Anatomic Allograft Reconstruction for Engaging Hill-Sachs Lesions: An Intraoperative Masterclass
Key Takeaway
This masterclass provides an exhaustive, real-time guide to anatomic allograft reconstruction for symptomatic, engaging Hill-Sachs lesions. We cover comprehensive anatomy, meticulous preoperative planning, and granular intraoperative execution, from initial incision to definitive graft fixation. Fellows will gain insights into managing glenohumeral instability, preventing pitfalls, and ensuring optimal patient outcomes through detailed surgical directives and postoperative strategies.
Comprehensive Introduction and Patho-Epidemiology
Welcome, fellows and colleagues, to the operating theater. Today, we are undertaking a highly complex and critical procedure in the realm of shoulder instability: the management of a symptomatic, engaging Hill-Sachs lesion presenting with a profound articular arc deficit. This is unequivocally not your routine arthroscopic Bankart repair. We are fundamentally addressing severe structural pathology to restore the native architecture of the humeral head, thereby preventing recurrent instability and mitigating catastrophic joint impingement. We will be performing an anatomic osteoarticular allograft reconstruction, a sophisticated technique that eliminates the mechanical defect while preserving essential glenohumeral kinematics.
Glenohumeral instability remains one of the most common orthopedic presentations, yet the nuance of the "engaging" Hill-Sachs lesion is frequently underappreciated in primary management. When the shoulder dislocates anteriorly, the posterolateral aspect of the humeral head impacts violently against the dense cortical bone of the anterior glenoid rim. This traumatic collision generates a compression fracture on the humeral head—the classic Hill-Sachs lesion. The critical biomechanical distinction, however, lies in whether this resultant defect "engages" the anterior glenoid rim during functional arcs of motion.

An engaging Hill-Sachs lesion is specifically defined by its behavior when the shoulder is placed in a functional position of abduction and external rotation (ABER). In this provocative position, the long axis of the humeral defect aligns parallel to the anterior glenoid rim. This geometric alignment allows the glenoid rim to physically "drop into" the Hill-Sachs void, acting as a cam mechanism that levers the humeral head out of the glenoid fossa, culminating in a sensation of severe subluxation or frank dislocation. This mechanical engagement is a primary driver of surgical failure in isolated soft-tissue stabilization procedures.

While Hill-Sachs lesions are ubiquitous—occurring in 32% to 51% of primary anterior dislocations and approaching 100% in recurrent cases—the vast majority are superficial, non-engaging, and clinically silent. However, lesions involving 25% to 30% or more of the humeral articular arc create a critical "articular arc length mismatch." In these scenarios, even an anatomically perfect soft-tissue Bankart repair will fail because the irregular contour of the humeral head mechanically overrides the repaired labrum. Addressing this structural deficit directly through anatomic bony reconstruction is paramount to restoring stability, preventing early-onset osteoarthritis, and returning the high-demand patient to unrestricted function.
Detailed Surgical Anatomy and Biomechanics
To master the anatomic allograft reconstruction, one must possess a profound understanding of glenohumeral biomechanics, specifically the interplay between humeral head defects and glenoid bone loss. The concept of the "glenoid track," elegantly described by Yamamoto and Itoi, is the cornerstone of modern instability surgery. The glenoid track is defined as the contact zone between the glenoid and the humeral head as the arm is elevated in maximum external rotation. If the medial margin of a Hill-Sachs lesion extends medially beyond the medial margin of the glenoid track, the lesion is "off-track" and will engage the anterior glenoid rim.
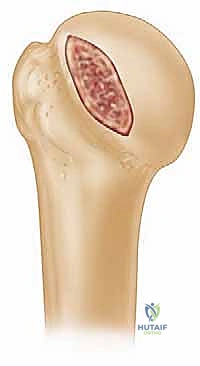
The native humeral head is not a perfect sphere; it possesses a specific radius of curvature that articulates congruently with the shallow glenoid fossa. A large Hill-Sachs lesion disrupts this congruency, effectively shortening the articular arc length of the humeral head. When the arm rotates externally, this shortened arc reaches the anterior glenoid rim prematurely. This articular arc length mismatch, initially championed by Burkhart, explains why soft-tissue repairs fail in the presence of large bony defects: the repair simply cannot compensate for the missing osseous fulcrum.
