differentiate the rigid, structural component of the curve from the flexible, reducible component.
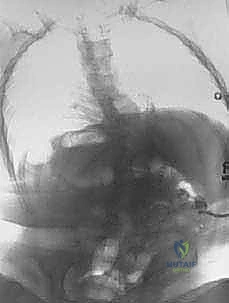
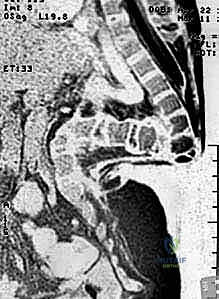
High-resolution, three-dimensional computed tomography (CT) is the cornerstone of preoperative templating. The CT scan delineates the highly dysplastic osseous anatomy, the exact dimensions of the splayed pedicles, the location of the apical vertebral bodies, and the available bone stock for fixation. This allows for precise, screw-by-screw trajectory planning.
Magnetic resonance imaging (MRI) of the entire neural axis is mandatory to assess for concurrent neurosurgical pathologies such as Chiari malformations, syringomyelia, or a tethered spinal cord.
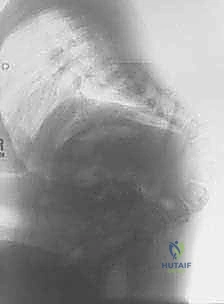
The relationship of the neural placode to the apical vertebrae must be clearly defined to plan the dural resection or mobilization strategy.
Hemodynamic Preparation and Patient Positioning
Given the anticipated massive blood loss, robust intravenous access is critical. Bilateral large-bore peripheral IVs, a central venous catheter, and an arterial line are standard. Intraoperative neurophysiological monitoring (IONM), including somatosensory evoked potentials (SSEPs) and motor evoked potentials (MEPs), is utilized to monitor the proximal, functional spinal cord, although signals from the lower extremities are typically absent in severe lumbar MMC.
Patient positioning is a highly orchestrated procedure. The patient is carefully log-rolled onto a specialized radiolucent spinal frame (e.g., Jackson table or OSI frame) that allows the abdomen to hang freely.
This free-hanging abdomen is vital to decompress the inferior vena cava, thereby reducing epidural venous engorgement and mitigating catastrophic intraoperative bleeding.
Meticulous padding of all bony prominences, particularly the insensate lower extremities, is essential to prevent iatrogenic pressure necrosis during the prolonged surgical duration.
The C-arm fluoroscopy unit is draped and brought into the field early to verify that orthogonal views of the entire planned construct can be obtained without obstruction.

Step-by-Step Surgical Approach and Fixation Technique
The surgical execution of a myelomeningocele kyphectomy is a masterclass in controlled aggression, requiring precise anatomical dissection and decisive deformity correction.
Exposure and Dural Management
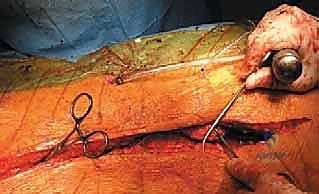
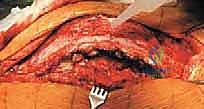
The procedure commences with an elliptical incision encompassing the previous surgical scars and any areas of atrophic, compromised skin overlying the apex. The dissection proceeds laterally, identifying the fascial planes. The laterally displaced erector spinae muscles are meticulously elevated subperiosteally from the rudimentary lateral masses and transverse processes. This dissection must be kept strictly subperiosteal to minimize bleeding and preserve the muscular blood supply.
The neural placode and the surrounding thin dura are identified. In severe, rigid deformities where the neural elements distal to the apex are completely non-functional, a controversial but often necessary step is the transection and ligation of the dural sac. The dura is carefully opened, and the non-functional neural elements are sharply divided. The proximal dural stump is then meticulously closed with a watertight, continuous non-absorbable suture. Two independent ligatures are often placed to ensure absolute hemostasis and prevent postoperative CSF leakage, which is a leading cause of deep surgical site infection in this population.
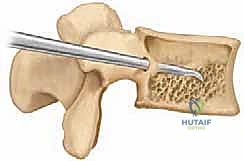
Apical Resection (The Kyphectomy)
With the neural axis managed and the bony anatomy exposed, the apical resection begins. The goal is to perform a closing-wedge osteotomy or a formal apical vertebrectomy to shorten the spinal column and allow for extension of the spine without stretching the anterior neurovascular structures.
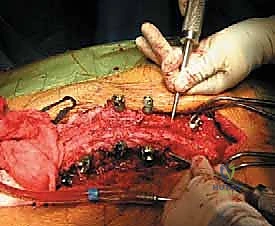
Using Leksell rongeurs, high-speed burrs, and ultrasonic bone scalpels, the rudimentary posterior elements and the lateral masses of the apical vertebrae (typically 1 to 3 vertebral levels) are resected.

