Knee Arthrofibrosis Release: An Intraoperative Masterclass for Restoring Motion
Key Takeaway
Join us in the OR for a masterclass on knee arthrofibrosis release. This guide details preoperative planning, precise patient positioning, and a granular, step-by-step arthroscopic technique. Learn to identify and address diffuse adhesions, restore patellar mobility, and manage posterior capsular contractures. We'll cover comprehensive surgical anatomy, neurovascular risks, and essential pearls for achieving optimal patient outcomes in knee motion restoration.
Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues, to the operating theater. Today, we are undertaking a rigorous, systematic approach to one of the most challenging and immensely rewarding procedures in our specialty: the arthroscopic release for knee loss of motion, clinically categorized as arthrofibrosis. This is a debilitating condition that, if left unaddressed, relentlessly degrades a patient's quality of life, leading to profound functional deficits, altered gait mechanics, and chronic, recalcitrant pain. Our operative mandate is to systematically deconstruct the pathologic fibrous tissue and restore the intricate, multi-planar kinematics of the knee joint, ensuring the achievement of a full, pain-free range of motion.
Before we make our initial incisions, it is imperative to establish a precise taxonomy and understand the underlying molecular and macroscopic pathology. "Loss of motion" is a descriptive umbrella term encompassing deficits in flexion, extension, or a combination thereof; it does not inherently imply a discrete etiology. A flexion contracture specifically denotes a loss of terminal extension, frequently resulting from the contracture or adaptive shortening of the posterior capsuloligamentous complex or the hamstring musculature. Arthrofibrosis, our primary surgical target today, describes a profound restriction in knee motion driven by the diffuse proliferation of intra-articular adhesions and dense fibrotic tissue. It represents a pathologic internal scarring process that obliterates the normal recesses and gliding planes of the knee. In its most severe, unremitting chronic form, arthrofibrosis can progress to ankylosis—the complete immobility of the joint mediated by dense fibrous, cartilaginous, or even heterotopic bony bridging.
Beyond diffuse arthrofibrosis, we must recognize distinct, localized fibrotic syndromes. The ACL Nodule, classically termed a Cyclops Lesion, is a focal, dense fibrous scar that typically forms following anterior cruciate ligament (ACL) reconstruction. Pathognomonically located anterolateral to the tibial tunnel, this nodule mechanically impinges within the intercondylar notch during terminal extension, creating a hard, mechanical block. Another critical entity is Infrapatellar Contracture Syndrome (IPCS). This involves pathologic fibrous hyperplasia of the anterior infrapatellar fat pad (Hoffa's fat pad), leading to the rigid adherence of the patellar tendon to the anterior tibia. This tethering severely limits patellar excursion, alters patellofemoral contact forces, and frequently results in a permanent patella infera (baja) if not aggressively released.
The pathogenesis of knee motion loss is a complex, multifactorial cascade, frequently initiated as a catastrophic complication of knee ligament trauma or surgical reconstruction. At the cellular level, an exaggerated inflammatory response triggers the localized over-expression of profibrotic cytokines, notably Transforming Growth Factor-beta (TGF-$\beta$) and Platelet-Derived Growth Factor (PDGF). This cytokine storm stimulates the aggressive proliferation of myofibroblasts and the disorganized deposition of Type I and Type III collagen within the joint space. Clinical risk factors for this fibrotic cascade include operating during the acute inflammatory phase of an injury, prolonged postoperative immobilization, suboptimal graft positioning or overtensioning during ligamentous reconstruction, and the presence of underlying complex regional pain syndrome (CRPS).
Detailed Surgical Anatomy and Biomechanics
The knee joint, while historically simplified as a ginglymus (hinge-type) articulation, is biomechanically exquisite, requiring a minimum of six degrees of freedom to execute normal ambulation and weight-bearing tasks. It comprises three distinct, yet functionally interdependent articulations: the patellofemoral joint, the medial tibiofemoral joint, and the lateral tibiofemoral joint. The capsular envelope extends expansively from the suprapatellar pouch proximally to the intricate posteromedial and posterolateral recesses distally. Pathologic fibroblastic proliferation can occur anywhere within these anatomic confines, obliterating the essential gliding planes.