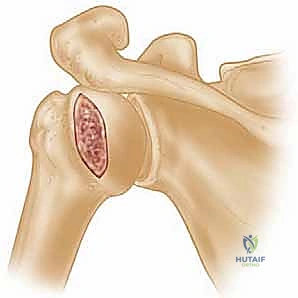
Furthermore, we must consider the phenomenon of "bipolar bone loss." A humeral head defect rarely exists in isolation; it is frequently accompanied by attritional bone loss of the anterior inferior glenoid. This glenoid bone loss effectively narrows the width of the glenoid track, making even a moderately sized Hill-Sachs lesion more likely to engage. Therefore, the biomechanical goal of our allograft reconstruction is not merely to fill a hole, but to restore the native radius of curvature of the humeral head, effectively converting an "off-track" lesion back into an "on-track" lesion, thereby restoring the normal articular arc and preventing mechanical engagement.
The structural integrity of the subscapularis and the anterior capsule also plays a vital role in this biomechanical equation. During our surgical approach, meticulous management of these anterior structures is critical. While procedures like the remplissage address the Hill-Sachs lesion by tenodesing the infraspinatus into the defect (effectively making it an extra-articular lesion), this non-anatomic solution can restrict external rotation. In contrast, the anatomic osteoarticular allograft reconstruction preserves native rotational kinematics by restoring the true spherical geometry of the humeral head, making it the superior biomechanical choice for large, engaging defects in high-demand patients.
Exhaustive Indications and Contraindications
Patient selection is the absolute bedrock of success for this complex procedure. Not every Hill-Sachs lesion, even if large, mandates an osteoarticular allograft. We reserve this operation for a highly specific subset of patients who exhibit clear clinical and radiographic evidence of symptomatic engagement that cannot be managed through less invasive means. The primary indication is ongoing symptomatic anterior glenohumeral instability—manifesting as pain, clicking, catching, or profound apprehension—in a patient with a large, engaging Hill-Sachs lesion that involves more than 25% to 30% of the articular surface.
A critical indication is the failure of previous soft-tissue stabilization procedures where an unrecognized or underappreciated bony defect was the primary cause of failure. Furthermore, patients with high-risk profiles for redislocation, such as those with poorly controlled seizure disorders (who often present with massive, atypical defects) or elite contact athletes with combined bipolar bone loss, may be considered for primary allograft reconstruction to minimize the risk of catastrophic failure and expedite return to sport.
Conversely, the contraindications must be strictly respected to avoid devastating complications. Routine medical comorbidities precluding safe general anesthesia or prolonged surgical times are absolute contraindications. Active or recent glenohumeral infection is an absolute contraindication, given the massive avascular bone graft being implanted. Furthermore, patients with non-engaging or functionally silent Hill-Sachs lesions, or those who have responded well to conservative management or isolated soft-tissue repair, should not be subjected to the morbidity of this open reconstructive procedure. Advanced glenohumeral osteoarthritis is also a contraindication, as these patients are better served by arthroplasty.
| Category | Specific Criteria | Clinical Rationale |
|---|---|---|
| Primary Indications | Symptomatic engaging Hill-Sachs lesion > 25-30% of articular arc. | High risk of failure with isolated soft-tissue repair due to mechanical override. |
| Failed prior soft-tissue stabilization (Bankart) with persistent engagement. | Bony architecture must be restored to provide a stable fulcrum. | |
| "Off-track" lesions in high-demand contact athletes or seizure patients. | High forces require anatomic restoration to prevent catastrophic redislocation. | |
| Relative Indications | Combined bipolar bone loss (glenoid + humeral head). | Reconstructing the humeral side may preclude the need for a Latarjet in specific cases. |
| Absolute Contraindications | Active or suspected glenohumeral joint infection. | High risk of deep-seated infection of the massive allograft. |
| Advanced glenohumeral osteoarthritis (Kellgren-Lawrence Grade 3 or 4). | Joint preservation is futile; arthroplasty is the definitive treatment. | |
| Medically unfit for prolonged open surgery/general anesthesia. | Unacceptable perioperative morbidity and mortality risk. | |
| Relative Contraindications | Non-compliant patients unable to follow strict postoperative rehabilitation. | High risk of graft displacement, hardware failure, or severe stiffness. |
| Small, non-engaging, or "on-track" Hill-Sachs lesions. | Morbidity of open allograft outweighs the benefits; arthroscopic management suffices. |
Pre-Operative Planning, Templating, and Patient Positioning
The success of an anatomic allograft reconstruction is largely determined before the first incision is ever made. Meticulous preoperative planning is mandatory, heavily relying on advanced cross-sectional imaging. While standard radiographs, including the Stryker notch and true AP views, provide a baseline understanding of the defect, they are insufficient for precise surgical templating.