The dissection is carried ventrally, subperiosteally along the lateral walls of the vertebral bodies, staying strictly within the periosteal sleeve to protect the laterally displaced great vessels.
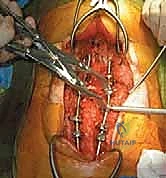
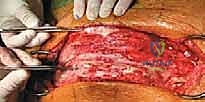
The apical vertebral bodies are then decancellized or completely excised.
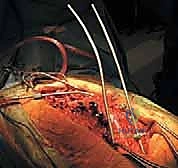
This is the most hemorrhagic phase of the operation. Copious use of bone wax, bipolar electrocautery, and topical hemostatic agents (e.g., Floseal, Surgiflo) is required to manage bleeding from the basivertebral veins. The anterior longitudinal ligament is often preserved if possible to act as a hinge, though in severe deformities, it may need to be sectioned to achieve adequate mobility.
Deformity Correction and Instrumentation
Once the apical resection is complete and the spine is mobilized, the correction is achieved by slowly extending the patient's hips and lower extremities, effectively closing the anterior wedge osteotomy. This maneuver must be performed under direct vision, carefully monitoring the anterior soft tissues and the great vessels to ensure they are not kinked or subjected to excessive tension.

Fixation is achieved using a robust, segmental pedicle screw construct. Given the poor bone quality and the massive cantilever forces involved, the construct must span multiple levels proximal to the apex and have absolute, rigid fixation distally in the pelvis.
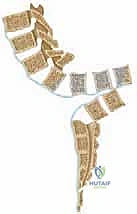
S2-Alar-Iliac (S2AI) screws or traditional iliac screws are mandatory to provide a stable foundation. The proximal pedicle screws are placed into the relatively normal thoracic or upper lumbar vertebrae.
The rods are contoured to the corrected sagittal profile and secured, locking the spine into its new, balanced alignment. A thorough decortication of the remaining bony elements is performed, and massive amounts of autograft (obtained from the resected apical vertebrae) and allograft are packed into the defect to promote a solid arthrodesis.
Soft Tissue Closure
The closure is as critical as the bony reconstruction.
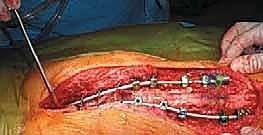
The previously elevated erector spinae muscles are mobilized medially and approximated in the midline over the hardware to provide a robust, vascularized soft-tissue envelope.

If the skin edges cannot be approximated without tension, the plastic surgery team will perform local tissue rearrangements, rotation flaps, or utilize the previously expanded tissue to achieve a durable, tension-free skin closure. Multiple subfascial and subcutaneous drains are placed to prevent hematoma formation.
Complications, Incidence Rates, and Salvage Management
The complication profile for MMC kyphectomy is among the highest in all of orthopedic surgery. The surgeon must be acutely aware of these risks and possess the armamentarium to manage them decisively.
| Complication | Estimated Incidence | Pathophysiology and Salvage Management |
|---|---|---|
| Massive Hemorrhage | 80 - 100% | Driven by engorged epidural/basivertebral veins and extensive osteotomies. Management: Preoperative optimization, routine use of antifibrinolytics (Tranexamic Acid), intraoperative cell salvage, rapid infusion protocols, and meticulous sequential hemostasis with bone wax and thrombin matrices. |
| Deep Surgical Site Infection | 15 - 25% | Multifactorial: poor soft tissue envelope, proximity to the perineum, neurogenic bladder, and potential CSF leaks. Management: Aggressive radical debridement, hardware retention (if stable), placement of antibiotic-impregnated beads/sponges, prolonged culture-directed IV antibiotics, and plastic surgery flap coverage. |
| Cerebrospinal Fluid (CSF) Leak | 10 - 20% | Resulting from violation of the fragile, adherent dura at the previous placode repair site or failure of the dural ligation. Management: Primary watertight repair, use of dural sealants, fascial patch grafts. Postoperatively, ensuring optimal VP shunt function to decrease thecal sac pressure is mandatory. |
| Pseudarthrosis / Hardware Failure | 10 - 15% | Driven by poor local bone stock, massive cantilever forces, and impaired biology. Often presents as rod fracture or screw pullout. Management: Revision surgery with extension of the fusion construct, aggressive decortication, bone morphogenetic protein (BMP) application, and potentially anterior column support if feasible. |
| Great Vessel Injury / Ischemia | < 5% | Caused by aggressive retraction or acute stretching of the bowstrung aorta/IVC during deformity correction. Management: Immediate release of the correction, vascular surgery consultation, primary repair of the vessel. Continuous intraoperative monitoring of lower extremity pulse oximetry and arterial lines is critical for early detection. |
| Skin Necrosis / Wound Dehiscence | 15 - 30% | Resulting from excessive tension on the compromised apical skin over prominent hardware. Management: Immediate debridement of necrotic tissue, vacuum-assisted closure (VAC) therapy (used cautiously over exposed dura), and definitive coverage with myocutaneous rotation flaps. |
Phased Post-Operative Rehabilitation Protocols
The postoperative management of the MMC kyphectomy patient is a delicate balance between protecting the massive surgical reconstruction and preventing the complications of prolonged immobility.