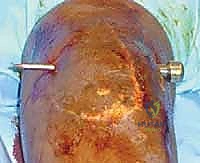
The suprapatellar pouch is a critical recess for knee flexion. Anatomically, it extends 3 to 4 centimeters proximal to the superior pole of the patella. It is bounded anteriorly by the quadriceps tendon and patella, and posteriorly by the anterior cortex of the distal femur. The vastus intermedius muscle originates from the anterior femoral shaft and forms the dynamic floor of the quadriceps tendon. During normal flexion, this pouch unfolds, allowing the extensor mechanism to glide smoothly over the trochlea. In an arthrofibrotic knee, dense adhesions obliterate this space, tethering the quadriceps tendon to the femur and effectively acting as a mechanical checkrein to flexion. Surgical dissection here must be meticulous to avoid iatrogenic injury to the quadriceps tendon or the vastus intermedius aponeurosis.
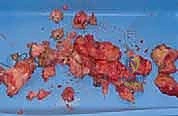
The anterior interval, encompassing the infrapatellar fat pad and the pretibial recess, is equally vital for normal kinematics. This space lies inferior to the patella, posterior to the patellar tendon, and anterior to the tibia. Hoffa's fat pad is a highly vascularized and innervated structure that facilitates the smooth excursion of the patellar tendon during flexion and extension. When fibrosis invades this interval—characteristic of Infrapatellar Contracture Syndrome—the patellar tendon adheres rigidly to the anterior tibia. This shortens the effective length of the tendon, dragging the patella distally (patella infera) and exponentially increasing patellofemoral joint reaction forces. Aggressive débridement of this space is required, but the surgeon must remain hyper-vigilant to protect the intermeniscal (transverse) ligament and the anterior horns of the menisci.
The posterior compartment anatomy presents the highest risk profile during arthrofibrosis release. The posteromedial interval lies between the superficial medial collateral ligament (sMCL) anteriorly and the pes anserine tendons posteriorly, harboring the medial head of the gastrocnemius and the posterior oblique ligament (POL). The posterolateral approach courses over the anterior aspect of the biceps femoris tendon. Here, the common peroneal nerve is the critical structure at risk as it wraps around the fibular neck. Furthermore, aggressive posterior capsulotomies or notchplasties place the popliteal artery and vein—located immediately posterior to the posterior cruciate ligament (PCL) and posterior capsule—in imminent danger. A thorough command of these neurovascular intervals is non-negotiable for a safe and effective release.
Exhaustive Indications and Contraindications
The decision to proceed with operative intervention for arthrofibrosis must be carefully weighed, balancing the profound functional impairment of the patient against the inherent risks of exacerbating the inflammatory cascade. Surgical management is not a first-line treatment; it is reserved for patients who have plateaued after a dedicated, aggressive trial of non-operative management. The timing of the intervention is as critical as the technical execution. Operating on a "hot," acutely inflamed knee is a guaranteed catalyst for recurrent, often more severe, fibrotic proliferation.
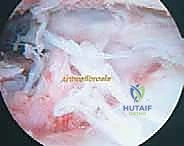
Absolute indications for arthroscopic arthrofibrosis release include a persistent loss of flexion greater than 10 to 15 degrees, or an extension deficit greater than 5 to 10 degrees, that remains refractory to a minimum of 8 to 12 weeks of intensive, supervised physical therapy. A localized mechanical block, such as a symptomatic cyclops lesion confirmed on magnetic resonance imaging (MRI) and correlating with a hard endpoint in terminal extension, is a definitive indication for localized arthroscopic excision. Furthermore, progressive patella infera secondary to infrapatellar contracture syndrome warrants prompt surgical release to prevent irreversible patellofemoral chondral degradation.