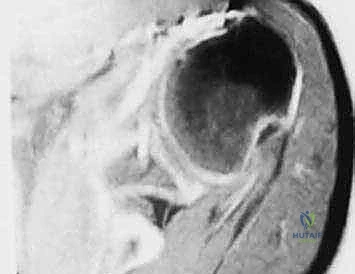
High-resolution CT scanning with 3D reconstructions is the gold standard for quantifying the volume, depth, and orientation of the Hill-Sachs lesion. We utilize these 3D models to calculate the exact percentage of articular surface involvement and to assess the glenoid track. MRI is also essential to evaluate the integrity of the rotator cuff, the anterior labrum, and the capsular tissues, which will need to be addressed concurrently during the open procedure.
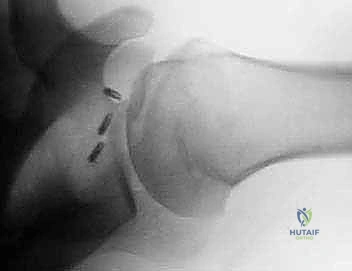
Procurement of the correct allograft is perhaps the most critical logistical step. We mandate the use of a fresh-frozen, cryopreserved osteoarticular humeral head allograft obtained from a certified tissue bank. The allograft must be size-matched to the patient's native humeral head to ensure the radius of curvature is perfectly recreated. We provide the tissue bank with the patient's CT data to ensure precise matching. We strictly avoid irradiated grafts, as the radiation process significantly degrades the biomechanical properties of the bone, increasing the risk of structural collapse and hardware failure upon aggressive impaction and cyclic loading.
In the operating room, the patient is placed in a highly modified beach chair position, elevated to approximately 45 to 60 degrees. The head is meticulously secured in a padded Mayfield or similar headrest, ensuring the cervical spine remains in neutral alignment to prevent catastrophic neurapraxia. The operative arm must be draped free, utilizing a sterile arm positioner that allows for dynamic intraoperative manipulation—specifically, the ability to place the arm in full abduction and external rotation to dynamically assess graft engagement. Fluoroscopy is positioned to allow for immediate, unobstructed AP and axillary views, which are critical for verifying screw trajectory and confirming that the hardware does not breach the articular surface.
Step-by-Step Surgical Approach and Fixation Technique
We begin with a standard extended deltopectoral approach. The skin incision starts just lateral to the tip of the coracoid process and extends distally toward the deltoid tuberosity, following the deltopectoral groove. We meticulously identify the cephalic vein, typically retracting it laterally with the deltoid to preserve its venous drainage, though medial retraction is acceptable depending on the specific venous branching pattern. The clavipectoral fascia is incised, and the conjoined tendon is identified and retracted medially. Care must be taken to identify and protect the musculocutaneous nerve entering the medial aspect of the conjoined tendon.
Exposure of the proximal humerus requires careful management of the subscapularis. Depending on the size of the patient and the exact location of the defect, we perform either a lesser tuberosity osteotomy or a vertical subscapularis tenotomy, leaving a robust lateral stump for later repair. The anterior capsule is then incised vertically, creating a T-capsulotomy to allow for excellent visualization of the glenohumeral joint. The arm is then externally rotated and extended to deliver the posterolateral humeral head—and the engaging Hill-Sachs lesion—into the anterior surgical window.
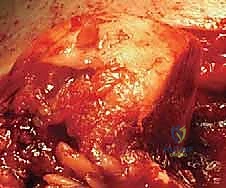
Once the defect is fully visualized, we prepare the recipient bed. Using a high-speed burr and sharp osteotomes, we debride the sclerotic base of the Hill-Sachs lesion down to healthy, bleeding cancellous bone. The margins of the defect are squared off to create a geometric, uniform recipient site that will intimately accept the allograft. This step is critical; a poorly prepared bed will lead to fibrous non-union and eventual graft failure. We then measure the precise dimensions of the prepared defect using a sterile caliper.
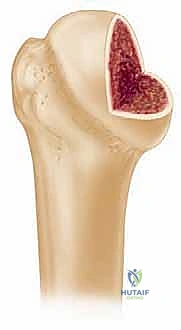
Attention is then turned to the back table for allograft preparation. The fresh-frozen humeral head allograft is thawed in warm saline. Using the measurements obtained from the recipient bed, we use an oscillating saw to harvest a precisely matched osteoarticular block from the corresponding anatomical location on the allograft humeral head. The goal is a "press-fit" matching of the graft's radius of curvature to the patient's native anatomy. The graft is repeatedly trialed in the recipient bed, and fine adjustments are made with a rasp or burr until a flawless, flush articular fit is achieved.