Phase I: Acute Inpatient Management (Days 0 - 14)
The immediate postoperative period is focused on hemodynamic stability, wound protection, and aggressive pulmonary toilet. Patients are typically managed in the Pediatric Intensive Care Unit (PICU) for the first 48 to 72 hours. They are kept strictly flat or log-rolled to prevent any sheer forces on the delicate soft-tissue closure and the nascent bony fusion. Drains are maintained until output is minimal (typically < 30 cc/shift) to prevent epidural hematoma and reduce tension on the skin closure. Broad-spectrum intravenous antibiotics are continued until all drains are removed, and often transitioned to culture-directed oral therapy for several weeks based on intraoperative tissue cultures.
Phase II: Mobilization and Orthotic Management (Weeks 2 - 12)
Once the soft tissue envelope demonstrates primary healing and the drains are removed, gradual mobilization is initiated. A custom-molded Thoracolumbosacral Orthosis (TLSO) is mandatory for all out-of-bed activities. The TLSO is designed to distribute pressure evenly and protect the instrumentation from catastrophic cantilever failure during transfers. Sitting time is strictly graduated, beginning with 15-30 minute intervals and slowly advancing as tolerated, with meticulous inspection of the skin after every sitting session. Physical therapy focuses on upper extremity strengthening and safe transfer techniques.
Phase III: Long-Term Surveillance and Integration (Months 3 - 24+)
Radiographic evaluation is performed at 6 weeks, 3 months, 6 months, 1 year, and 2 years postoperatively to assess the progression of the fusion mass and monitor for hardware failure or loss of correction. The TLSO is typically weaned between 6 and 9 months once robust bridging trabecular bone is visible on plain radiographs or CT scan. Long-term surveillance remains critical, as these patients are at a lifetime risk for late-onset infections, adjacent segment disease, and recurrent skin breakdown if their seating systems are not continuously optimized to their new sagittal profile.
Summary of Landmark Literature and Clinical Guidelines
The evolution of the myelomeningocele kyphectomy reflects a journey from high-mortality, uninstrumented resections to modern, highly rigid, segmentally instrumented reconstructions.
Historically, the foundational work by Sharrard (1968) introduced the concept of neonatal kyphectomy, advocating for early resection of the apical deformity at the time of initial placode closure to facilitate skin coverage and prevent the vicious cycle of deformity progression. However, neonatal procedures were plagued by high rates of recurrent kyphosis due to the lack of rigid internal fixation and the continued growth of the anterior column.
Lindseth (1979) revolutionized the approach in the older child by introducing the concept of the apical vertebrectomy combined with posterior wire fixation. This technique addressed the fundamental length mismatch between the anterior and posterior columns. Warner and Fackler (1993) further advanced the biomechanical stability of the construct by popularizing the Luque-Galveston technique, driving the rods deep into the ilium to counteract the massive cantilever forces that routinely caused proximal pullout in shorter constructs.
In the modern era, the consensus guidelines, driven by multi-center pediatric spine study groups, universally mandate the use of rigid, segmental pedicle screw instrumentation extending from the stable thoracic spine to the pelvis. The advent of S2-Alar-Iliac (S2AI) screws has significantly decreased the profile of the pelvic fixation, reducing the risk of hardware prominence and subsequent skin breakdown compared to traditional iliac screws. Current literature strongly emphasizes the necessity of the multidisciplinary approach, demonstrating that preoperative nutritional optimization, aggressive management of hydrocephalus, and the integration of plastic surgery for soft-tissue coverage are equally as critical to a successful outcome as the orthopedic reconstruction itself.
Mastering the MMC kyphectomy requires a synthesis of this historical context, a profound respect for the dysplastic anatomy, and the technical fortitude to execute a massive physiological reconstruction. It remains one of the most challenging, yet undeniably transformative, procedures in the armamentarium of the pediatric spinal surgeon.