Contraindications must be strictly observed to prevent catastrophic outcomes. The most critical absolute contraindication is the presence of an active intra-articular infection, which must be eradicated with appropriate surgical débridement and targeted antimicrobial therapy prior to any reconstructive or motion-restoring procedures. Similarly, operating during an active flare of Complex Regional Pain Syndrome (CRPS) or Reflex Sympathetic Dystrophy (RSD) is contraindicated. These patients require aggressive preoperative optimization, often involving regional sympathetic blocks and pharmacological modulation, to quiet the central nervous system before any surgical trauma is introduced.
| Parameter | Indications for Arthrofibrosis Release | Contraindications for Arthrofibrosis Release |
|---|---|---|
| Range of Motion | Flexion deficit > 10-15°; Extension deficit > 5-10° | Functional ROM for patient's baseline demands |
| Conservative Care | Failure of 8-12 weeks of aggressive, guided PT | Inadequate trial of non-operative management |
| Pathology | Symptomatic Cyclops Lesion; Infrapatellar Contracture | Active intra-articular infection (Absolute) |
| Inflammatory State | "Cold," non-inflamed joint (resolved acute phase) | Acute inflammatory phase; "Hot" swollen joint |
| Neurologic Status | Stable neurovascular baseline | Active, uncontrolled CRPS/RSD (Absolute) |
Patient psychology and compliance also play a massive role in patient selection. A relative contraindication is a patient who demonstrates a profound lack of compliance or an inability to tolerate the intense, painful, and prolonged postoperative rehabilitation protocol. The surgical release is merely the first step in a long continuum of care; without immediate and relentless postoperative physical therapy, the joint will inevitably re-stiffen, rendering the surgical intervention futile.
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning is the absolute cornerstone of a successful arthrofibrosis release. The evaluation begins with a meticulous clinical assessment, quantifying the precise degrees of flexion and extension loss using a goniometer. The surgeon must differentiate between a soft, resilient endpoint (suggesting muscular tightness or capsular contracture) and a hard, abrupt endpoint (suggesting a bony block, hardware impingement, or a dense cyclops lesion). Patellar mobility must be rigorously tested; restricted superior-inferior glide or the inability to tilt the patella are hallmark signs of anterior interval scarring and retinacular contracture.
Advanced imaging is mandatory to define the architectural distortion within the joint. Plain radiographs (anteroposterior, lateral, sunrise, and notch views) are scrutinized for hardware failure, heterotopic ossification, osteochondral defects, and joint space narrowing. The lateral radiograph is critical for assessing patellar height using the Insall-Salvati or Caton-Deschamps ratios to identify patella infera. Magnetic Resonance Imaging (MRI) remains the gold standard for soft tissue evaluation. It allows the surgeon to visualize the extent of capsular thickening, the presence of a cyclops lesion in the intercondylar notch, the integrity of previous ligamentous grafts, and the degree of obliteration within the suprapatellar pouch and anterior interval.
The Examination Under Anesthesia (EUA) is an indispensable, dynamic component of the preoperative planning phase, performed immediately prior to prepping the limb. With the patient fully relaxed and devoid of muscular guarding, the true, unrestrained range of motion is documented. The surgeon gently manipulates the knee, assessing for crepitus, mechanical blocks, and the precise limits of flexion and extension. This baseline EUA is critical; it distinguishes true mechanical arthrofibrosis from pain-mediated stiffness and sets the intraoperative goal for motion restoration.
Patient positioning and anesthetic management are tailored to facilitate both the intraoperative release and the immediate postoperative rehabilitation. We strongly advocate for regional anesthesia, such as an indwelling epidural or continuous peripheral nerve blocks (e.g., adductor canal block), to provide profound intraoperative muscle relaxation and paramount postoperative analgesia. The patient is positioned supine with a lateral post to provide a stable fulcrum for valgus and varus stress. A pneumatic tourniquet is applied to the proximal thigh but is typically left deflated. Operating without a tourniquet allows the surgeon to distinguish highly vascularized, hyperemic scar tissue from normal, avascular capsular structures, though the tourniquet remains available if visualization is compromised by excessive bleeding.
Step-by-Step Surgical Approach and Fixation Technique
The intraoperative execution of an arthrofibrosis release requires meticulous precision, patience, and a deep respect for the compromised intra-articular anatomy. The procedure is inherently dangerous due to the loss of normal anatomic landmarks and the profound contraction of the joint space. We proceed systematically, compartment by compartment, to sequentially dismantle the fibrotic constraints.