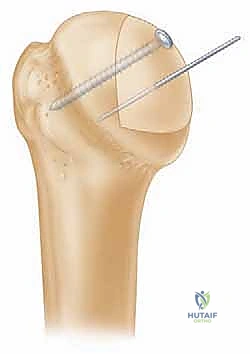
With the graft perfectly seated, we proceed with rigid internal fixation. We typically utilize two or three headless compression screws (usually 3.0mm or 3.5mm), placed perpendicular to the graft-host interface. Guide pins are placed first, and their trajectory is confirmed via intraoperative fluoroscopy to ensure they do not breach the far cortex or the articular surface. The screws are then advanced, ensuring the heads are countersunk at least 2mm below the articular cartilage of the graft to prevent devastating postoperative glenoid abrasion.
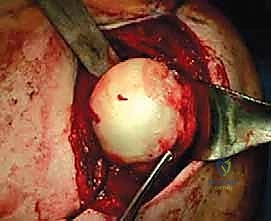
Following fixation, the arm is taken through a full, dynamic range of motion, specifically into the provocative ABER position. We must visually confirm that the reconstructed humeral head articulates smoothly with the glenoid and that there is absolutely no residual engagement.
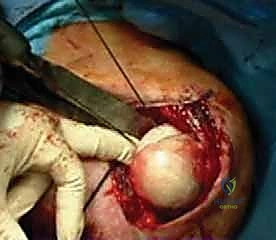
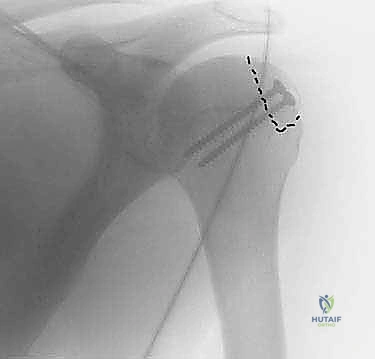
Once stability and smooth kinematics are confirmed, we irrigate the joint copiously. The anterior capsule is meticulously repaired, often with an inferior capsular shift if concurrent capsular laxity is present. The subscapularis is repaired anatomically using heavy non-absorbable sutures through transosseous tunnels or suture anchors. A final radiographic check is performed before routine layered closure of the deltopectoral interval, subcutaneous tissues, and skin.
Complications, Incidence Rates, and Salvage Management
While anatomic allograft reconstruction is highly effective, it is a massive open procedure carrying significant risks. The surgeon must be acutely aware of potential complications and possess the technical armamentarium to manage them. The most devastating complication is deep infection, which, while rare (<2%), often necessitates complete removal of the allograft, thorough debridement, and placement of an antibiotic spacer, effectively destroying the joint.
Graft resorption and partial collapse are recognized complications, occurring in approximately 5% to 10% of cases, particularly if irradiated grafts are erroneously used or if initial fixation was inadequate. Minor resorption may be asymptomatic, but significant collapse leads to recurrent articular arc mismatch and secondary osteoarthritis. Hardware complications, such as screw back-out or intra-articular penetration, occur in roughly 2% to 5% of cases. If a headless compression screw is not adequately countersunk, it will rapidly abrade the native glenoid cartilage, leading to catastrophic chondrolysis.
Postoperative stiffness is perhaps the most common complication, affecting up to 15% of patients. This is typically secondary to prolonged immobilization or aggressive capsular plication during the closure. While most cases resolve with aggressive, phased physical therapy, recalcitrant stiffness may require arthroscopic capsular release. Avascular necrosis (AVN) of the native humeral head is a rare but severe complication (<1%) related to excessive disruption of the vascular supply during the surgical approach and defect preparation.