Establishing initial portal access in a severely arthrofibrotic knee is exceptionally challenging and carries a high risk of iatrogenic chondral injury. The joint capsule is often completely scarred down to the femoral condyles and patella. To mitigate this risk, we perform aggressive capsular distention by injecting 120 to 180 mL of sterile saline through a superolateral approach before any instruments are introduced. We establish a superolateral portal for high-flow inflow, followed by standard inferolateral (viewing) and inferomedial (working) portals. If the anterior interval is completely obliterated, a "blind" insertion of the arthroscope is strictly avoided; instead, we use a blunt trocar to gently sweep and create a rudimentary viewing space posterior to the patellar tendon.
The systematic release begins in the suprapatellar pouch, which is frequently the primary culprit in flexion loss. The arthroscope is directed superiorly. In a fibrotic knee, the normal vastus intermedius gliding plane is replaced by dense, white, avascular scar tissue bridging the anterior femur to the quadriceps tendon. Using a combination of blunt sweeping with the arthroscope sheath, motorized shavers, and radiofrequency ablation wands, we meticulously re-establish this pre-femoral space. The dissection must remain strictly intra-articular, carefully peeling the adhesions off the anterior femur while vehemently protecting the overlying quadriceps tendon from thermal or mechanical perforation.
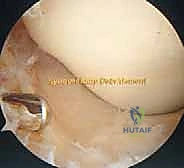
Once the suprapatellar pouch is reconstituted, attention is turned to the medial and lateral gutters. Adhesions in these recesses tether the patellar retinaculum and the peripheral margins of the menisci, severely restricting patellar mobility and tibiofemoral rotation. The medial and lateral retinacular adhesions are released using electrocautery, taking extreme care to achieve hemostasis of the superior and inferior genicular vessels. The menisci must be visually confirmed and freed from any capsular tethering, restoring their normal translation during the arc of motion.

The anterior interval and infrapatellar fat pad are addressed next. This is a critical step for restoring patellar tracking and addressing patella infera. The dense fibrotic tissue binding the posterior aspect of the patellar tendon to the anterior tibia is resected. This "Millett release" requires careful, deliberate use of a radiofrequency wand to vaporize the scar tissue while protecting the underlying anterior transverse meniscal ligament. As this interval is cleared, the surgeon will often notice an immediate, palpable improvement in superior patellar glide.
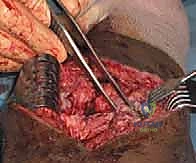
Finally, the arthroscope is advanced into the intercondylar notch to address extension deficits. The notch is frequently obscured by hypertrophic synovial tissue or a discrete cyclops lesion. The cyclops nodule is systematically excised using a motorized shaver and biting punches until the anterior aspect of the ACL graft (or native ACL) is clearly visualized and free of impingement against the intercondylar roof during terminal extension. If a posterior capsular contracture is present, posteromedial and posterolateral portals may be necessary to elevate the posterior capsule off the posterior femoral condyles. This is an advanced maneuver requiring absolute vigilance to protect the popliteal neurovascular bundle lying immediately extra-capsular.

Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, arthroscopic release of arthrofibrosis carries a distinct and formidable complication profile. The surgeon must be acutely aware of these risks, employ rigorous preventative strategies, and possess the technical acumen to manage them should they arise. The tight, distorted anatomy inherently increases the margin for error compared to routine knee arthroscopy.
Iatrogenic chondral injury is arguably the most frequent intraoperative complication, occurring primarily during the establishment of portals or the initial insertion of instruments into an obliterated joint space. The incidence can be as high as 5-10% in severe cases of ankylosis. Prevention relies on aggressive preoperative joint distention and the strict avoidance of forceful, blind instrument insertion. If a significant full-thickness chondral defect is created, salvage management may require microfracture, chondroplasty, or, in severe cases, delayed cartilage restoration procedures, though these are heavily complicated by the underlying fibrotic tendency of the joint.