| Complication | Estimated Incidence | Pathophysiology / Risk Factors | Salvage Management Strategy |
|---|---|---|---|
| Postoperative Stiffness | 10% - 15% | Over-tensioning of subscapularis/capsule; poor rehab compliance. | Aggressive physical therapy; arthroscopic capsular release if refractory > 6 months. |
| Graft Resorption / Collapse | 5% - 10% | Use of irradiated grafts; inadequate initial press-fit; biologic failure. | Observation if asymptomatic; revision bone grafting or arthroplasty if symptomatic/unstable. |
| Hardware Prominence / Chondrolysis | 2% - 5% | Failure to countersink screws >2mm; graft collapse exposing hardware. | Immediate arthroscopic hardware removal; microfracture/chondral procedures for glenoid damage. |
| Deep Joint Infection | < 2% | Contaminated allograft; prolonged open surgical time; patient comorbidities. | Urgent I&D, complete allograft/hardware removal, antibiotic spacer, long-term IV antibiotics. |
| Recurrent Instability | 2% - 4% | Unaddressed glenoid bone loss; undersized graft failing to restore arc. | Revision stabilization (Latarjet) if glenoid deficient; revision allograft if humeral side failed. |
| Avascular Necrosis (AVN) | < 1% | Excessive soft tissue stripping around the anatomic neck during exposure. | Humeral head resurfacing or total shoulder arthroplasty depending on patient age and cuff status. |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following an osteoarticular allograft reconstruction is a delicate balance between protecting the healing bony integration and preventing debilitating arthrofibrosis. The protocol must be strictly phased and closely monitored by both the surgical team and the physical therapist.
Phase I: Protection and Early Passive Motion (Weeks 0-4)
Immediately postoperatively, the patient is placed in a specialized shoulder immobilizer with a small abduction pillow to relieve tension on the repaired subscapularis and anterior capsule. Absolute protection of the subscapularis repair is paramount. Pendulum exercises and strictly passive range of motion (PROM) are initiated on postoperative day one. Forward elevation is typically limited to 90 degrees, and external rotation is strictly limited to neutral (0 degrees) to protect the anterior soft tissue repairs and avoid stressing the graft-host interface.
Phase II: Active-Assisted Motion and Weaning (Weeks 4-8)
At four weeks, assuming radiographic evidence of stable hardware and early graft incorporation, the sling is gradually weaned. Active-assisted range of motion (AAROM) is initiated. Forward elevation is progressed to full as tolerated. External rotation is slowly advanced, aiming for 30 degrees by week 6. Submaximal, pain-free isometric exercises for the deltoid and rotator cuff are introduced, strictly avoiding internal rotation isometrics to protect the healing subscapularis.
Phase III: Active Motion and Early Strengthening (Weeks 8-12)
By week eight, the patient should have achieved nearly full active range of motion (AROM). The focus shifts to progressive isotonic strengthening of the rotator cuff and periscapular stabilizers. Light resistance bands and light weights are utilized. Scapular dyskinesia, which is common following prolonged immobilization, must be aggressively addressed. Radiographs are obtained at 12 weeks to confirm complete bony union of the allograft.
Phase IV: Advanced Strengthening and Return to Sport (Months 3-6+)
Once bony union is confirmed and full, painless AROM is achieved, advanced strengthening and plyometric exercises are initiated. For athletes, sport-specific functional training begins. Return to contact sports or heavy manual labor is generally prohibited until at least 6 months postoperatively, and only after the patient demonstrates symmetrical strength, normal kinematics, and absolute confidence in the shoulder without apprehension.
Summary of Landmark Literature and Clinical Guidelines
The evolution of anatomic allograft reconstruction is heavily grounded in the biomechanical principles established by several landmark studies. Burkhart and De Beer's seminal work on the "engaging" Hill-Sachs lesion fundamentally shifted the paradigm, demonstrating that soft-tissue repairs fail predictably in the presence of significant unaddressed bony defects. This established the absolute necessity of evaluating the articular arc length.
The concept was further refined by Yamamoto and Itoi, who introduced the "glenoid track" paradigm. Their rigorous biomechanical studies provided surgeons with a quantifiable method (using CT or MRI) to determine whether a specific Hill-Sachs lesion will engage based on its medial extension relative to the native glenoid width and any concurrent anterior glenoid bone loss. The glenoid track formula is now the standard of care for preoperative templating.
Clinical outcomes of osteoarticular allograft reconstruction have been extensively documented by authors such as Miniaci and Provencher. Miniaci's early series demonstrated excellent restoration of stability and high rates of return to sport in patients with massive defects, establishing the procedure's viability. Provencher's subsequent systematic reviews and long-term follow-up studies have confirmed that when performed with meticulous technique and strict patient selection, fresh-frozen allograft reconstruction yields reliable graft incorporation, durable stability, and prevents the progression of instability-arthropathy, cementing its place as a master-level technique in the orthopedic surgeon's armamentarium.