Neurovascular complications, while less frequent (incidence < 1%), are potentially catastrophic. The popliteal artery and vein are at extreme risk during posterior capsular releases or aggressive notchplasties. The common peroneal nerve is vulnerable during posterolateral portal placement and lateral capsular releases, while the saphenous nerve and its infrapatellar branch are frequently irritated or transected during medial portal placement, leading to painful neuromas or localized numbness. Salvage management for major vascular injury requires immediate open exploration and vascular repair. Nerve injuries are typically managed expectantly, though painful neuromas may eventually require surgical excision.
| Complication | Estimated Incidence | Preventative Strategy | Salvage Management / Treatment |
|---|---|---|---|
| Iatrogenic Chondral Injury | 5 - 10% | Aggressive joint distention; blunt trocars; avoid blind insertion | Chondroplasty; Microfracture; Delayed cartilage restoration |
| Extensor Mechanism Rupture | < 1% | Avoid aggressive manipulation; protect quad tendon during pouch release | Immediate open primary repair of quad/patellar tendon |
| Neurovascular Injury | < 1% | Meticulous portal placement; strict adherence to anatomic boundaries | Immediate vascular surgery consult; Neuroma excision |
| Recurrent Arthrofibrosis | 15 - 30% | Immediate post-op CPM; aggressive pain control; strict PT compliance | Revision release; Manipulation under anesthesia; CRPS management |
| Hemarthrosis / Infection | 1 - 3% | Meticulous hemostasis (genicular arteries); prophylactic antibiotics | Joint aspiration; Arthroscopic lavage and targeted antibiotics |
Recurrent arthrofibrosis is the most frustrating complication for both the surgeon and the patient, with incidence rates ranging from 15% to 30% depending on the severity of the initial pathology and patient compliance. The surgical trauma inherently triggers a new inflammatory cascade. If the patient fails to mobilize the joint immediately, the fibrotic tissue will rapidly reform. Salvage management for recurrence involves a multidisciplinary approach: aggressive pain management (often involving pain specialists for sympathetic blocks), potential manipulation under anesthesia (MUA) within the first 4-6 weeks, or, in refractory cases, a revision arthroscopic release once the joint has completely "cooled down."
Phased Post-Operative Rehabilitation Protocols
The surgical release of arthrofibrosis represents merely 20% of the therapeutic equation; the remaining 80% is predicated entirely on a rigorous, unrelenting, and perfectly executed postoperative rehabilitation protocol. The primary objective is to maintain the range of motion achieved intraoperatively while managing the inevitable postoperative pain and inflammatory effusion. A paradigm shift in patient mindset is required: the knee must be moved immediately and continuously.
Phase I (Immediate Post-Operative Phase, Days 0-7) focuses on aggressive motion preservation and profound pain control. We routinely utilize indwelling epidural catheters or continuous peripheral nerve blocks to blunt the nociceptive response, allowing the patient to tolerate intensive physical therapy. Continuous Passive Motion (CPM) machines are initiated in the recovery room, set to the maximum range of motion achieved during the intraoperative EUA. The patient is instructed to use the CPM for 12 to 16 hours a day. Concurrently, physical therapists initiate aggressive patellar mobilization techniques (superior, inferior, medial, and lateral glides) to prevent the recurrence of anterior interval and retinacular tethering. Cryotherapy and high-voltage galvanic stimulation are employed to control edema and joint effusion.
Phase II (Early Rehabilitation, Weeks 1-4) transitions the patient towards active and active-assisted range of motion. The reliance on the CPM is gradually weaned as the patient demonstrates the ability to independently achieve and maintain the target motion parameters. Closed kinetic chain exercises, such as wall slides and stationary cycling (initially with pendulum arcs, progressing to full revolutions), are introduced to stimulate quadriceps activation without placing excessive shear forces on the patellofemoral joint. Aggressive hamstring stretching is mandatory to combat flexion contractures. The therapist must remain vigilant; any loss of motion during this phase is a critical warning sign that may necessitate an early manipulation under anesthesia.
Phase III (Strengthening and Return to Function, Months 2-6) focuses on restoring muscular strength, endurance, and proprioception. Once full, symmetric range of motion is achieved and the joint effusion has resolved, the protocol shifts toward progressive resistance training. Open kinetic chain quadriceps strengthening is introduced cautiously, monitoring for patellofemoral irritability. Agility drills, plyometrics, and sport-specific training are gradually integrated. Throughout this entire continuum
